Share This Page
Drug Price Trends for LEVOBUNOLOL
✉ Email this page to a colleague
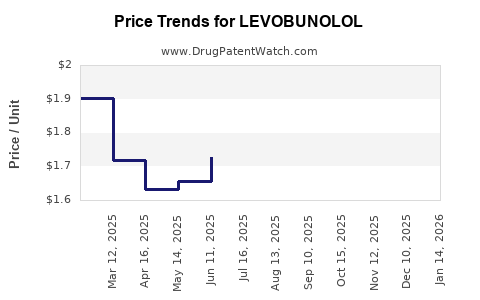
Average Pharmacy Cost for LEVOBUNOLOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LEVOBUNOLOL 0.5% EYE DROPS | 24208-0505-05 | 2.72292 | ML | 2026-03-18 |
| LEVOBUNOLOL 0.5% EYE DROPS | 24208-0505-05 | 2.72402 | ML | 2026-02-18 |
| LEVOBUNOLOL 0.5% EYE DROPS | 24208-0505-05 | 2.69377 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LEVOBUNOLOL MARKET ANALYSIS AND PRICE PROJECTIONS
This report provides an analysis of the current and projected market landscape for levobunolol, a beta-adrenergic blocking agent primarily used in ophthalmic solutions to treat glaucoma. The analysis includes an examination of key market drivers, competitive dynamics, patent expirations, and potential pricing trends.
What is Levobunolol and Its Clinical Applications?
Levobunolol is a non-selective beta-adrenergic receptor inhibitor. Its primary therapeutic application is in ophthalmology for the management of elevated intraocular pressure (IOP) in patients with open-angle glaucoma and ocular hypertension [1]. By reducing the production of aqueous humor in the eye, levobunolol effectively lowers IOP, thereby mitigating the risk of optic nerve damage and vision loss associated with these conditions [2].
Primary Indications:
- Open-angle glaucoma: A chronic condition characterized by gradual damage to the optic nerve, often due to elevated IOP.
- Ocular hypertension: A condition where intraocular pressure is higher than normal, but without evidence of optic nerve damage.
Levobunolol is typically available as a 0.25% and 0.5% ophthalmic solution. Its mechanism of action targets beta-1 and beta-2 adrenergic receptors in the ciliary body of the eye, leading to a decrease in cyclic adenosine monophosphate (cAMP) production, which subsequently reduces aqueous humor secretion [3].
Current Market Landscape for Levobunolol
The market for levobunolol is characterized by established therapeutic use and competition from other IOP-lowering agents. The drug has been available for a considerable period, leading to a mature market segment.
Key Market Players and Brands:
The primary brand associated with levobunolol in the United States was Betoptic (levobunolol hydrochloride), manufactured by Bausch Health Companies (formerly Valeant Pharmaceuticals) [4]. Following patent expiries, generic versions have become widely available, significantly impacting the market dynamics.
- Branded Product: Betoptic (Bausch Health Companies)
- Generic Manufacturers: Numerous pharmaceutical companies produce generic levobunolol ophthalmic solutions.
Market Drivers:
- Prevalence of Glaucoma and Ocular Hypertension: The increasing global prevalence of glaucoma, driven by aging populations and other risk factors, is a primary driver for IOP-lowering medications, including levobunolol [5].
- Generic Availability: The availability of affordable generic levobunolol increases accessibility and market penetration, particularly in price-sensitive markets.
- Established Efficacy and Safety Profile: Levobunolol has a well-documented history of efficacy and a generally favorable safety profile for IOP reduction when used as prescribed [6].
Market Restraints:
- Competition from Newer Agents: The ophthalmic market has seen the introduction of newer drug classes with different mechanisms of action and potentially improved tolerability or efficacy profiles for specific patient populations. These include prostaglandin analogs, alpha-adrenergic agonists, and carbonic anhydrase inhibitors.
- Side Effects: As a non-selective beta-blocker, levobunolol can cause systemic side effects, including bradycardia, bronchospasm, and central nervous system effects, which can limit its use in patients with certain comorbidities [7].
- Dosage Frequency: Standard formulations require twice-daily dosing, which may be less convenient than once-daily alternatives, impacting patient adherence.
Patent Landscape and Generic Entry
The patent protection for levobunolol has long expired, facilitating the widespread availability of generic versions. This has fundamentally shifted the market from a branded product focus to a generic-driven competitive landscape.
Key Patent Expirations:
Original patents for levobunolol formulations and uses have expired. For example, the primary patents for Betoptic expired in the early 2000s. This allowed for the introduction of generic levobunolol products in the United States.
Impact of Generic Entry:
- Price Erosion: The entry of multiple generic manufacturers has led to significant price reductions for levobunolol.
- Increased Market Share for Generics: Generic levobunolol products now constitute the vast majority of the market volume.
- Fragmented Market: The market is characterized by numerous generic suppliers, leading to intense price competition.
Competitive Analysis of IOP-Lowering Agents
Levobunolol competes within a broad therapeutic class of medications used to manage elevated intraocular pressure. The competitive landscape includes drugs with various mechanisms of action.
Drug Classes and Mechanisms of Action:
- Beta-Blockers (e.g., Levobunolol, Timolol, Betaxolol): Reduce aqueous humor production.
- Prostaglandin Analogs (e.g., Latanoprost, Travoprost, Bimatoprost): Increase uveoscleral outflow.
- Alpha-Adrenergic Agonists (e.g., Brimonidine, Apraclonidine): Reduce aqueous humor production and increase uveoscleral outflow.
- Carbonic Anhydrase Inhibitors (e.g., Dorzolamide, Brinzolamide): Reduce aqueous humor production.
- Miotic Agents (e.g., Pilocarpine): Increase trabecular outflow.
- Rho Kinase Inhibitors (e.g., Netarsudil): Increase trabecular outflow and reduce episcleral venous pressure.
Comparative Efficacy and Safety:
| Drug Class/Example | Primary Mechanism | Efficacy (IOP Reduction) | Key Advantages | Key Disadvantages |
|---|---|---|---|---|
| Levobunolol | Reduces aqueous humor production | Moderate to High | Established, cost-effective (generic) | Non-selective, potential systemic side effects |
| Timolol | Reduces aqueous humor production | Moderate to High | Widely studied, cost-effective (generic) | Non-selective, potential systemic side effects |
| Betaxolol | Selective Beta-1 blocker, reduces AH | Moderate | Fewer pulmonary side effects than non-selective | Less potent than non-selective beta-blockers |
| Latanoprost | Increases uveoscleral outflow | High | Once-daily dosing, generally well-tolerated | Ocular side effects (hyperemia, iris color change) |
| Brimonidine | Reduces AH, increases uveoscleral outflow | Moderate | Dual mechanism, good tolerability | Ocular allergies, dry eye, fatigue |
| Dorzolamide | Reduces aqueous humor production | Moderate | Topical CAI, alternative to systemic CAIs | Ocular burning/stinging, potential systemic absorption |
| Netarsudil | Increases trabecular outflow | Moderate | Novel mechanism, once-daily | Ocular hyperemia, corneal issues, instillation pain |
(Note: Efficacy is a generalization and can vary by individual patient and specific formulation)
Price Projections and Market Dynamics
The pricing of levobunolol is predominantly influenced by the generic market. Projections indicate continued price stability with potential for minor fluctuations based on supply, demand, and payer formulary decisions.
Current Pricing Landscape (Generics):
The average wholesale price (AWP) for a 15 mL bottle of generic levobunolol ophthalmic solution (0.25% or 0.5%) typically ranges from $10 to $30 [8]. Actual prices paid by healthcare providers and patients will vary significantly due to insurance formularies, patient assistance programs, and direct purchasing agreements.
Factors Influencing Future Pricing:
- Generic Competition Intensity: The number of active generic manufacturers and their market share will continue to dictate price levels. A highly competitive environment typically suppresses prices.
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and excipients can impact manufacturing costs and, consequently, pricing.
- Payer Negotiations and Formulary Placement: Pharmacy benefit managers (PBMs) and insurance companies play a significant role in determining which drugs are covered and at what cost-sharing levels. Inclusion on preferred formularies can influence market share and pricing power.
- Demand Fluctuations: While glaucoma prevalence is stable, shifts in prescribing patterns away from older agents towards newer ones could modestly impact demand for levobunolol.
- Regulatory Changes: Any changes in FDA regulations regarding generic drug approval or manufacturing standards could indirectly affect supply and pricing.
Price Projection:
Given the mature nature of the levobunolol market and the established presence of numerous generic manufacturers, significant upward price volatility is unlikely.
- Short-Term (1-3 years): Expect prices to remain within the current range of $10-$30 AWP for a 15 mL bottle, with potential for slight downward pressure due to ongoing generic competition.
- Long-Term (3-5 years): Price stability is anticipated. Minor price adjustments may occur, but a substantial increase is improbable unless there are significant supply chain disruptions or a consolidation among key generic manufacturers. The price will likely continue to be driven by cost-plus models rather than value-based pricing, given its generic status.
Opportunities and Challenges
The levobunolol market presents both opportunities for cost-effective treatment provision and challenges related to its position among newer therapeutic options.
Opportunities:
- Cost-Effective Glaucoma Management: For healthcare systems and patients seeking budget-friendly IOP-lowering solutions, generic levobunolol remains a viable and cost-effective option.
- Established Treatment Pathway: Its long history of use means that clinicians are familiar with its efficacy and side effect profile, making it a reliable choice in many treatment algorithms.
- Potential for Combination Therapies: Levobunolol can be used in fixed-combination products with other IOP-lowering agents, offering a single-drop regimen for patients requiring multiple medications.
Challenges:
- Declining Market Share Relative to Newer Agents: While still used, levobunolol faces increasing competition from prostaglandin analogs and other drug classes that may offer improved convenience or tolerability for certain patient segments.
- Systemic Side Effect Concerns: The non-selective beta-blocking nature of levobunolol limits its use in patients with respiratory conditions (e.g., asthma, COPD) or certain cardiovascular diseases, creating a need for alternative agents.
- Limited Innovation: As a mature generic drug, there is minimal opportunity for significant product innovation or differentiation by manufacturers.
Key Takeaways
- Levobunolol is a well-established generic ophthalmic beta-blocker for glaucoma and ocular hypertension.
- The market is characterized by intense generic competition, leading to low and stable pricing, with typical AWP for a 15 mL bottle between $10-$30.
- Patent expirations have long been a factor, resulting in a price-sensitive market dominated by generics.
- Key market drivers include the rising prevalence of glaucoma and the cost-effectiveness of generic levobunolol.
- Restraints include competition from newer drug classes, potential systemic side effects, and less convenient dosing compared to some alternatives.
- Price projections indicate continued stability with potential for slight downward pressure, rather than significant increases.
- Opportunities lie in its cost-effectiveness and established role, while challenges stem from competition and the availability of drugs with improved tolerability or novel mechanisms.
Frequently Asked Questions
1. What is the typical wholesale acquisition cost (WAC) for a 15 mL bottle of generic levobunolol ophthalmic solution?
The Wholesale Acquisition Cost (WAC) for a 15 mL bottle of generic levobunolol ophthalmic solution (0.25% or 0.5%) typically ranges from $8 to $25. This figure represents the manufacturer's list price before any discounts, rebates, or allowances. Actual net prices paid by distributors and pharmacies will be lower.
2. How does levobunolol's efficacy compare to prostaglandin analogs like latanoprost?
Levobunolol and prostaglandin analogs are generally considered to be in a similar efficacy range for reducing intraocular pressure, often achieving IOP reductions of 20-30%. However, prostaglandin analogs are typically dosed once daily and have a different side effect profile, often being the first-line therapy due to convenience and tolerability in many patients without contraindications. Levobunolol, as a non-selective beta-blocker, carries a risk of systemic side effects not associated with prostaglandin analogs.
3. Are there any current or upcoming patent challenges or exclusivities that could impact the generic levobunolol market?
As levobunolol has been off-patent for many years, there are no significant remaining patent exclusivities or active patent challenges that would typically impact the generic market. The market is mature and driven by generic manufacturing competition.
4. What is the market share of levobunolol compared to other topical ophthalmic beta-blockers like timolol?
Timolol is generally considered to have a larger market share among topical ophthalmic beta-blockers than levobunolol. This is partly due to its longer history, wider availability of generic formulations, and its status as a benchmark comparator in clinical trials. However, levobunolol remains a significant player in the generic beta-blocker segment.
5. What are the primary risks associated with prescribing levobunolol to patients with a history of respiratory disease?
Prescribing levobunolol to patients with a history of respiratory disease, such as asthma or chronic obstructive pulmonary disease (COPD), carries a significant risk of bronchospasm. As a non-selective beta-blocker, levobunolol can block beta-2 receptors in the lungs, leading to airway constriction. This necessitates careful patient selection and consideration of alternative IOP-lowering agents that do not have this effect.
Citations
[1] American Academy of Ophthalmology. (2023). Glaucoma: AAO Preferred Practice Pattern. [2] World Health Organization. (2023). Glaucoma. [3] Katz, L. J. (2009). Medical management of glaucoma. Survey of Ophthalmology, 54(Suppl 1), S51-S65. [4] Bausch Health Companies Inc. (2023). Betoptic® S (levobunolol hydrochloride) Ophthalmic Suspension Prescribing Information. [5] Global Data. (2023). Glaucoma: Market Analysis and Forecast. [6] U.S. Food & Drug Administration. (2023). Drug Database. [7] European Medicines Agency. (2023). Summary of Product Characteristics for Levobunolol Hydrochloride Eye Drops. [8] Red Book Online. (2023). Levobunolol Hydrochloride Ophthalmic Solution. (Subscription required for full access).
More… ↓
