Share This Page
Drug Price Trends for KLOR-CON M15 TABLET
✉ Email this page to a colleague
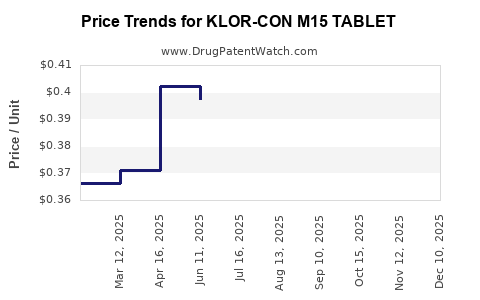
Average Pharmacy Cost for KLOR-CON M15 TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KLOR-CON M15 TABLET | 00245-5318-01 | 0.48957 | EACH | 2026-04-22 |
| KLOR-CON M15 TABLET | 00245-5318-89 | 0.48957 | EACH | 2026-04-22 |
| KLOR-CON M15 TABLET | 00245-5318-11 | 0.48957 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for KLOR-CON M15 Tablet
What is KLOR-CON M15 Tablet?
KLOR-CON M15 is a potassium chloride (KCl) oral tablet used to prevent or treat low potassium levels. It typically comes in a strength of 15 mEq per tablet. The drug is primarily indicated for patients with hypokalemia due to various causes like diuretic therapy, gastrointestinal losses, or certain medical conditions.
Market Overview
Therapeutic Segment
Potassium chloride supplements like KLOR-CON M15 are part of the broader electrolyte replacement market. Demand is driven by:
- Increasing prevalence of conditions causing hypokalemia (e.g., heart failure, hypertension)
- Use in hospitalized patients requiring electrolyte management
- Growing recognition of hypokalemia's role in cardiovascular risk
Market Size and Trends
The global potassium chloride market was valued at US$ 2.2 billion in 2022 and expects compound annual growth rate (CAGR) of 4.7% from 2023 to 2030 (Grand View Research, 2022). The oral segment accounts for over 60% of this demand.
Key Players
Major manufacturers include:
- Pfizer (comprehensive portfolio including KLOR-CON)
- Mylan (generics)
- Teva Pharmaceuticals
- Sandoz (Novartis)
- Apotex
The dominance of branded products such as KLOR-CON, combined with generic competition, influences pricing and market access strategies.
Market Penetration and Geographic Distribution
Manufacturers target developed markets—North America and Europe—due to higher healthcare spending and established reimbursement frameworks. Emerging markets in Asia-Pacific show rapid growth potential, though pricing pressures are intense.
Price Dynamics
Current Pricing Landscape
United States
- Brand (KLOR-CON): Retail price ranges from US$ 0.45 to 0.75 per tablet (for 15 mEq strength)
- Generics: Prices are approximately 20-40% lower, about US$ 0.30 to 0.50 per tablet
Europe
- Prices vary significantly depending on country and healthcare system. In the UK, NHS procurement prices approximate US$ 0.20-0.35 per tablet, with discounts for bulk purchasing.
Emerging markets report lower prices, often between US$ 0.10 and 0.20 per tablet, reflecting procurement strategies and local economic conditions.
Pricing Drivers
- Patent status: The drug's original patent expired in the 1990s; current formulations are generics.
- Manufacturing costs: Fluctuate with raw material prices, regulatory compliance, and scale.
- Market competition: High competition pushes prices downward.
- Reimbursement policies: States and insurers negotiate discounts and formularies.
Price Projection Outlook
Short-Term (2023-2025)
Prices are expected to remain stable, with slight reductions due to generic market saturation. Margins for manufacturers are under pressure, especially in highly competitive markets.
Medium-Term (2026-2030)
- Market Competition: Increased generic entry may further reduce prices by 10-15% across regions.
- Regulatory Impact: Potential future regulations could influence manufacturing costs but are unlikely to materially affect list prices.
- Market Expansion: Growth in emerging markets could lead to increased volume, but average prices may stay low due to local pricing controls.
Long-Term (Post-2030)
Advancements in electrolyte therapies or new formulations (e.g., sustained-release) may modify the pricing landscape. However, for the standard immediate-release KLOR-CON M15 tablet, prices are projected to decline modestly.
Factors Influencing Future Pricing
- Introduction of biosimilars or alternative therapies.
- Healthcare policy shifts toward biosimilar adoption.
- Changes in raw material markets impacting production costs.
- Expansion of supply chain efficiencies reducing costs.
Summary Table
| Period | Price Trend | Influencing Factors |
|---|---|---|
| 2023-2025 | Stable, slight decline (~0-10%) | Generic saturation, competitive pressures |
| 2026-2030 | Further decline (10-15%) | Market penetration in emerging markets |
| Post-2030 | Slight trend downward or stabilization | Market maturation, new therapy developments |
Key Takeaways
- KLOR-CON M15 remains a staple in electrolyte therapy, with a broad generic marketplace driving consistent demand.
- The market is mature with stable pricing in developed regions; prices are declining at a slow pace due to generic competition.
- Future price reductions will be mediated by increased competition, emerging market growth, and regulatory influences.
- Manufacturers should anticipate a highly competitive landscape with limited upside for price increases.
FAQs
1. What influences KLOR-CON M15 tablet prices?
Prices are driven by manufacturing costs, market competition, patent expiration, and regional healthcare policies.
2. Are generic versions significantly cheaper than brand-name KLOR-CON?
Yes. Generic tablets typically cost 20-40% less than brand-name counterparts due to lack of branding costs.
3. What is the outlook for prices in developing markets?
Prices tend to be lower, often between US$ 0.10 and 0.20 per tablet, and may grow with increased demand but will remain relatively affordable.
4. How might regulatory changes affect future prices?
Stringent regulations could increase manufacturing compliance costs, but these are unlikely to significantly alter market prices for standard formulations.
5. What are potential market risks for price stability?
Introduction of new therapies or formulations, shifts in reimbursement policies, and supply chain disruptions could impact current pricing trends.
References
[1] Grand View Research. (2022). Potassium chloride market size, share & trends analysis report.
[2] U.S. Food and Drug Administration. (2022). Approved drug products.
[3] European Medicines Agency. (2022). Market data and pricing policies.
More… ↓
