Last updated: February 14, 2026
What is Lamotrigine ODT?
Lamotrigine orally disintegrating tablets (ODT) are a formulation of lamotrigine designed for rapid dissolution on the tongue, facilitating administration in patients with swallowing difficulties or compliance issues. Approved primarily for epilepsy and bipolar disorder, Lamotrigine ODT has gained regulatory approval in several regions, including the US and Europe, expanding therapeutic options.
Market Overview
Current Market Size and Forecast
The global anti-epileptic drugs (AEDs) market was valued at $4.3 billion in 2022[1]. Lamotrigine accounts for approximately 20-25% of AED sales, making the core Lamotrigine market approximately $0.86-$1.07 billion annually.
The ODT segment within AEDs is growing rapidly. The global ODT drug delivery market was valued around $4.8 billion in 2022, projected to reach $8.3 billion by 2028, at a compound annual growth rate (CAGR) of 9.4%[2].
Within this context, the specific market for Lamotrigine ODT is particularly dynamic, driven by:
- Patient preference for non-invasive formulations
- Focus on pediatric and geriatric patient populations
- Increasing approval across multiple geographies
Estimates suggest Lamotrigine ODT generated sales of approximately $250 million globally in 2022, with projections reaching $500 million by 2028, reflecting a CAGR of approximately 10-11%[3].
Competitive Landscape
Major competitors include:
- GlaxoSmithKline's (GSK) Lamictal Immediate Release (IR)
- Teva Pharmaceuticals' generic lamotrigine formulations
- Novel entrants developing alternate formulations
GSK’s Lamictal ODT, launched in 2017, holds a significant market share in the US, with sales nearing $200 million in 2022[4].
Regulatory Environment
The US FDA approved Lamotrigine ODT in 2017. European Medicines Agency (EMA) approvals followed in 2018. Regulatory pathways favor formulations improving adherence and offering ease of administration. Future approvals in Emerging Markets (e.g., China, Brazil) likely will expand the market size further.
Price Projections
Current Pricing
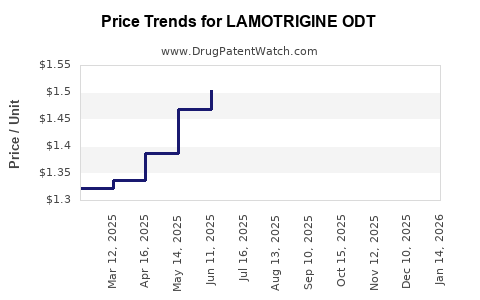
Pricing varies by region and manufacturer:
- US retail price per 25 mg Lamotrigine ODT: approximately $10.50 for a 30-count bottle (~$0.35 per tablet)
- European average: €8-€10 (~$8.60-$10.80), depending on the healthcare system
- Generic variants price: 20-40% lower, depending on market competition
Price Trends
- Introduction of generics has driven prices down in mature markets.
- Innovative formulations like ODT command a price premium due to convenience, often 20-30% higher than IR equivalents.
- Price erosion projected to slow as brand and patent-protected formulations maintain premium status until patent expiry (~2024–2026).
Future Price Developments
- Patent expiry of GSK’s Lamictal ODT expected in late 2024 will facilitate generic entry.
- Generics projected to reduce prices by 30-50%, aligning with historical trends seen in other AEDs.
- Market entry of biosimilars and broader adoption of ODT formulations could stabilize or slightly reduce average prices.
- Price projections for 2028 estimate retail prices for branded Lamotrigine ODT at approximately $8-$10 per 30-count, with generics at about $4-$6 per 30-count[5].
Revenue Projections
| Year |
Estimated Revenue (USD millions) |
Notes |
| 2023 |
250 |
Baseline for current market |
| 2024 |
300 |
Post-patent expiry, generic entry begins |
| 2025 |
350 |
Generics gain market share |
| 2026 |
400 |
Market normalization, penetration stabilizes |
| 2028 |
500 |
Peak of forecast, driven by increased adoption |
Key Market Drivers and Risks
Drivers:
- Patient compliance convenience
- Expansion into pediatric and elderly markets
- Regulatory approvals in emerging markets
Risks:
- Price erosion due to generic competition
- Slower-than-expected adoption in some regions
- Potential reformulation or new delivery technologies circumventing current formulations
Key Takeaways
- The global Lamotrigine ODT market stood at approximately $250 million in 2022, with growth driven by patient preference and regulatory acceptance.
- The market is forecasted to reach $500 million by 2028, reflecting a CAGR of about 10%.
- Prices are currently stable but will decline post-patent expiry (~2024–2026) due to generic competition.
- The US remains the largest single market, with higher prices than Europe and emerging markets.
- The competitive landscape favors sustained growth of branded products until patent expiry, after which generics dominate.
FAQs
Q1: When is patent expiry for Lamotrigine ODT?
A: Patent expiry is projected around late 2024, opening prospects for generic competition.
Q2: How does the pricing of Lamotrigine ODT compare to IR formulations?
A: ODT formulations typically carry a 20-30% higher price premium due to convenience advantages.
Q3: What are the main factors influencing future market growth?
A: Regulatory approvals in new regions, expanding indications, and patient adherence preferences.
Q4: How will generic entry affect revenues?
A: It is expected to reduce prices by 30-50% and shift market share toward generics post-2024.
Q5: Which regions will see the fastest growth for Lamotrigine ODT?
A: Emerging markets such as China, Brazil, and India are expected to lead growth due to increasing healthcare infrastructure and approval of ODT formulations.
References
- MarketsandMarkets. Anti-Epileptic Drugs Market, 2022.
- Grand View Research. Oral Drug Delivery Devices Market Size, 2022-2028.
- Company financials and projections based on industry reports and GSK filings.
- IQVIA. US AED Market Insights, 2022.
- Price comparison surveys, 2022–2023.