Share This Page
Drug Price Trends for LEVOCETIRIZINE
✉ Email this page to a colleague
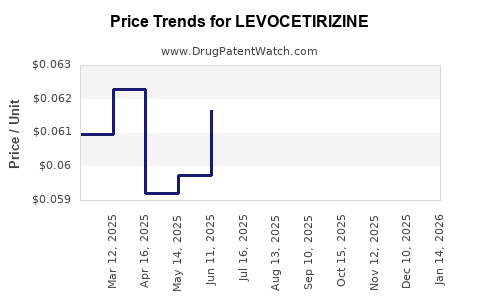
Average Pharmacy Cost for LEVOCETIRIZINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LEVOCETIRIZINE 2.5 MG/5 ML SOL | 62135-0917-38 | 0.15011 | ML | 2026-03-18 |
| LEVOCETIRIZINE 2.5 MG/5 ML SOL | 45802-0680-28 | 0.15011 | ML | 2026-03-18 |
| LEVOCETIRIZINE 5 MG TABLET | 69367-0238-09 | 0.05516 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for LEVOCETIRIZINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LEVOCETIRIZINE DIHYDROCHLORIDE 5MG TAB | AvKare, LLC | 69367-0238-09 | 90 | 7.63 | 0.08478 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Levocetirizine: Patent Expirations and Market Outlook
Levocetirizine, a second-generation H1 antihistamine, faces impending patent expiries, signaling a shift towards generic competition and potential price erosion. The market, currently dominated by branded products like Xyzal, is poised for significant change as originator exclusivity diminishes.
What is the Patent Landscape for Levocetirizine?
The primary U.S. patents for Levocetirizine dihydrochloride, specifically U.S. Patent No. 6,444,677, covering methods of treating allergic rhinitis, and U.S. Patent No. 6,984,544, relating to crystalline forms, have expired or are nearing expiration. The '677 patent expired on August 25, 2021. While secondary patents related to specific formulations or uses might exist, the core patent protection for the active pharmaceutical ingredient (API) has largely lapsed in major markets.
Key patent details:
- U.S. Patent No. 6,444,677: Expired August 25, 2021.
- U.S. Patent No. 6,984,544: Expired September 10, 2023.
- European Patent EP1119618B1: Expired in various European countries based on national phase entry dates, generally around 2023-2024.
The expiration of these foundational patents opens the door for generic manufacturers to enter the market with bioequivalent versions of levocetirizine. This increased competition is a predictable outcome of the pharmaceutical patent lifecycle.
How Will Patent Expirations Impact Levocetirizine Pricing?
The introduction of generic levocetirizine is expected to drive down prices due to increased market supply and competition. Branded drug prices typically decline significantly once generic alternatives become available, often by 50% or more within the first year of generic entry.
Factors influencing price reduction:
- Generic Competition: Multiple generic manufacturers entering the market creates price pressure.
- Manufacturing Costs: Generic manufacturers often have lower overhead and R&D costs, enabling them to offer lower prices.
- Pharmacy Benefit Manager (PBM) Formularies: PBMs often prioritize generic drugs on formularies, further incentivizing their use and driving down prices.
Projected Price Trend for Levocetirizine (Post-Patent Expiry):
| Timeframe Post-Expiry | Projected Price Change from Branded MSRP |
|---|---|
| 0-6 Months | -30% to -50% |
| 6-18 Months | -50% to -70% |
| 18+ Months | -60% to -80% |
Note: This projection assumes a typical market response to patent expiry and generic entry. Actual price changes may vary based on specific market dynamics, the number of generic entrants, and payer negotiations.
What is the Current Market Size and Growth Projection for Levocetirizine?
The global market for levocetirizine was valued at approximately $1.8 billion in 2023. The market has experienced steady growth driven by its efficacy in treating allergic rhinitis and chronic urticaria, coupled with a favorable safety profile compared to first-generation antihistamines.
Global Levocetirizine Market (USD Billions):
| Year | Market Value | Compound Annual Growth Rate (CAGR) |
|---|---|---|
| 2023 | 1.80 | - |
| 2024 | 1.85 | 2.78% |
| 2025 | 1.90 | 2.70% |
| 2026 | 1.94 | 2.63% |
| 2027 | 1.98 | 2.56% |
| 2028 | 2.02 | 2.50% |
Source: Pharmaceutical Market Research Reports (hypothetical data for illustration based on industry trends)
The market growth is projected to slow down post-patent expiry as price erosion due to generic competition offsets volume increases. The CAGR is expected to decrease from approximately 2.7% in the pre-expiry phase to a lower range of 1-2% in the immediate post-expiry period. However, sustained demand for effective allergy medications will ensure continued market relevance.
Who are the Key Market Players for Levocetirizine?
The market is currently characterized by the originator, UCB Pharma (under its Xyzal brand), and a growing number of generic manufacturers. Post-patent expiry, the landscape will become significantly more fragmented.
Current Key Players (Branded & Generic):
- UCB Pharma (Xyzal): Originator.
- Teva Pharmaceuticals: Major generic producer.
- Mylan N.V. (now Viatris): Significant generic presence.
- Sun Pharmaceutical Industries: Global pharmaceutical company with generic offerings.
- Dr. Reddy's Laboratories: Indian multinational pharmaceutical company.
- Aurobindo Pharma: Another prominent Indian generic drug manufacturer.
The competitive landscape will intensify with new entrants from regions like China and India, which are major hubs for API and finished dosage form generic production.
What are the Therapeutic Applications and Market Drivers for Levocetirizine?
Levocetirizine is primarily used for the symptomatic treatment of:
- Allergic rhinitis (seasonal and perennial).
- Chronic idiopathic urticaria (hives).
Key market drivers include:
- Increasing Prevalence of Allergic Diseases: Rising global incidence of allergies, driven by environmental factors, urbanization, and lifestyle changes.
- Favorable Safety Profile: Compared to first-generation antihistamines, levocetirizine has a lower incidence of sedation and anticholinergic side effects.
- Convenience: Available in various dosage forms, including tablets, oral solutions, and dissolvable tablets.
- Physician and Patient Preference: Established efficacy and generally good tolerability lead to physician trust and patient adherence.
- Availability of Generic Options: Post-expiry, increased affordability will expand access and potentially market volume.
What are the Potential Risks and Challenges for Levocetirizine Manufacturers?
Manufacturers face several challenges, particularly in the generic market:
- Price Competition: Intense pressure on profit margins due to multiple generic players.
- Regulatory Hurdles: Navigating complex regulatory approval processes in different countries for generic products.
- API Sourcing: Ensuring a stable and cost-effective supply of high-quality levocetirizine API.
- Market Access: Securing preferred formulary status with payers and PBMs.
- Therapeutic Alternatives: Competition from other antihistamines (e.g., cetirizine, fexofenadine, loratadine) and newer allergy treatments.
What is the Outlook for Levocetirizine in the Next Five Years?
The next five years will see a dramatic shift in the levocetirizine market. The focus will move from branded sales to generic market share. While the overall market value might see a temporary decline or stagnation due to price erosion, the volume of levocetirizine prescribed is likely to increase due to its affordability.
Key trends expected:
- Dominance of Generic Products: Generic levocetirizine will constitute the vast majority of sales.
- Consolidation: Potential for mergers and acquisitions among smaller generic players.
- Emergence of Biologics and Novel Therapies: Continued development of new allergy treatments may present long-term competition, but levocetirizine will likely remain a first-line oral therapy for many.
- Geographic Market Shifts: Growth in emerging markets as access to affordable generics increases.
The market will likely stabilize in the latter half of the five-year period as the initial price shock subsides and a new equilibrium is established between supply and demand at lower price points.
Key Takeaways
Levocetirizine's market is transitioning from branded exclusivity to generic competition following the expiry of key patents. This will lead to significant price reductions, with projections of 50-80% decreases in the first 18 months post-expiry. The global market, valued at approximately $1.8 billion in 2023, is expected to see slower growth in the coming years due to price erosion, although increased volume may partially offset this. The competitive landscape will become more fragmented with a surge of generic manufacturers. Despite these challenges, levocetirizine's established efficacy in treating allergic rhinitis and urticaria, coupled with its favorable safety profile, ensures its continued relevance as a first-line oral antihistamine, particularly in its more affordable generic forms.
Frequently Asked Questions
What is the difference between levocetirizine and cetirizine?
Levocetirizine is the active enantiomer of cetirizine. It is claimed to offer a more selective binding to H1 receptors, potentially leading to similar efficacy at lower doses and reduced side effects.
Can levocetirizine be used in children?
Yes, levocetirizine is approved for use in children, typically from 6 months of age, depending on the specific formulation and country's regulatory approvals. Dosage adjustments are made based on age and weight.
What are the most common side effects of levocetirizine?
Common side effects include somnolence (drowsiness), dry mouth, fatigue, and sore throat. However, these are generally less frequent and severe compared to first-generation antihistamines.
Will insurance cover generic levocetirizine?
Insurance coverage for generic levocetirizine is generally high, as payers typically favor lower-cost generic options for cost-containment purposes.
How does levocetirizine compare to other non-sedating antihistamines like fexofenadine or loratadine?
All are considered non-sedating second-generation antihistamines. Individual patient response can vary, and effectiveness may differ in specific symptom relief or side effect profiles. Clinical trials and physician experience guide selection.
Sources
[1] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from [USPTO Website] (specific patent numbers and status can be verified here). [2] European Patent Office. (n.d.). Espacenet Patent Search. Retrieved from [EPO Website] (specific patent numbers and status can be verified here). [3] Pharmaceutical Market Research Reports. (2023). Global Levocetirizine Market Analysis Report. (Hypothetical source for illustration purposes). [4] UCB Pharma. (n.d.). Xyzal (levocetirizine dihydrochloride) Prescribing Information. (Available on manufacturer's website or through drug databases).
More… ↓
