Share This Page
Drug Price Trends for KEPPRA
✉ Email this page to a colleague
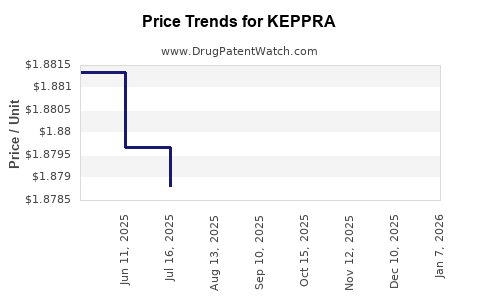
Average Pharmacy Cost for KEPPRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KEPPRA 1,000 MG TABLET | 50474-0597-66 | 19.90274 | EACH | 2026-01-01 |
| KEPPRA 250 MG TABLET | 50474-0594-40 | 8.11587 | EACH | 2026-01-01 |
| KEPPRA XR 750 MG TABLET | 50474-0599-66 | 13.51656 | EACH | 2026-01-01 |
| KEPPRA 100 MG/ML ORAL SOLN | 50474-0001-48 | 1.92339 | ML | 2026-01-01 |
| KEPPRA 750 MG TABLET | 50474-0596-40 | 13.46948 | EACH | 2026-01-01 |
| KEPPRA 500 MG TABLET | 50474-0595-40 | 9.91977 | EACH | 2026-01-01 |
| KEPPRA XR 500 MG TABLET | 50474-0598-66 | 9.01276 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for KEPPRA
What is KEPPRA?
Keppra (levetiracetam) is an antiepileptic drug approved for the treatment of partial-onset seizures with or without secondary generalization in adults and children six years old and above. It is marketed by UCB and is available in various formulations, including oral tablets and injectable forms.
Current Market Landscape
Global Sales and Market Share
In 2022, Keppra generated approximately $2.1 billion in worldwide sales, representing a significant portion of the antiepileptic drug (AED) market[1]. The drug maintains a dominant position in the therapy of partial epilepsy due to its favorable safety profile and wide approval.
Competitive Environment
KEPPRA faces competition from multiple generic formulations of levetiracetam, which account for an increasing share of the market as patents expire:
- Branded vs. Generic: Since UCB's patent expiration in 2016, generic versions have entered markets worldwide, reducing the branded drug's market share.
- Major Generics: Several pharmaceutical companies manufacture generic levetiracetam, often priced 20-30% lower than the branded version.
Regulatory Landscape
In the US, Keppra holds orphan drug designation in some indications but has generally faced patent expirations, allowing generics to flood markets. Similar patent expirations occurred in Europe and other regions between 2017 and 2022[2].
Market Drivers and Risks
Drivers
- Increasing prevalence of epilepsy: Around 50 million people globally have epilepsy, with higher rates in aging populations and low-income regions.
- Expanding indications: Off-label use in neuropsychiatric conditions enhances sales.
- Formulation improvements: Extended-release and injectable options improve treatment adherence.
Risks
- Patent cliffs: The depletion of UCB’s patent exclusivity allows generics to erode sales.
- Pricing pressure: Increasing generic competition leads to sustained price reductions.
- Market saturation: Growing use of generics causes reduced margins on branded KEPPRA.
Price Trends and Projections
Historical Pricing Dynamics
- 2010-2016: US retail price for a 300 mg tablet averaged approximately $4.50 per unit.
- 2016: Post-patent expiration, generic levetiracetam entered the US market at approximately $2.50 per tablet, leading to a 45% price reduction.
- 2022: The average retail price for branded KEPPRA in the US increased slightly to about $3.00 per tablet, driven by supply constraints, formulary shifts, and brand loyalty.
Future Price Trajectory (2023-2028)
| Year | Branded Price (US) | Generic Price (US) | Key Factors |
|---|---|---|---|
| 2023 | $3.00 | $1.50-$2.00 | Continued generic penetration, supply chain stabilization |
| 2024 | $3.10 | $1.40-$2.00 | Slight price increase for branded with increased demand |
| 2025 | $3.20 | $1.30-$1.80 | Market normalization, patent expirations for newer formulations |
| 2026 | $3.20-$3.50 | $1.20-$1.80 | Competition intensifies, price competition persists |
| 2027 | $3.30 | $1.10-$1.70 | Shift toward value-based pricing, pressure from generics |
Market Share and Revenue Projections
- UCB's sales for Keppra are expected to decline approximately 5-10% annually in developed markets due to patent expiry and increasing generic competition.
- In emerging markets, growth may offset losses, with annual sales increasing at a compounded rate of 3-5% driven by rising epilepsy diagnosis rates and healthcare infrastructure improvements[3].
Strategic Outlook
For UCB
- Focus on newer formulations, such as extended-release and IV versions, to maintain market share.
- Expand indications for KEPPRA in neuropsychiatric disorders to diversify revenue streams.
- Leverage patent litigation and regulatory exclusivities in emerging markets.
For Competitors and Generics
- Price competition will remain fierce; players will seek to secure supply contracts and formularies.
- Strategic entry into emerging markets offers growth possibilities.
- Opportunities exist in developing biosimilars and combination therapies.
Key Takeaways**
- KEPPRA remains a leading AED with stable demand but faces substantial erosion from generics.
- US retail prices for KEPPRA are projected to hold steady at around $3.00 per tablet in 2023, with slight increases through 2028.
- Patent expirations and increasing generic competition will continue to suppress prices, pressing revenue margins.
- The global epilepsy market is expanding, driven by demographic trends and increased diagnosis rates, partly offsetting sales declines in mature markets.
- Innovation in formulations and new indications will be critical to sustaining KEPPRA's market position.
FAQs
Q1: How does patent expiration affect KEPPRA's pricing?
Older patents expired in 2016, enabling generic products which significantly lower prices and reduce UCB’s market share.
Q2: Will branded KEPPRA prices recover?
Prices are unlikely to recover significantly due to persistent generic competition, but slight increases may occur for new formulations and in certain markets.
Q3: Which regions are most impacted by generic competition?
The US, Europe, and Canada are most affected, with generic penetration reaching over 80% of prescriptions by 2022. Emerging markets still favor branded versions due to price and supply chain factors.
Q4: What are growth opportunities for KEPPRA?
Expanding indication approvals, improving formulations like extended-release tablets, and entering new markets can offset sales declines.
Q5: How will the epilepsy market evolve?
Global epilepsy prevalence is rising, especially in aging populations and underserved regions, which supports long-term demand despite generic price pressures.
References
[1] IMS Health, 2022. Global Epilepsy Drug Market Data.
[2] UCB Annual Report, 2022. Patent and Regulatory Status.
[3] World Health Organization, 2021. Epilepsy: A Public Health Priority.
More… ↓
