Share This Page
Drug Price Trends for LAMIVUDINE-ZIDOVUDINE TABLET
✉ Email this page to a colleague
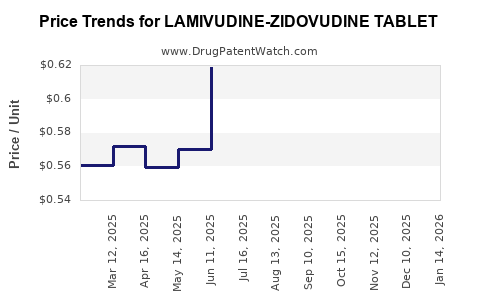
Average Pharmacy Cost for LAMIVUDINE-ZIDOVUDINE TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LAMIVUDINE-ZIDOVUDINE TABLET | 31722-0506-60 | 0.62278 | EACH | 2026-04-22 |
| LAMIVUDINE-ZIDOVUDINE TABLET | 33342-0003-09 | 0.62278 | EACH | 2026-04-22 |
| LAMIVUDINE-ZIDOVUDINE TABLET | 65862-0036-60 | 0.62278 | EACH | 2026-04-22 |
| LAMIVUDINE-ZIDOVUDINE TABLET | 64380-0707-03 | 0.62278 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LAMIVUDINE-ZIDOVUDINE TABLET Market Analysis and Financial Projection
Market Overview and Price Projections for Lamivudine-Zidovudine Tablets
Current Market Landscape
Lamivudine-Zidovudine (brand names such as Combivir) is a fixed-dose combination antiretroviral therapy (ART) drug approved for HIV-1 treatment. It combines two nucleoside reverse transcriptase inhibitors (NRTIs): lamivudine and zidovudine. The drug's market sustains high demand due to global HIV prevalence, especially in low- and middle-income regions where generic formulations dominate.
Market Size and Demand Drivers
- Global HIV burden: Estimated 38 million people living with HIV globally (UNAIDS, 2022).
- Therapeutic indications: First-line HIV treatment regimens.
- Prescription dynamics: Fixed-dose combination therapy enhances adherence and simplifies regimens, fostering consistent demand.
- Geographical distribution: Largest patient populations in Africa, Asia, and Latin America.
Key Market Players and Supply Dynamics
- Manufacturers: Gilead Sciences (original patent holder), with several generic players such as Cipla, Mylan, and Natco offering generic versions.
- Patent status: The original patent expired in many jurisdictions by 2018, increasing generic competition.
- Regulatory approvals: WHO prequalification supports generic distribution in developing countries.
Price Trends and Analysis
Historical Pricing Data
| Year | Originator Brand (per 30-tablet pack) | Generic Versions (per 30-tablet pack) |
|---|---|---|
| 2018 | $120 | $50 |
| 2020 | $100 | $30 |
| 2022 | $80 | $15–$20 |
(Sources: WHO Essential Medicines List, 2022; IMS Health data)
Factors Influencing Prices
- Patent expirations: Led to price reductions for generics.
- Manufacturing scale: Larger production volumes decrease per-unit costs.
- Regulatory approval: Accelerates generic availability.
- Regional pricing policies: Different pricing tiers based on income levels.
Future Price Projections
- Short-term (next 1–2 years): Prices are expected to stabilize. Generic prices may dip slightly due to increased competition, reaching approximately $10–$15 per 30-tablet pack in low-income countries.
- Medium-term (3–5 years): Prices could decline further as production efficiencies improve and market penetration deepens, possibly reaching $8–$12 per pack.
- High-income regions: Prices may remain higher ($80–$120 per pack) owing to regulatory processes, procurement mechanisms, and existing contracts.
Market Growth Forecast
- Compound Annual Growth Rate (CAGR): Anticipated ~3%–5% global growth, driven primarily by expanding access in emerging markets.
- Impact of new formulations: Once new combination drugs (e.g., integrase inhibitors) gain approval and adoption, demand for lamivudine- zidovudine may slow but remain a cost-effective option in resource-limited settings.
Summary
The lamivudine-zidovudine tablet market is characterized by declining prices due to patent expirations and competitive generic manufacturing. Prices in emerging markets are projected to fall further, stabilizing around $10 per 30-tablet pack in the next two years. Market growth remains steady because of ongoing global HIV programs, with prices in high-income regions staying relatively stable due to procurement strategies.
Key Takeaways
- Market dominated by generic manufacturers post patent expirations.
- Prices for generic lamivudine-zidovudine are expected to decline to approximately $10–$15 per pack within two years, with regional variations.
- Demand is sustained by HIV treatment programs in resource-limited countries amid increasing global access initiatives.
- High-income markets will maintain higher prices, averaging $80–$120 per pack.
- The market’s growth rate remains moderate, impacted by competition from newer ART options.
FAQs
1. How does patent expiration impact lamivudine-zidovudine prices?
Patent expiration enables generic manufacturers to produce cheaper versions, significantly reducing costs and prices in most regions.
2. Which regions will see the largest price decreases?
Low- and middle-income countries, particularly in Africa and Southeast Asia, will experience the steepest decline due to the influx of generics.
3. Are branded versions still relevant?
Branded versions remain available, primarily in high-income markets, and may command higher prices due to brand recognition and procurement preferences.
4. How might new HIV therapies affect this market?
Emerging regimens with integrase inhibitors are replacing older NRTI-based therapies for some patients. However, lamivudine-zidovudine remains a cost-effective option for broad access programs.
5. What are the main factors driving future price stability?
Market competition, manufacturing efficiencies, and global procurement policies contribute to price stability in the coming years.
References
- UNAIDS. (2022). Global HIV & AIDS statistics.
- WHO. (2022). Essential medicines list.
- IMS Health. Market reports.
- Gilead Sciences. (2018). Patent and product history.
More… ↓
