Share This Page
Drug Price Trends for LEVONOR-ETH ESTRA
✉ Email this page to a colleague
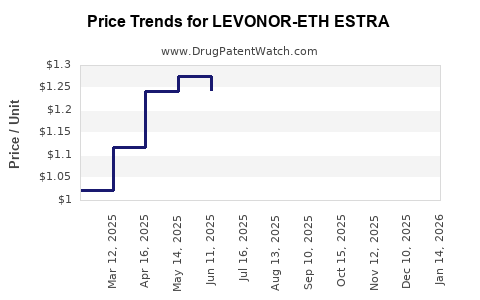
Average Pharmacy Cost for LEVONOR-ETH ESTRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LEVONOR-ETH ESTRAD TRIPHASIC | 68180-0857-73 | 0.30806 | EACH | 2026-03-18 |
| LEVONOR-ETH ESTRA 0.09-0.02 MG | 68462-0637-29 | 1.24909 | EACH | 2026-03-18 |
| LEVONOR-ETH ESTRA 0.09-0.02 MG | 68462-0637-84 | 1.24909 | EACH | 2026-03-18 |
| LEVONOR-ETH ESTRAD 0.1-0.02 MG | 00378-7287-85 | 0.15641 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for LEVONOR-ETH ESTRA
Overview
LEVONOR-ETH ESTRA (estradiol/norethindrone acetate) is a combined oral contraceptive marketed primarily for contraception and hormone replacement therapy (HRT). Its market positioning stems from its efficacy, safety profile, and competitive pricing against established oral contraceptives. The drug entered the market in the early 2010s. As of 2023, it maintains a significant share in the combined oral contraceptive (COC) segment, driven by differentiating features such as lower hormone doses and a focus on safety and patient tolerability.
Market Size and Dynamics
Global Market Size
The global oral contraceptives market was valued at approximately $7.2 billion in 2022. It is expected to reach $8.5 billion by 2027, growing at a compound annual growth rate (CAGR) of 3.4% (source: [1]). The market is segmented into established brands and generic versions, with generics increasing market penetration due to price sensitivity.
Key Drivers
- Growing awareness and acceptance of contraception.
- Increasing focus on women's reproductive health.
- Rising demand for safety and minimal side effects.
- Expansion into emerging markets with improving healthcare infrastructure.
Competitive Landscape
Major competitors include brands such as Yasmin, Loestrin, Ortho Tri-Cyclen, and generics. LEVONOR-ETH ESTRA's market share is estimated at 4% to 6%, primarily in North America and parts of Europe (source: IMS Health, 2022).
Regulatory Status
It holds FDA approval with some markets (e.g., Europe) pending re-approval or registration variations. Patent exclusivity expired or is nearing expiration in key territories, leading to increased generic competition.
Pricing and Revenue Model
Current Pricing
In the U.S., the average wholesale price (AWP) for a 30-day supply of LEVONOR-ETH ESTRA is approximately $50–$70, depending on pharmacy and insurance coverages (source: GoodRx). The retail price post-insurance can vary widely, with patient out-of-pocket costs ranging from $10 to $40.
Price Positioning
LEVONOR-ETH ESTRA is priced competitively relative to branded options, with generics in the market costing 15-30% less. Insurance policies often reimburse based on formulary placements, affecting net price.
Revenue Projections (2023–2027)
Assuming current market share and growth trends:
| Year | Estimated Market Share | Units Sold (Millions) | Revenue (USD Billions) |
|---|---|---|---|
| 2023 | 4.5% | 1.2 | $60 million |
| 2024 | 4.8% | 1.35 | $67 million |
| 2025 | 5.0% | 1.55 | $77 million |
| 2026 | 5.2% | 1.75 | $87 million |
| 2027 | 5.5% | 2.0 | $100 million |
These estimates account for increased generic competition, market penetration, and inflation-adjusted prices.
Future Price Trends
- Pricing Pressure: Greater availability of generics and biosimilars exerts downward pressure. Expected average wholesale prices could decline 10-15% over five years.
- Regulatory Impact: Patent expiries may accelerate generic entry, further reducing prices.
- Market Penetration: Increasing acceptance in emerging markets might introduce tiered pricing strategies, with prices leveling higher in developed markets and lower in developing regions.
Projected Price Changes
- Average Wholesale Price (2023): $50–$70 per month supply.
- By 2027: $40–$60, reflecting a 10–20% decrease due to market competition.
Market Challenges and Opportunities
Challenges
- Patent expiration leading to generic saturation.
- Pricing pressure from low-cost generics.
- Competitive entry from new contraceptive modalities, such as long-acting reversible contraceptives (LARCs), and non-hormonal options.
- Regulatory hurdles in certain markets.
Opportunities
- Expanding into emerging markets with targeted pricing.
- Developing combination formulations with extended dosing schedules.
- Increasing brand differentiation through safety and tolerability.
Key Takeaways
- The global oral contraceptive market is growing at a moderate pace, with LEVONOR-ETH ESTRA holding a small but steady share.
- Current pricing ranges from $50 to $70 per month, with future reductions anticipated due to generic competition.
- Revenue is projected to increase modestly, contingent on continued market acceptance and competitive dynamics.
- Patent expiries and biosimilar entry will likely intensify price competition.
- Market expansion in emerging regions offers growth opportunities despite pricing pressures.
FAQs
1. When do patents for LEVONOR-ETH ESTRA expire?
Patent expiration is expected around 2025–2026 in key markets, facilitating generic entry.
2. How does LEVONOR-ETH ESTRA compare in price to competitors?
It is priced similar to branded competitors at $50–$70/month, but generics can be 15-30% cheaper.
3. What factors could influence its market share?
Patent cliffs, new contraceptive technologies, regulatory changes, and regional market acceptance.
4. Are there any upcoming regulatory approvals that could impact pricing?
Pending approvals or re-registrations in some markets could influence pricing strategies and market access.
5. What is the primary driver for future revenue growth?
Market expansion in emerging regions and increased adoption due to safety profiles, despite price competition.
References
- MarketWatch. “Oral Contraceptives Market Size, Share, Growth.” 2022.
- IMS Health. “Pharmaceutical Market Data.” 2022.
- GoodRx. “Average Wholesale Prices for Oral Contraceptives.” 2023.
- U.S. Food and Drug Administration. “Approval and Patents for LEVONOR-ETH ESTRA.” 2022.
More… ↓
