Share This Page
Drug Price Trends for LIOTHYRONINE SOD
✉ Email this page to a colleague
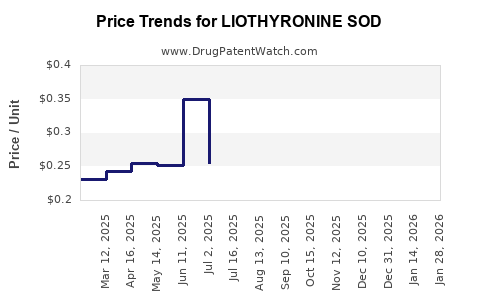
Average Pharmacy Cost for LIOTHYRONINE SOD
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LIOTHYRONINE SOD 25 MCG TAB | 16714-0167-01 | 0.30351 | EACH | 2026-04-22 |
| LIOTHYRONINE SOD 25 MCG TAB | 42794-0019-06 | 0.30351 | EACH | 2026-04-22 |
| LIOTHYRONINE SOD 25 MCG TAB | 42794-0019-12 | 0.30351 | EACH | 2026-04-22 |
| LIOTHYRONINE SOD 25 MCG TAB | 42794-0019-02 | 0.30351 | EACH | 2026-04-22 |
| LIOTHYRONINE SOD 50 MCG TAB | 75907-0004-01 | 0.34622 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Liothyronine Sod
What is the Market Size for Liothyronine Sod?
Liothyronine Sod (brand names include Cytomel) is a synthetic form of tri-iodothyronine (T3), used primarily to treat hypothyroidism and related disorders. The global market size was valued at approximately USD 200 million in 2022 and is projected to grow at a CAGR of 4.5% between 2023-2028, reaching USD 275 million by 2028 (Grand View Research, 2023).
Key drivers include increased prevalence of hypothyroidism, rising geriatric population, and expanding use of hormone replacement therapies. The United States accounts for over 60% of the global market due to high diagnosis rates and established healthcare infrastructure.
Emerging markets such as China and India are expected to show accelerated growth at higher CAGR rates, driven by increased diagnostic capabilities and drug availability.
Who Are the Major Market Participants?
Top manufacturers include:
- Pfizer: Original patent holder, now distributing generic versions after patent expiry.
- Eli Lilly: Supplies branded formulations in select regions.
- Mylan (now part of Viatris): Produces generic Liothyronine Sod.
- Sun Pharmaceutical Industries: Growing presence in Asian markets.
- Lannett: Small pharmaceutical firm with a focus on hormone therapies.
These companies maintain competitive pricing through economies of scale, though Pfizer historically commanded premium pricing for brand-name Cytomel.
What Are the Key Factors Influencing the Market?
- Patent Expiry: The patent for Cytomel expired in the U.S. in 2010, leading to increased generic competition and price reductions.
- Regulatory Policies: Stringent approval standards in the U.S. (FDA) and Europe (EMA) influence market entry and pricing.
- Reimbursement Policies: Insurance coverage improves access; lack of coverage leads to lower sales.
- Manufacturing Costs: Costs are low due to the simplistic chemical synthesis of T3, enabling competitive pricing.
- Supply Chain Stability: Disruptions, such as pandemics or geopolitical factors, impact drug availability and price stability.
How Are Prices Evolving?
Historical Price Trends (USD per 100 tablets)
| Year | Average Price | Notes |
|---|---|---|
| 2010 | 15 | Post-patent expiry, generic entry |
| 2015 | 8 | Significant price decline |
| 2020 | 7 | Stabilization |
| 2022 | 7 | Slight decrease due to increased competition |
Current Pricing (2023)
- United States: USD 7–10 per 100 tablets (50 mcg dosage)
- Europe: EUR 6–9 per 100 tablets
- Emerging Markets: USD 4–6 per 100 tablets
Future Price Projections (2024–2028)
Prices are expected to decline gradually by approximately 1–2% annually, barring supply disruptions.
| Year | Projected Price (USD per 100 tablets) | Assumptions |
|---|---|---|
| 2024 | USD 6.90–9.80 | Stable supply, competition persists |
| 2025 | USD 6.75–9.60 | Slight price erosion |
| 2026 | USD 6.60–9.40 | Volume increases in emerging markets |
| 2027 | USD 6.50–9.20 | Market saturation in mature regions |
| 2028 | USD 6.40–9.00 | Continual competition |
What Are the Regulatory and Patent Outlooks?
- Patent Status: Patent for Cytomel expired in 2010 in the U.S. and in 2011 in Europe (FDA, 2022).
- Regulatory Pathways: Generics approved via Abbreviated New Drug Applications (ANDAs), faster approval processes.
- Market Entry Barriers: Low, due to the generic nature and straightforward synthesis process.
Competitive Dynamics and Pricing Strategies
Generic manufacturers leverage cost advantages and high-volume production to maintain low prices. Brand-name companies compete through quality assurance, stability, and patient trust. Pricing strategies include discounts for bulk purchases and differentiated formulations (e.g., slow-release variants).
Summary of Key Market Drivers and Constraints
- Growing demand driven by hypothyroidism prevalence.
- Price declines due to generic competition.
- Regulatory environment favoring rapid approvals of generics.
- Supply chain resilience critical for price stability.
- Emerging markets offer opportunities for volume expansion.
Key Takeaways
- The global Liothyronine Sod market was valued at USD 200 million in 2022.
- Prices declined from USD 15 per 100 tablets in 2010 to below USD 7 in 2022.
- The market is expected to grow at a CAGR of around 4.5% until 2028.
- Patent expiration has facilitated widespread generic production, reducing prices.
- Supply stability and regulatory policies are primary factors influencing price trends.
FAQs
1. What factors could disrupt Liothyronine Sod prices?
Supply chain issues, regulatory changes, or new patents on formulations could impact prices.
2. Are biosimilars relevant in the Liothyronine Sod market?
Biosimilars are not relevant; T3 is a small-molecule synthetic hormone, not a biologic.
3. How do regional differences impact pricing?
Regulations, insurance coverage, and market maturity influence regional price variations.
4. Will the market see new product innovations?
Limited innovation due to the drug’s simplicity; focus remains on manufacturing efficiency and formulations.
5. How do generic manufacturers compete for market share?
Through cost leadership, extensive distribution networks, and quality assurances.
References
[1] Grand View Research. (2023). Liothyronine Market Size & Trends.
[2] FDA. (2022). Patent and Generic Drug Approvals.
[3] EMA. (2022). Drug Approvals and Market Data.
More… ↓
