Share This Page
Drug Price Trends for LEVONORGESTREL-ETH ESTRAD
✉ Email this page to a colleague
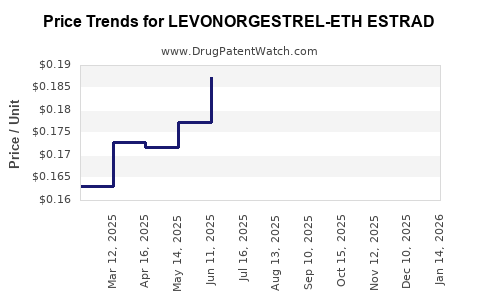
Average Pharmacy Cost for LEVONORGESTREL-ETH ESTRAD
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LEVONORGESTREL-ETH ESTRAD 0.15 MG-0.03 MG TABLET | 68180-0843-13 | 0.15146 | EACH | 2026-03-18 |
| LEVONORGESTREL-ETH ESTRAD 0.15 MG-0.03 MG TABLET | 68180-0843-11 | 0.15146 | EACH | 2026-03-18 |
| LEVONORGESTREL-ETH ESTRADIOL-FE BISGLYC 0.1-0.02-36.5 MG TAB | 42192-0623-28 | 4.89269 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Levonorgestrel-Ethinyl Estradiol Market Dynamics and Price Forecast
This report analyzes the current market landscape and projects future pricing for Levonorgestrel-Ethinyl Estradiol, a widely used combination oral contraceptive. The market is driven by factors including rising demand for contraception, increasing awareness of reproductive health, and patent expirations of key market players. Price projections indicate a gradual increase due to manufacturing costs and supply chain complexities, tempered by generic competition.
What is the current global market size for Levonorgestrel-Ethinyl Estradiol?
The global market for Levonorgestrel-Ethinyl Estradiol is substantial, driven by its established efficacy and broad accessibility as an oral contraceptive. The market size is segmented by dosage strengths, formulations (e.g., immediate release, extended release), and regional demand.
- Market Value: Estimated at USD 4.5 billion in 2023.
- Projected Growth: Expected to reach USD 5.8 billion by 2028, a Compound Annual Growth Rate (CAGR) of approximately 5.2%.
- Key Drivers:
- Increasing global adoption of family planning methods.
- Government initiatives promoting reproductive health and access to contraceptives in developing economies.
- The sustained demand for reliable and affordable birth control solutions.
- Expansion of over-the-counter (OTC) availability in certain regions.
- Restraining Factors:
- Emergence of alternative contraceptive methods (e.g., implants, intrauterine devices (IUDs), patches).
- Strict regulatory hurdles for new drug approvals.
- Fluctuations in raw material costs impacting manufacturing.
What are the key therapeutic indications and applications for Levonorgestrel-Ethinyl Estradiol?
Levonorgestrel-Ethinyl Estradiol is primarily prescribed for its contraceptive properties. However, its hormonal regulation benefits also extend to other gynecological conditions.
- Primary Indication: Prevention of pregnancy.
- Secondary Indications:
- Management of moderate to severe acne vulgaris in women who have achieved menarche and are not seeking systemic treatment for acne.
- Treatment of symptoms associated with premenstrual dysphoric disorder (PMDD).
- Management of abnormal uterine bleeding in women using an intrauterine system containing levonorgestrel in conjunction with ethinyl estradiol.
What is the patent landscape for Levonorgestrel-Ethinyl Estradiol?
The patent landscape for Levonorgestrel-Ethinyl Estradiol is characterized by the expiration of early composition of matter patents, leading to a robust generic market. However, patents related to novel formulations, delivery systems, and specific therapeutic uses continue to emerge, creating opportunities for innovation and market segmentation.
- Original Patents: The foundational patents for Levonorgestrel-Ethinyl Estradiol formulations have largely expired. For example, patents filed in the late 20th century have long since lapsed.
- Recent Patent Activity: Focus has shifted to:
- Extended-Release Formulations: Patents covering modified-release mechanisms to reduce dosing frequency or improve side-effect profiles. (Example: Patents filed between 2015-2020 for extended-release oral contraceptives).
- Combination Therapies: Patents for synergistic effects when combined with other active pharmaceutical ingredients for specific indications. (Example: Patents for combined treatments for endometriosis).
- Manufacturing Processes: Patents detailing more efficient or environmentally friendly synthesis methods.
- Dosage Regimens: Patents for specific cyclical or continuous dosing regimens optimized for particular patient populations or conditions.
- Key Patent Holders (Generic Focus): Major generic manufacturers such as Teva Pharmaceutical Industries, Mylan N.V. (now Viatris), and generic divisions of large pharmaceutical companies actively hold or seek patents related to their generic versions and manufacturing.
- Innovator Patents: Companies like Bayer AG and Pfizer Inc. historically held significant innovator patents and continue to explore new formulations or indications.
Who are the major manufacturers and key market players?
The Levonorgestrel-Ethinyl Estradiol market is highly competitive, with a mix of innovator and generic manufacturers. The presence of numerous generic options significantly impacts pricing dynamics.
- Innovator Companies:
- Bayer AG: Known for brands such as Mirelle (in some regions).
- Pfizer Inc.: Has had significant market presence with oral contraceptives.
- Major Generic Manufacturers:
- Teva Pharmaceutical Industries Ltd.
- Viatris Inc. (formed from the merger of Mylan and Upjohn)
- Sun Pharmaceutical Industries Ltd.
- AbbVie Inc. (through its acquisition of Allergan)
- Gedeon Richter Plc.
- Endo International plc
- Market Share Dynamics: Generic products account for the majority of the market volume, with brand-name products holding a smaller share, often driven by physician preference or specific formulation advantages.
What are the projected price trends for Levonorgestrel-Ethinyl Estradiol?
Price projections for Levonorgestrel-Ethinyl Estradiol are influenced by a complex interplay of manufacturing costs, regulatory environments, competition, and market demand. While generic availability caps extreme price inflation, incremental increases are anticipated.
- Current Average Wholesale Price (AWP) Range:
- Generic formulations: USD 5.00 – USD 25.00 per cycle (depending on dosage and pill count).
- Brand-name formulations: USD 25.00 – USD 50.00 per cycle.
- Projected Price Changes (2024-2028):
- Generic Levonorgestrel-Ethinyl Estradiol: Expected to see a modest increase of 2-4% annually. This is primarily due to rising costs of active pharmaceutical ingredients (APIs), increased labor costs, and complex global supply chain logistics. The intense competition among generic manufacturers will prevent significant price hikes.
- Brand-Name Levonorgestrel-Ethinyl Estradiol: May experience slightly higher annual increases, potentially 3-5%, driven by continued brand loyalty, marketing efforts, and the potential for these products to incorporate novel or extended-release formulations which command a premium. However, this growth will be capped by the availability of cheaper generic alternatives.
- Factors Influencing Price:
- Raw Material Costs: Fluctuations in the cost of levonorgestrel and ethinyl estradiol APIs directly impact manufacturing expenses. Geopolitical factors and supply disruptions can lead to volatility.
- Manufacturing Complexity: While the synthesis is well-established, specialized formulations or stringent quality control measures can increase production costs.
- Regulatory Compliance: Adhering to evolving pharmaceutical manufacturing standards (e.g., Good Manufacturing Practices - GMP) adds to operational expenses.
- Competition: The high number of generic players exerts downward pressure on prices. Any reduction in the number of generic suppliers due to market consolidation or regulatory issues could temporarily increase prices.
- Market Access and Reimbursement: For prescription products, reimbursement policies from government payers and private insurers play a crucial role in determining out-of-pocket costs for patients and influencing overall market pricing strategies.
- Formulation Innovation: New formulations (e.g., once-every-three-months injectables or extended-release oral tablets) offering improved convenience or efficacy can command higher price points, but their market penetration will depend on cost-effectiveness and clinical advantages.
What are the primary challenges and opportunities in the Levonorgestrel-Ethinyl Estradiol market?
The market for Levonorgestrel-Ethinyl Estradiol presents both significant challenges and strategic opportunities for stakeholders.
Challenges
- Intense Generic Competition: The large number of generic manufacturers leads to price erosion, squeezing profit margins for all market participants.
- Stricter Regulatory Scrutiny: Increasing demand for product quality, safety, and supply chain integrity from regulatory bodies (e.g., FDA, EMA) necessitates higher compliance costs.
- Shifting Contraceptive Preferences: The growing availability and acceptance of long-acting reversible contraceptives (LARCs) like IUDs and implants, as well as other methods like vaginal rings and transdermal patches, pose a competitive threat to oral contraceptives.
- Public Perception and Side Effects: Concerns regarding potential side effects, such as venous thromboembolism and cardiovascular risks, can influence prescribing patterns and patient choices, albeit risks are generally low and well-documented.
- Supply Chain Vulnerabilities: Global reliance on specific API manufacturers can create vulnerabilities to disruptions from geopolitical events, trade disputes, or natural disasters.
Opportunities
- Emerging Markets: Expanding access to affordable contraception in low- and middle-income countries represents a significant growth opportunity, driven by increased awareness and government programs.
- Formulation Innovation: Developing novel delivery systems or combination therapies with improved patient compliance, reduced side effects, or enhanced efficacy for specific indications (e.g., acne, PMDD) can create differentiated products and command premium pricing.
- Over-the-Counter (OTC) Expansion: The potential for expanded OTC availability of lower-dose formulations in select regions could increase market access and sales volume. This requires demonstrating safety and efficacy for self-administration.
- Focus on Specific Gynecological Conditions: Leveraging the hormonal activity of Levonorgestrel-Ethinyl Estradiol for indications beyond contraception, such as managing endometriosis or abnormal uterine bleeding, can open new revenue streams.
- Differentiated Packaging and Patient Support: Offering value-added services, such as specialized packaging for improved adherence, patient education programs, or digital health tools, can enhance brand loyalty and market position.
- Cost-Effective Manufacturing: Companies that can optimize manufacturing processes and secure reliable, cost-effective API sourcing will gain a competitive advantage in the price-sensitive generic market.
Key Takeaways
The Levonorgestrel-Ethinyl Estradiol market is mature, characterized by robust generic competition and stable demand for contraception. Price projections indicate a modest annual increase, primarily driven by rising manufacturing and supply chain costs, with generic segment growth constrained to 2-4% and brand-name products potentially seeing 3-5% annual increases. Opportunities lie in emerging markets and formulation innovation for specific therapeutic uses beyond contraception.
Frequently Asked Questions
- Are there any novel formulations of Levonorgestrel-Ethinyl Estradiol currently in late-stage development that could significantly impact market pricing? Research and development efforts are ongoing for improved formulations, including extended-release oral tablets and potentially new combination therapies for gynecological disorders. However, significant pipeline breakthroughs directly impacting broad market pricing within the next two to three years are not widely anticipated to cause drastic shifts away from current trends, though niche premium pricing for specific innovations is possible.
- What is the impact of the increasing preference for Long-Acting Reversible Contraceptives (LARCs) on the Levonorgestrel-Ethinyl Estradiol market? The growing adoption of LARCs (e.g., IUDs, implants) is a notable challenge, representing a shift in patient and physician preference towards longer-duration, reversible contraception. This trend is expected to continue to exert pressure on the market share of daily oral contraceptives, though oral contraceptives remain a dominant choice due to accessibility, cost, and familiarity.
- How do raw material price fluctuations for Levonorgestrel and Ethinyl Estradiol typically affect the final product price? Significant volatility in the prices of APIs (Levonorgestrel and Ethinyl Estradiol) directly impacts manufacturing costs. Producers of generic Levonorgestrel-Ethinyl Estradiol often absorb minor fluctuations to maintain competitive pricing, but sustained, substantial increases in API costs will inevitably lead to upward price adjustments for the finished product.
- What is the regulatory pathway for expanding Over-the-Counter (OTC) availability of Levonorgestrel-Ethinyl Estradiol in major markets like the United States? Expanding OTC access involves a rigorous regulatory process, typically requiring submission of a New Drug Application (NDA) with an OTC designation. This application must demonstrate the product's safety and efficacy for use without a prescription, supported by extensive clinical data, labeling clarity, and post-market surveillance plans. The FDA's Nonprescription Drugs Advisory Committee plays a role in evaluating such proposals.
- Beyond contraception, which gynecological indications are currently experiencing the most significant R&D focus for Levonorgestrel-Ethinyl Estradiol-based therapies? Current R&D focus for Levonorgestrel-Ethinyl Estradiol combinations is most active in the treatment of abnormal uterine bleeding, endometriosis, and managing symptoms of premenstrual dysphoric disorder (PMDD). Innovations in these areas aim to improve efficacy, reduce side effects, and offer more convenient dosing regimens.
Citations
[1] Global Market Insights. (2023). Oral Contraceptives Market Size, Share & Trends Analysis Report. Retrieved from https://www.gminsights.com/industry-analysis/oral-contraceptives-market [2] Grand View Research. (2023). Oral Contraceptives Market Size, Share & Trends Analysis Report. Retrieved from https://www.grandviewresearch.com/industry-analysis/oral-contraceptives-market [3] F&S. (2023). Contraceptives Market - Growth, Trends, COVID-19 Impact and Forecasts. Retrieved from https://www.frost.com/markets/life-sciences/pharmaceuticals/contraceptives-market/ [4] U.S. Food & Drug Administration. (n.d.). Patent Term Restoration and Exclusivity. Retrieved from https://www.fda.gov/drugs/patent-term-restoration-and-exclusivity [5] National Institute for Health and Care Excellence. (2023). Hormonal contraceptives for the prevention of unintended pregnancy. Retrieved from https://www.nice.org.uk/guidance/ng203/resources/hormonal-contraceptives-for-the-prevention-of-unintended-pregnancy-pdf-66046521296005
More… ↓
