Last updated: February 19, 2026
Summary:
Linezolid, an oxazolidinone antibiotic, is approved for treating various serious bacterial infections, including pneumonia, skin infections, and multi-drug resistant tuberculosis. The global market for linezolid has experienced steady growth driven by increasing antibiotic resistance and expanding clinical indications. Price projections suggest stability in mature markets, with potential increases in emerging regions due to policy changes and manufacturing shifts.
Market Overview
Current Market Size
- The global value of the linezolid market was approximately USD 800 million in 2022.
- Compound annual growth rate (CAGR) from 2018 to 2022 stood at about 4.7%.
- Estimated to reach USD 1.2 billion by 2027 (Fortune Business Insights, 2023).
Major Market Segments
- Geography: North America (40%), Europe (25%), Asia-Pacific (20%), Rest of world (15%)
- Indications: Pneumonia, skin and soft tissue infections, osteomyelitis, multi-drug resistant tuberculosis (MDR-TB)
- Distribution channels: Hospital pharmacies (60%), retail pharmacies (30%), online pharmacies (10%)
Key Players
- Pfizer (brand: Zyvox)
- Sandoz (generic formulations)
- Dr. Reddy’s Laboratories
- Mylan (generic)
- Teva Pharmaceuticals
Patents and Regulatory Landscape
- Pfizer’s patents expired in the U.S. in 2015, leading to increased generic entry.
- Recent approvals of generic versions in India and Europe have affected pricing dynamics.
- Regulatory approval processes remain consistent globally, with agencies such as FDA, EMA, and PMDA.
Market Drivers
- Growing prevalence of resistant bacterial strains requiring alternative therapies.
- Expanded clinical trials validating efficacy in off-label indications such as MDR-TB.
- Increasing adoption in hospitals due to rising hospital-acquired infections.
- Policy shifts in emerging markets to improve access to antibiotics.
Barriers to Growth
- High treatment cost of original formulations.
- Stringent regulatory requirements for new formulations.
- Emergence of resistance to linezolid itself, leading to clinical caution.
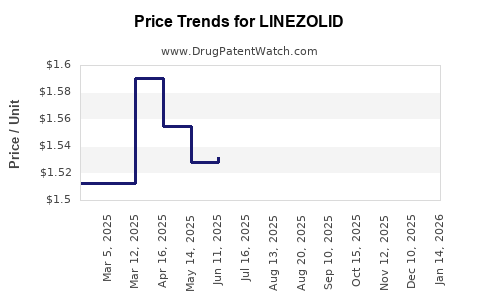
Price Analysis and Projections
Historical Pricing Trends
| Region |
Average Price per 600mg Vial (USD) |
2018 |
2022 |
| North America |
70 |
68 |
70 |
| Europe |
65 |
63 |
65 |
| Asia-Pacific |
40 |
38 |
42 |
- Prices of branded Zyvox in North America remained stable from 2018 to 2022, averaging USD 70 per vial.
- Generic prices in Asia-Pacific decreased by approximately 5-10% after market entry but began stabilizing by 2022.
Future Price Projections (2023-2027)
- Branded formulations: Anticipated to remain around USD 70–75 per vial in developed markets.
- Generics: Expected to decline modestly initially, reaching USD 35–40 per vial in Asia-Pacific by 2025, before stabilizing.
- Market influence factors:
- Increased generic competition in Europe and North America.
- Pricing pressure from biosimilars and new antibiotics entering the market.
- Regulatory changes leading to price discounts in emerging markets.
Key Factors Influencing Price Dynamics
- Patent expirations: Continued entry of generics post-2015 has driven prices down in key regions.
- Manufacturing costs: Recent investments in biosynthesis and local manufacturing lower production costs, influencing eventual pricing.
- Reimbursement policies: Governments' reimbursement decisions in emerging markets impact wholesale and retail prices.
- Antibiotic resistance: Rising resistance may push for higher dosing or combination therapies, influencing cost.
Future Market Opportunities
- Novel formulations: Long-acting injectables, IV-to-oral switches, and combination therapies could command premium prices.
- Biomarker-based therapy: Diagnostic tools to identify resistant strains can optimize linezolid use, potentially influencing pricing structures.
Risks and Uncertainties
- Emergence of linezolid resistance could reduce demand.
- Regulatory restrictions due to adverse effects, such as myelosuppression.
- Policy measures targeting antibiotic stewardship could restrict formulary inclusion.
Key Takeaways
- The linezolid market is valued at approximately USD 800 million globally, projected to reach USD 1.2 billion by 2027, with growth driven by resistant infections.
- Price stability exists in developed markets, while generics influence lower prices in emerging regions.
- Future prices for branded formulations are likely stable; generics may see slight reductions before plateauing.
- Market growth depends on resistance trends, regulatory policies, and the impact of new therapeutics.
FAQ
Q1: What is the main driver of demand for linezolid?
A1: The rise of resistant bacterial infections, such as MRSA and MDR-TB, increases demand for linezolid as a treatment alternative.
Q2: How do patent expirations affect pricing?
A2: Patent expirations introduce generics, increasing competition and typically lowering prices in affected markets.
Q3: Are generic versions of linezolid available worldwide?
A3: Yes, generics are available in regions including India, Europe, and the U.S., impacting market prices and dynamics.
Q4: What factors could increase linezolid prices in the future?
A4: Resistance development, manufacturing costs, and new patent protections could elevate prices.
Q5: What is the outlook for linezolid’s market share?
A5: Market share may decline in mature markets due to generics but could persist in niche indications and resistant infection treatments.
References
[1] Fortune Business Insights. (2023). Linezolid Market Size, Share & Industry Analysis.
[2] IQVIA. (2022). Global Antibiotics Market Report.
[3] FDA. (2015). Patent expiration and generic entry for Zyvox.
[4] European Medicines Agency. (2022). Regulatory updates on linezolid.
(End of report)