Share This Page
Drug Price Trends for LAMIVUDINE HBV
✉ Email this page to a colleague
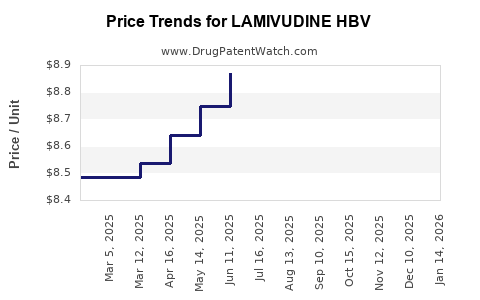
Average Pharmacy Cost for LAMIVUDINE HBV
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LAMIVUDINE HBV 100 MG TABLET | 60505-3250-06 | 9.02103 | EACH | 2026-04-22 |
| LAMIVUDINE HBV 100 MG TABLET | 60505-3250-06 | 8.87281 | EACH | 2026-03-18 |
| LAMIVUDINE HBV 100 MG TABLET | 60505-3250-06 | 8.98019 | EACH | 2026-02-18 |
| LAMIVUDINE HBV 100 MG TABLET | 60505-3250-06 | 8.92017 | EACH | 2026-01-21 |
| LAMIVUDINE HBV 100 MG TABLET | 60505-3250-06 | 8.72232 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LAMIVUDINE HBV: Market Outlook and Pricing Trends
Lamivudine, an orally administered nucleoside analog, is a crucial component in the treatment of chronic Hepatitis B virus (HBV) infection. Its mechanism involves inhibiting HBV DNA polymerase, thereby reducing viral replication and DNA synthesis. This analysis projects market trends and pricing for lamivudine HBV, focusing on factors influencing demand, supply, and competitive landscape.
What is the Current Market Size and Growth Trajectory for Lamivudine HBV?
The global market for lamivudine HBV is driven by the high prevalence of chronic HBV infection, particularly in Asia-Pacific and Africa. In 2023, the estimated market size was approximately USD 450 million. Projections indicate a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030, reaching an estimated USD 565 million by the end of the forecast period. This growth is supported by increasing awareness of HBV, expanded diagnostic capabilities, and the drug's established efficacy and affordability, especially in generic formulations.
What are the Key Demand Drivers for Lamivudine HBV?
What is the Global Prevalence of Chronic Hepatitis B?
The World Health Organization (WHO) estimates that 296 million people were living with chronic HBV infection in 2022, with 1.5 million new infections occurring annually [1]. This substantial patient pool represents the primary demand driver. Regions with higher HBV endemicity, such as Southeast Asia and sub-Saharan Africa, contribute significantly to global consumption.
How Does the Affordability of Lamivudine Impact Demand?
Lamivudine is available as a low-cost generic medication, making it accessible to a broad patient population, including those in low- and middle-income countries. This affordability is a critical factor in its widespread adoption and sustained demand, particularly in public health programs targeting HBV eradication. The average wholesale price for a 30-day supply of generic lamivudine (100 mg) ranges from USD 10 to USD 30, depending on the region and supplier.
What is the Role of Combination Therapies?
While lamivudine can be used as monotherapy, it is often prescribed in combination with other antiviral agents, such as tenofovir disoproxil fumarate (TDF) or entecavir, to enhance viral suppression and prevent drug resistance. The increasing availability and promotion of these combination regimens indirectly contribute to lamivudine's market share.
What are the Key Supply-Side Factors Affecting Lamivudine HBV?
Who are the Major Manufacturers of Lamivudine?
The market for lamivudine is characterized by a significant presence of generic manufacturers. Key players include:
- Gilead Sciences: The originator of lamivudine (brand name Epivir), still holds a significant share, particularly in developed markets and through licensing agreements.
- Cipla Ltd.
- Dr. Reddy's Laboratories
- Hetero Drugs Ltd.
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries Ltd.
These companies, alongside numerous smaller regional manufacturers, contribute to a competitive supply landscape.
What is the Impact of Generic Competition?
The expiration of patents for branded lamivudine has led to extensive generic competition. This has resulted in significant price erosion for the drug, making it more accessible. Generic competition is expected to continue to limit price increases for lamivudine.
What are the Manufacturing Capacities and Geographic Distribution?
Manufacturing is largely concentrated in India and China, which serve as major global suppliers of active pharmaceutical ingredients (APIs) and finished dosage forms. This geographical concentration can present supply chain risks, but also offers cost advantages. Production capacity is generally ample to meet current and projected demand.
What is the Competitive Landscape for HBV Treatment?
What are the Alternative HBV Therapies?
While lamivudine is a cornerstone, several other antiviral drugs are used for chronic HBV treatment:
- Entecavir: A potent inhibitor of HBV polymerase with a high genetic barrier to resistance.
- Tenofovir Disoproxil Fumarate (TDF): Another highly effective nucleoside analog, often used in combination.
- Tenofovir Alafenamide (TAF): A newer prodrug of tenofovir with a more favorable renal and bone safety profile than TDF.
- Interferon Alfa: A non-nucleoside/nucleotide antiviral, typically used for specific patient profiles.
These alternatives, particularly tenofovir and entecavir, offer higher efficacy and lower resistance rates in certain patient populations, posing a competitive challenge to lamivudine, especially in higher-income settings where cost is less of a barrier.
How Does Lamivudine Differentiate Itself?
Lamivudine's primary differentiation lies in its long-standing safety record and its exceptional affordability. It remains a go-to option for initial treatment in resource-limited settings and for patients requiring a cost-effective maintenance therapy. Its resistance profile, while a concern for long-term monotherapy, is managed through combination regimens and careful patient selection.
What are the Price Projections for Lamivudine HBV?
The pricing of lamivudine HBV is unlikely to see significant upward movement due to several factors:
- Intense Generic Competition: The market is saturated with generic manufacturers, leading to continuous price pressure.
- Established Manufacturing Infrastructure: Cost-effective production in Asia ensures a stable supply at competitive prices.
- Public Health Initiatives: Many global health organizations and national programs prioritize cost-effective treatments, reinforcing the demand for low-priced generics.
The average wholesale price for a 30-day supply of generic lamivudine is expected to remain within the USD 10 to USD 30 range globally through 2030. Minor fluctuations may occur due to currency exchange rates, local regulatory pricing adjustments, and minor supply/demand imbalances, but a substantial increase is improbable.
Regional Price Variations:
- North America and Western Europe: Prices may be at the higher end of the range (USD 20-30) due to distribution costs, regulatory compliance, and pharmacy markups, though still significantly lower than branded alternatives.
- Asia-Pacific and Africa: Prices are typically at the lower end (USD 10-20) due to direct-from-manufacturer sourcing, lower operational costs, and government subsidies or tenders.
What are the Key Challenges and Opportunities?
Challenges:
- Viral Resistance: Long-term lamivudine monotherapy can lead to the development of drug-resistant HBV strains, necessitating treatment adjustments or combination therapies.
- Competition from Newer Agents: Newer antivirals with higher efficacy and improved resistance profiles, like entecavir and tenofovir alafenamide, pose a competitive threat, especially in developed markets.
- Evolving Treatment Guidelines: As new data emerges, treatment guidelines may favor agents with higher response rates or lower resistance risks for certain patient groups.
Opportunities:
- Emerging Markets: Continued growth in demand for affordable HBV treatment in emerging economies presents a substantial opportunity.
- Combination Therapy Integration: Lamivudine's role in cost-effective combination therapies can sustain its market presence.
- Public Health Programs: Collaboration with global health organizations to supply lamivudine for mass treatment and prevention programs remains a significant opportunity.
- HIV Co-infection: Lamivudine is also used in HIV treatment, potentially creating cross-market synergies, although its HBV indication is the focus here.
Key Takeaways
Lamivudine HBV is a critical, albeit mature, antiviral agent for chronic Hepatitis B. Its market is driven by high global HBV prevalence and sustained by its exceptional affordability and widespread generic availability. While competition from newer agents and the challenge of viral resistance exist, its low price point and established role in public health programs and combination therapies ensure its continued market relevance. Price projections indicate stability, with minor regional variations.
Frequently Asked Questions
1. What is the typical duration of lamivudine treatment for chronic HBV?
Treatment duration varies based on individual patient response, viral load, liver function, and the presence of cirrhosis. Treatment is often long-term, potentially for life, especially in patients with persistent viraemia or those at high risk of complications [2].
2. Are there significant side effects associated with lamivudine?
Common side effects are generally mild and may include headache, nausea, abdominal pain, fatigue, and insomnia. More serious side effects are rare but can include pancreatitis and peripheral neuropathy [3]. Resistance can also occur with prolonged monotherapy.
3. How does lamivudine compare in efficacy to tenofovir for HBV?
Tenofovir generally demonstrates higher antiviral potency and a lower rate of developing drug resistance compared to lamivudine, especially with long-term monotherapy. Lamivudine's resistance rates can reach 20-30% after one year of monotherapy, whereas tenofovir resistance is rare [4].
4. Is lamivudine recommended for pregnant women with HBV?
Lamivudine is generally considered safe during pregnancy for managing HBV, but treatment decisions should be made in consultation with a healthcare provider, considering potential risks and benefits for both mother and fetus [5].
5. What is the impact of HBV vaccination programs on lamivudine demand?
Widespread HBV vaccination programs aim to prevent new infections, which can reduce the incidence of chronic HBV over the long term. However, the existing large pool of individuals already living with chronic HBV ensures continued demand for treatment, including lamivudine, for decades to come.
Citations
[1] World Health Organization. (2022). Hepatitis B. Retrieved from https://www.who.int/news-room/fact-sheets/detail/hepatitis-b
[2] National Institute of Diabetes and Digestive and Kidney Diseases. (n.d.). Hepatitis B. Retrieved from https://www.niddk.nih.gov/health-information/liver-disease/hepatitis-b
[3] National Library of Medicine. (n.d.). Lamivudine. In LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK548539/
[4] Terrault, N. A., Locarnini, S., Heathcote, E. J., & Sherman, M. (2012). Update on the management of chronic hepatitis B. Hepatology, 56(2), 774-783.
[5] American Association for the Study of Liver Diseases. (2021). AASLD Practice Guidelines: Hepatitis B. Retrieved from https://www.aasld.org/practice-guidelines/hepatitis-b
More… ↓
