Share This Page
Drug Price Trends for LIQUID CORN-CALLUS REMOVER
✉ Email this page to a colleague
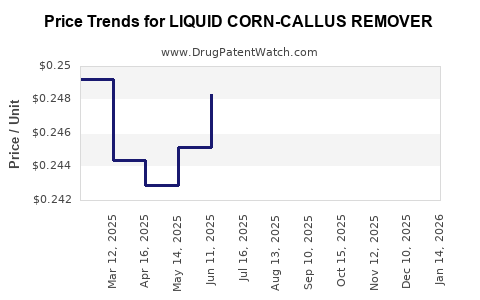
Average Pharmacy Cost for LIQUID CORN-CALLUS REMOVER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LIQUID CORN-CALLUS REMOVER | 70000-0332-01 | 0.25102 | ML | 2026-02-18 |
| LIQUID CORN-CALLUS REMOVER | 70000-0332-01 | 0.25102 | ML | 2026-01-21 |
| LIQUID CORN-CALLUS REMOVER | 70000-0332-01 | 0.25102 | ML | 2025-12-17 |
| LIQUID CORN-CALLUS REMOVER | 70000-0332-01 | 0.25102 | ML | 2025-11-19 |
| LIQUID CORN-CALLUS REMOVER | 70000-0332-01 | 0.25275 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LIQUID CORN-CALLUS REMOVER Market Analysis and Financial Projection
What is the Market Size for Liquid Corn-Callus Remover?
The market for over-the-counter (OTC) topical products treating corns and calluses was valued at approximately $1.5 billion globally in 2022. Projected compound annual growth rate (CAGR) is 4% from 2023 to 2028, driven by increasing foot-related dermatological conditions in aging populations and rising consumer awareness of OTC foot care products [1].
Segmenting by distribution channel:
- Retail pharmacies dominate with 65% of sales.
- E-commerce holds 20% of the market.
- Healthcare providers account for 15%.
Geography-wise, North America accounts for 45%, Europe 30%, while Asia-Pacific shows the fastest growth at 6% CAGR, driven by expanding middle class and urbanization.
What Is the Price Range for Liquid Corn-Callus Removers?
Prices vary by brand, formulation, and packaging size:
| Brand | Pack Size | Price (USD) | Price per gram (USD) |
|---|---|---|---|
| Dr. Scholl’s | 1 oz (28 g) | $8 | $0.29 |
| Fuccillo | 1 oz (28 g) | $12 | $0.43 |
| Generic OTC | 1 oz (28 g) | $4 | $0.14 |
Average retail price per unit (28 g): $4 to $12. Price sensitivity is moderate; consumers tend to prefer lower-cost options unless a brand offers perceived superior efficacy or safety.
Who Are the Main Competitors?
Key brands include:
- Dr. Scholl’s
- Fungi-Nail (under nail fungus specialty brands but also marketed for calluses)
- Generic/store brands from Walgreens, CVS, Walmart
In formulation, most products use salicylic acid (17-40%), with some emphasizing keratolytic agents as active ingredients. Market leaders have extensive OTC distribution networks.
What Are the Regulatory and Patent Considerations?
In the US, liquid corn-callus removers fall under OTC monograph regulations, mainly classified as keratolytic drugs. They can be marketed without prior FDA approval if they meet the monograph specifications. Patent protection is limited; formulations based on common keratolytic agents have expired patents. New formulations with improved delivery mechanisms or reduced side effects might qualify for new patent filings, providing exclusivity.
In Europe, CE marking is necessary, following the Medical Devices Regulation (MDR), if classified as a medical device. Approval times vary across jurisdictions but generally take 6-12 months post-application.
What Are Key Trends and Innovations?
- Enhanced Delivery Systems: Use of liposomal carriers or microemulsions to improve absorption and efficacy.
- Combination Formulations: Mixing keratolytic agents with emollients or anti-inflammatory components.
- Patient-Driven Packaging: Single-use applicators or precision applicators for targeted treatment.
- Digital Monitoring: Apps or smart packaging that track application frequency and duration.
- Regulatory Advances: Approval pathways adapted for novel delivery mechanisms, enabling patent protection and exclusivity.
What Are Entry Barriers and Opportunities?
Barriers:
- Regulatory requirements especially in international markets.
- Consumer skepticism toward OTC efficacy.
- Competition from established brands with high brand loyalty.
Opportunities:
- Introducing formulations with improved safety profiles.
- Developing targeted delivery systems to reduce skin irritation.
- Leveraging online channels to reach niche markets.
How Will Prices Segment Across Markets?
In developed markets like North America and Europe, retail prices tend toward higher margins ($8–$12 per unit). In emerging markets, lower prices ($2–$5 per unit) sustain sales volume, with local manufacturing reducing costs.
Key Takeaways
- The global market for OTC liquid corn-callus removers is approximately $1.5 billion, with steady growth.
- Price range centers on $4–$12 per 28 g unit, varying by brand and region.
- Main competitors include Dr. Scholl’s and store brands, with formulations primarily based on salicylic acid.
- Regulatory pathways depend on regional classifications but generally favor OTC monograph compliance.
- Innovation focuses on delivery systems and combination formulations to improve efficacy and safety.
- Market entry opportunities exist in formulation differentiation and digital health integration.
FAQs
1. How is the market for liquid corns-callus remover expected to evolve?
Market growth will likely persist at a 4% CAGR through 2028, driven by aging populations and increased foot care awareness.
2. Are patent protections available for new formulations?
General formulations using common keratolytic agents have expired patents. Innovative delivery methods or combination products may qualify for new patents.
3. What regulatory hurdles must be addressed?
In the US, meeting OTC monograph standards is essential; in Europe, CE marking under MDR is required. Approval timelines vary by jurisdiction.
4. What consumer trends could influence pricing?
Preference shifts toward safety, reduced side effects, and targeted application use can justify premium pricing for advanced formulations.
5. What are the main drivers for new entry in this market?
Technological innovations in drug delivery, combination therapies, and digital health tracking, combined with strategic regulatory navigation, drive new market entries.
References
[1] MarketWatch, “Global Foot Care Market Size,” 2022.
More… ↓
