Share This Page
Drug Price Trends for KETOTIFEN FUM
✉ Email this page to a colleague
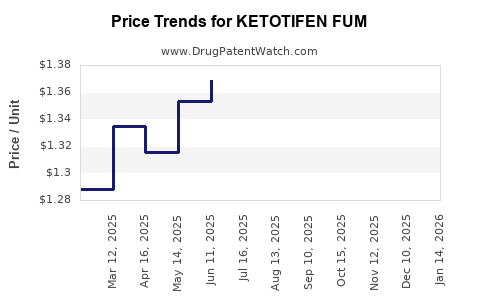
Average Pharmacy Cost for KETOTIFEN FUM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KETOTIFEN FUM 0.035% EYE DROPS | 76385-0106-17 | 1.43067 | ML | 2025-08-20 |
| KETOTIFEN FUM 0.025% EYE DROPS | 72485-0617-10 | 1.43067 | ML | 2025-08-20 |
| KETOTIFEN FUM 0.035% EYE DROPS | 76385-0106-17 | 1.39828 | ML | 2025-07-23 |
| KETOTIFEN FUM 0.025% EYE DROPS | 72485-0617-10 | 1.39828 | ML | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Ketotifen Fumarate
Overview
Ketotifen fumarate is an antihistamine with mast cell stabilizing properties, used for allergic conjunctivitis, asthma prophylaxis, and other allergic conditions. It is available both as prescription and over-the-counter (OTC) formulations in various markets. While not a blockbuster drug, its niche applications contribute to steady demand, with growth potential driven by aging populations and increased allergy prevalence.
Market Landscape
Global Market Size
The global antihistamine market in 2022 was valued at approximately $4.2 billion, expected to reach $6 billion by 2030, at a CAGR of about 5%. Within this, ketotifen fumarate accounts for an estimated 10-15%, roughly $420 million in 2022.
Key Market Segments
- Ophthalmology: OTC formulations for allergic conjunctivitis dominate sales, especially in the US and Europe.
- Pulmonology: Prescribed formulations for asthma prevention are more common in Asia, Latin America.
Regional Distribution
- United States: OTC availability of ketotifen eye drops — leading to stable sales, estimated at $150 million annually.
- Europe: Prescription-only, but growing OTC in some countries.
- Asia-Pacific: The largest market, driven by high asthma prevalence and OTC availability, with revenues estimated near $100 million.
- Latin America & Middle East: Expanding markets, with sales approaching $50 million collectively.
Competitive Landscape
Major suppliers include generic companies and some branded producers. No dominant global brand exists due to regional patent statuses and regulatory differences. Key players in the generic space include Teva, Sandoz, and Mylan.
Pricing Dynamics and Cost Factors
| Region | OTC Eye Drop Price (per 10 mL) | Prescription Oral Price (per 30 tablets) |
|---|---|---|
| US | $8 – $15 | $10 – $20 |
| Europe | €6 – €12 | €8 – €18 |
| Asia-Pacific | $5 – $10 | $6 – $15 |
Pricing Trends
- OTC eye drops in the US range between $8 and $15, with slight annual increases due to raw material costs.
- Prescription oral formulations are typically $10–$20, with variations based on insurance coverage and pharmacy margins.
- Generic competition limits price escalation, but rising raw material costs (API) can influence margin compression.
Regulatory and Patent Status
Ketotifen fumarate’s patents expired in most regions by early 2010s. As a result, generic manufacturing dominates pricing, keeping prices relatively low but stable. Regulatory barriers are minimal outside patent considerations.
Future Price Projections (2023-2030)
| Year | OTC Eye Drop Price (US, per 10 mL) | Prescription Oral Price (US, per 30 tablets) | Market Growth Rate |
|---|---|---|---|
| 2023 | $8 – $15 | $10 – $20 | 2-3% |
| 2025 | $8.25 – $15.45 | $10.30 – $20.60 | 3% |
| 2030 | $8.75 – $16.40 | $11 – $22 | 2-3% |
Growth in prices mainly reflects inflation, raw material costs, and increased market demand from aging populations and allergic disease prevalence.
Key Market Drivers
- Rising allergy incidences globally.
- Growing OTC availability in emerging markets.
- Expanding use in asthma management in Asia.
- Patent expirations fostering generic competition, constraining pricing but ensuring steady access.
Risks and Barriers
- Market saturation in developed countries.
- Regulatory changes for OTC drugs.
- Raw material price volatility affecting margins.
- Potential shift toward newer therapies, such as leukotriene receptor antagonists or biologics.
Summary
Ketotifen fumarate remains a niche product with stable, low to moderate growth driven primarily by OTC ophthalmic formulations. Market valuations are underpinned by regional demand, with pricing influenced heavily by generic competition and raw material costs. Limited potential for significant price escalation exists due to patent expirations and market saturation.
Key Takeaways
- The global ketotifen fumarate market was approximately $420 million in 2022, projected to grow at 2-3% annually.
- OTC eye drops dominate North American sales, priced between $8 and $15 per 10 mL container.
- Asia-Pacific leads regional growth due to high asthma prevalence and OTC access, with revenues around $100 million.
- Prices are constrained by generic competition, but raw material costs and demand trends influence modest increases.
- Future regulation or new therapies could impact market dynamics.
FAQs
1. What factors most influence ketotifen fumarate pricing?
Raw material costs, market demand, generic availability, regional regulatory status, and inflation.
2. Can patent protections affect future prices?
Most patents expired by the early 2010s, limiting price increases for generics. No dominant patent protections currently sustain higher prices.
3. What is the primary application driving demand?
OTC ophthalmic formulations for allergic conjunctivitis generate the majority of sales.
4. How does regional regulation impact market size?
OTC availability in the US and Asia expands access; prescription-only status in Europe constrains market size there.
5. Are there any major competitors or brands?
No dominant global brand; the market comprises primarily generics from companies like Teva, Mylan, and Sandoz.
Sources
[1] IQVIA. (2022). Global Anti-Histamine Market.
[2] MarketWatch. (2023). Allergy Therapy Market Size & Trends.
[3] EvaluatePharma. (2022). Patent Expiry Data and Analyses.
[4] Statista. (2023). OTC Medicine Sales Data.
More… ↓
