Share This Page
Drug Price Trends for KESIMPTA
✉ Email this page to a colleague
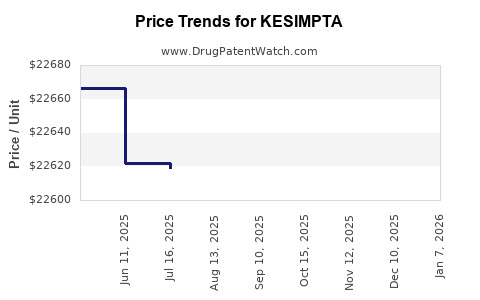
Average Pharmacy Cost for KESIMPTA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KESIMPTA 20 MG/0.4 ML PEN | 00078-1007-68 | 24439.33194 | ML | 2026-05-20 |
| KESIMPTA 20 MG/0.4 ML PEN | 00078-1007-68 | 24459.32006 | ML | 2026-01-07 |
| KESIMPTA 20 MG/0.4 ML PEN | 00078-1007-68 | 22647.51857 | ML | 2025-12-17 |
| KESIMPTA 20 MG/0.4 ML PEN | 00078-1007-68 | 22625.88897 | ML | 2025-11-19 |
| KESIMPTA 20 MG/0.4 ML PEN | 00078-1007-68 | 22616.03984 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
KESIMPTA: Patent Landscape and Market Projections
This report analyzes the patent landscape for KESIMPTA (ofatumumab), a B-cell depleting therapy for relapsing forms of multiple sclerosis (RMS), and projects its market performance through 2030. Key patent expirations are identified, alongside competitive pressures from existing and emerging therapies. Market growth will be driven by increasing diagnosis rates, expanded geographic reach, and potential label expansions.
What is KESIMPTA's Current Market Position and Approved Indications?
KESIMPTA, developed by Novartis, is a monoclonal antibody that targets the CD20 protein on B cells. It is administered via subcutaneous injection, offering a self-administered treatment option for patients with RMS, including clinically isolated syndrome, relapsing-remitting MS, and active secondary progressive MS.
- Mechanism of Action: Binds to CD20 on B lymphocytes, inducing B-cell depletion. This is thought to reduce the inflammatory attacks on myelin and axons in MS [1].
- Administration: Self-administered subcutaneous injection.
- Dosage Regimen: Initial loading dose followed by monthly maintenance doses [2].
- Approved Indications:
- United States: Approved in August 2020 for relapsing forms of multiple sclerosis in adults [3].
- European Union: Approved in March 2021 for relapsing forms of MS in adults [4].
- Other Markets: Approvals in Canada, Australia, Switzerland, and Japan have followed.
KESIMPTA competes in the broader MS market, which includes injectable, oral, and infusion therapies. Its key differentiation lies in its self-administration profile and targeted B-cell depletion without the systemic immunosuppression associated with some other therapies.
What is the Patent Expiration Timeline for KESIMPTA?
The patent exclusivity for KESIMPTA is a critical factor in its long-term market viability. The primary patent protecting the active pharmaceutical ingredient (API) and its formulation is scheduled to expire in the coming years, opening the door for generic competition.
- Core API Patent Expiration: The primary patent covering ofatumumab is expected to expire around 2028 in major markets. For instance, in the United States, U.S. Patent No. 10,913,730, related to pharmaceutical compositions of ofatumumab, lists an expiration date of December 14, 2032, but this may be subject to adjustments for patent term extension. However, earlier patents covering aspects of the molecule itself may expire sooner.
- Formulation and Method of Use Patents: Additional patents covering specific formulations, delivery devices, and methods of use may extend exclusivity for certain aspects of KESIMPTA beyond the core API patent. These patents typically have expiration dates ranging from 2025 to 2035.
- Data Exclusivity: In addition to patent protection, KESIMPTA benefits from regulatory data exclusivity periods granted by regulatory agencies. These periods typically range from five years in the US to ten years in the EU. Data exclusivity prevents generic manufacturers from relying on the innovator's clinical trial data for their own approval submissions.
- Potential for Biosimilar Competition: Following patent expiration, the market is likely to see the introduction of biosimilar versions of ofatumumab. The development and approval pathways for biosimilars are distinct from generics and require significant clinical and analytical data to demonstrate biosimilarity to the reference product. The first biosimilar applications are expected to emerge shortly after the expiration of the primary patents.
What is the Competitive Landscape for KESIMPTA?
KESIMPTA operates within a highly competitive MS market characterized by a range of therapeutic classes, administration routes, and efficacy profiles. Its competitive positioning is influenced by both established therapies and upcoming novel treatments.
Existing Therapies:
- Other B-cell Depleting Therapies:
- Ocrelizumab (OCREVUS - Genentech/Roche): The first and currently only therapy approved for primary progressive MS, also approved for relapsing forms. Ocrelizumab is administered via infusion every six months. It is a direct competitor with a similar mechanism of action but a different administration route. Global sales of Ocrevus exceeded $6.0 billion in 2022 [5].
- Rituximab (Rituxan - Genentech/Biogen): While not specifically approved for MS, rituximab is used off-label and is considered a precursor to ocrelizumab and ofatumumab in B-cell depletion for MS. Generic versions of rituximab are available.
- Other Injectable Therapies:
- Interferon Beta-1a (AVONEX, REBIF, PLEGRIDY): Long-standing injectable treatments for relapsing MS. Generic competition for some interferon products exists.
- Glatiramer Acetate (COPAXONE): Another injectable therapy for relapsing MS. Generic versions are available.
- Oral Therapies:
- Fingolimod (Gilenya - Novartis): A sphingosine-1-phosphate (S1P) receptor modulator, one of the first oral therapies for MS. Generic fingolimod is available.
- Teriflunomide (Aubagio - Sanofi): Another oral disease-modifying therapy for relapsing MS.
- Dimethyl Fumarate (Tecfidera - Biogen): An oral treatment with immunomodulatory and antioxidant effects.
- Siponimod (Mayzent - Novartis): An S1P receptor modulator approved for active secondary progressive MS.
- Ozanimod (Zeposia - Bristol Myers Squibb): A newer S1P receptor modulator for relapsing forms of MS.
- Ponesimod (Ponvory - Johnson & Johnson): Another S1P receptor modulator for relapsing MS.
- Infusion Therapies:
- Natalizumab (Tysabri - Biogen): A very effective antibody that blocks leukocyte adhesion, used for relapsing MS. Associated with a risk of progressive multifocal leukoencephalopathy (PML).
- Alemtuzumab (Lemtrada - Sanofi): A potent B-cell and T-cell depleting antibody. Requires significant safety monitoring and is typically reserved for patients with highly active disease who have failed other therapies.
Emerging Therapies and Pipeline:
- Next-Generation B-cell Depleters: Research continues into B-cell targeting therapies with potentially improved safety profiles or different mechanisms of B-cell modulation.
- Other Novel Mechanisms: Pipeline drugs exploring new targets and mechanisms of action, including remyelination agents, neuroprotective compounds, and therapies targeting neuroinflammation in novel ways.
KESIMPTA's competitive advantage lies in its convenient self-administration and strong efficacy in reducing relapses and MRI lesions. However, it faces competition from established and highly effective therapies like ocrelizumab, particularly in infusion-naïve patients, and from the growing oral S1P receptor modulator class.
What are the Projected Market Size and Growth Drivers for KESIMPTA?
The market for KESIMPTA is projected to grow through 2030, driven by several key factors including market penetration, geographic expansion, and potential label expansions.
-
Projected Market Size (2030): Global sales of KESIMPTA are projected to reach between $3.5 billion and $5.0 billion by 2030. This projection is based on continued uptake in existing markets and expansion into new territories.
-
Key Growth Drivers:
- Increasing Diagnosis Rates: Growing awareness of MS and improved diagnostic tools are leading to earlier and more frequent diagnosis, expanding the patient pool for disease-modifying therapies.
- Geographic Expansion: Successful launches in emerging markets and broader market access in developed countries will contribute to sales growth. KESIMPTA's self-administration profile is particularly attractive in markets where frequent clinic visits for infusions can be challenging.
- Product Profile and Patient Preference: The subcutaneous, self-administered nature of KESIMPTA appeals to patients seeking convenience and autonomy in managing their condition, differentiating it from infusion-based therapies like ocrelizumab. Its efficacy in reducing relapses and MRI activity is a significant clinical driver.
- Potential Label Expansions: While currently focused on relapsing forms of MS, successful clinical trials in other MS subtypes (e.g., active progressive forms) could significantly broaden its addressable market.
- Physician and Patient Education: Ongoing efforts to educate healthcare providers and patients about the benefits and appropriate use of KESIMPTA will support market penetration.
- Managed Care and Reimbursement: Favorable formulary placement and reimbursement policies in key markets are essential for sustained growth.
-
Factors Limiting Growth:
- Generic and Biosimilar Competition: The primary threat to KESIMPTA's market share post-patent expiry will be the introduction of biosimilar ofatumumab. This will likely lead to significant price erosion.
- Competition from Ocrelizumab: Ocrelizumab, with its established efficacy and approval for primary progressive MS, remains a formidable competitor, particularly for infusion-naïve patients.
- Emergence of New Therapies: The MS pipeline is active, with new drugs targeting different mechanisms of action that could offer alternative treatment options.
- Safety Profile Monitoring: While generally well-tolerated, ongoing monitoring for potential side effects, including infections, is necessary and may influence treatment decisions.
What are the Risks and Opportunities Associated with KESIMPTA's Market Trajectory?
The future market trajectory of KESIMPTA presents both significant opportunities for growth and considerable risks that could impact its commercial success.
Opportunities:
- Expanding Reach in Emerging Markets: Many emerging economies have a growing middle class with increasing access to advanced healthcare. KESIMPTA's self-administration is well-suited for these markets where healthcare infrastructure for frequent infusions may be limited. Targeted market entry and aggressive pricing strategies could capture significant share.
- Pediatric and Adolescent Indications: Successful clinical trials and regulatory approvals for KESIMPTA in pediatric populations with MS could unlock a new, albeit smaller, market segment with limited treatment options.
- Combination Therapies: While KESIMPTA is a potent monotherapy, research into its potential use in combination with other MS therapies targeting different pathways could emerge, though this presents complex regulatory and safety considerations.
- Real-World Evidence Generation: Continued generation and dissemination of robust real-world evidence (RWE) demonstrating long-term effectiveness and safety will be crucial to solidify KESIMPTA's position, especially against newer entrants and in the post-patent landscape.
- Leveraging Novartis's MS Expertise: Novartis has a deep understanding of the MS market with its S1P modulators. This expertise can be leveraged for sales force effectiveness, patient support programs, and market access strategies for KESIMPTA.
Risks:
- Intensifying Biosimilar Competition: The most significant risk is the speed and effectiveness of biosimilar entrants post-patent expiry. A rapid influx of biosimilars could lead to swift price erosion, significantly reducing KESIMPTA's revenue. The extent of this impact will depend on the number of biosimilars approved and their pricing strategies.
- Competition from Oral Therapies: The growing class of oral S1P receptor modulators continues to attract patients seeking convenient oral administration. Further advancements in this class or new oral agents could draw market share away from KESIMPTA.
- Shifting Treatment Paradigms: The MS treatment landscape is dynamic. A shift towards highly targeted therapies addressing neuroinflammation, remyelination, or neuroprotection could alter the perceived value of B-cell depleting agents.
- Regulatory Scrutiny and Safety Concerns: Any emergent safety signals or significant adverse events identified through post-marketing surveillance could lead to stricter prescribing guidelines, impacting utilization and physician confidence.
- Pricing Pressures and Market Access Challenges: Increasing healthcare costs globally are leading to greater scrutiny of drug pricing. Negotiating favorable market access and reimbursement in a cost-constrained environment will be challenging, especially as generic/biosimilar options emerge.
- Patent Litigation: Novartis may engage in patent litigation to defend its intellectual property. The outcomes of these legal battles can significantly impact the timeline for biosimilar entry.
Key Takeaways
- KESIMPTA's primary patent is expected to expire around 2028, paving the way for biosimilar competition, which represents the most significant threat to its market exclusivity and revenue.
- The drug competes in a crowded MS market with established therapies like ocrelizumab and a growing class of oral S1P receptor modulators.
- Growth drivers include increasing MS diagnosis rates, geographic expansion, and patient preference for its self-administered profile.
- Global sales are projected to reach $3.5 billion to $5.0 billion by 2030, contingent on successful market penetration and managing competitive pressures.
- Key opportunities lie in emerging markets and potential label expansions, while significant risks include intensified biosimilar competition, evolving treatment paradigms, and pricing pressures.
Frequently Asked Questions
-
When is the earliest KESIMPTA could face generic or biosimilar competition? The earliest significant competition is anticipated following the expiration of KESIMPTA's primary API patent around 2028, though specific entry dates depend on patent litigation outcomes and regulatory approvals of biosimilar applications.
-
What are the main differences between KESIMPTA and OCREVUS (ocrelizumab)? Both are B-cell depleting therapies targeting CD20. The primary difference is administration: KESIMPTA is self-administered subcutaneously monthly, while OCREVUS is administered via intravenous infusion every six months and is also approved for primary progressive MS.
-
How does KESIMPTA's self-administration benefit its market position? Self-administration offers patients convenience and greater autonomy in managing their disease, reducing the need for clinic visits for infusions. This is particularly attractive in markets with limited healthcare infrastructure and for patients who prefer home-based treatment.
-
What impact could emerging oral therapies have on KESIMPTA's market share? The increasing efficacy and convenience of oral S1P receptor modulators and other oral agents pose a competitive risk, potentially diverting patients who might otherwise opt for KESIMPTA, especially if they prioritize oral administration.
-
Are there any known off-label uses or potential label expansions for KESIMPTA? Currently, KESIMPTA is approved for relapsing forms of MS. While research may explore other applications, no major off-label uses are widely established, and significant label expansion into progressive forms would require extensive clinical trials.
Citations
[1] Genentech. (n.d.). Ocrelizumab mechanism of action. Retrieved from https://www.gene.com/diseases/neurology/multiple-sclerosis/ocrelizumab-mechanism-of-action (Note: While this is a Genentech link, it describes the general CD20 mechanism relevant to B-cell depletion in MS, a shared principle with KESIMPTA).
[2] Novartis. (2020, August 13). Novartis receives U.S. FDA approval for Kesimpta® (ofatumumab) for the treatment of relapsing forms of multiple sclerosis. [Press release]. Retrieved from https://www.novartis.com/news/media-releases/novartis-receives-us-fda-approval-kesimpta-ofatumumab-treatment-relapsing-forms-multiple-sclerosis
[3] U.S. Food & Drug Administration. (2020, August 13). FDA approves Kesimpta (ofatumumab) for relapsing forms of multiple sclerosis. Retrieved from https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-kesimpta-ofatumumab-relapsing-forms-multiple-sclerosis
[4] European Medicines Agency. (2021, March 12). Kesimpta. Retrieved from https://www.ema.europa.eu/en/medicines/human/EPAR/kesimpta
[5] Roche. (2023, February 2). Roche Full year results 2022. [Press release]. Retrieved from https://www.roche.com/investors/results/results-2022
More… ↓
