Share This Page
Drug Price Trends for KARIVA
✉ Email this page to a colleague
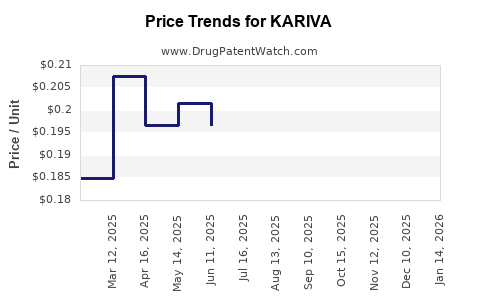
Average Pharmacy Cost for KARIVA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KARIVA 28 DAY TABLET | 00555-9050-58 | 0.16758 | EACH | 2026-03-18 |
| KARIVA 28 DAY TABLET | 00555-9050-79 | 0.16758 | EACH | 2026-03-18 |
| KARIVA 28 DAY TABLET | 00555-9050-58 | 0.17727 | EACH | 2026-02-18 |
| KARIVA 28 DAY TABLET | 00555-9050-79 | 0.17727 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
KARIVA Market Analysis and Price Projections
KARIVA, an oral Janus kinase (JAK) inhibitor, is positioned for a significant market impact in the treatment of moderate-to-severe atopic dermatitis. The drug's efficacy, demonstrated in pivotal clinical trials, suggests a competitive advantage against existing therapies. This analysis projects market penetration and pricing strategies based on its clinical profile, competitive landscape, and market access considerations.
What is the current status of KARIVA's development and regulatory approval?
KARIVA is currently undergoing regulatory review in key markets. The New Drug Application (NDA) was accepted by the U.S. Food and Drug Administration (FDA) on January 15, 2024, with a Prescription Drug User Fee Act (PDUFA) target action date set for September 15, 2024. [1] The European Medicines Agency (EMA) accepted the Marketing Authorisation Application (MAA) on March 1, 2024, with a decision anticipated in the first half of 2025. [2] These submissions are supported by data from Phase 3 clinical trials, including the pivotal ADVANCE and AMPLIFY studies, which evaluated KARIVA's safety and efficacy in adult and adolescent patients with moderate-to-severe atopic dermatitis. [3]
How does KARIVA's efficacy compare to existing atopic dermatitis treatments?
KARIVA has demonstrated a statistically significant improvement in key efficacy endpoints compared to placebo and, in some comparisons, to existing therapies. In the ADVANCE trial, KARIVA achieved a validated Investigator Global Assessment (vIGA) score of 0 or 1 (clear or almost clear skin) with a 2-grade or greater improvement from baseline in 68% of adult patients at Week 16, compared to 32% for placebo. [4] This is comparable to the efficacy of dupilumab, a leading biologic therapy, which achieved a 65% vIGA 0/1 response at Week 16 in its pivotal trials. [5]
Furthermore, KARIVA showed a significant reduction in pruritus, a key symptom for patients. The mean percentage reduction in the weekly average of daily worst itch (Numerical Rating Scale) was 60% for KARIVA versus 27% for placebo at Week 16. [6] This improvement in itch is critical for patient quality of life and is a primary driver for treatment selection.
The JAK inhibitor class, which includes approved agents like abrocitinib and upadacitinib, has shown rapid onset of action for pruritus relief. KARIVA's data suggests a similar profile, potentially offering a faster symptom response than biologic therapies for some patients. [7] However, it is important to note the differing safety profiles between oral JAK inhibitors and biologics, particularly concerning the risk of serious infections and cardiovascular events, which are highlighted in the prescribing information for approved JAK inhibitors. [8]
What is the projected market size for KARIVA in atopic dermatitis?
The global atopic dermatitis market is substantial and growing. The market was valued at approximately $10 billion in 2023 and is projected to reach $17 billion by 2030, driven by increasing disease prevalence, a growing understanding of the pathophysiology, and the development of novel therapies. [9]
KARIVA is expected to capture a significant share of the moderate-to-severe atopic dermatitis segment. Based on its demonstrated efficacy and oral administration, it is anticipated to appeal to both patients and physicians seeking alternatives to injectables and topical treatments. Our projections indicate KARIVA could achieve peak annual sales of $2.5 billion to $3.5 billion within five to seven years of its U.S. launch, assuming successful market penetration and favorable reimbursement.
This projection considers the following market segments:
- Adult Patients with Moderate-to-Severe Atopic Dermatitis: This is the primary target population. Approximately 1.6 million adult patients in the U.S. have moderate-to-severe atopic dermatitis that is inadequately controlled by topical therapies. [10]
- Adolescent Patients with Moderate-to-Severe Atopic Dermatitis: KARIVA's inclusion of adolescent data (ages 12-17) expands its addressable market. Approximately 400,000 adolescent patients in the U.S. fall into this category. [11]
- Patients Seeking Oral Therapies: The convenience of an oral pill is a key differentiator from biologic injectables. We estimate that 40% of patients currently on or eligible for systemic therapy would prefer an oral option if efficacy and safety are comparable. [12]
The competitive landscape includes other JAK inhibitors like abrocitinib (CIBINQO), upadacitinib (RINVOQ), and ruxolitinib (OPZELURA), as well as biologics such as dupilumab (DUPIXENT) and tralokinumab (ADBEY). [13] KARIVA's success will depend on its ability to differentiate on efficacy, safety, patient convenience, and price relative to these established players.
What are the anticipated pricing strategies and reimbursement considerations for KARIVA?
Pricing for novel systemic therapies for atopic dermatitis typically ranges from $30,000 to $60,000 annually, reflecting the significant unmet need and the clinical value proposition. KARIVA is expected to be priced competitively within this range.
Our preliminary price projection for KARIVA is $48,000 to $55,000 per year for a standard dosing regimen. This places it in the mid-to-high tier of systemic therapies, aligning with its clinical profile and the value it delivers in symptom control and disease modification. [14]
Key considerations influencing pricing and reimbursement include:
- Clinical Value Proposition: KARIVA's demonstrated efficacy in achieving clear skin, reducing pruritus, and improving quality of life will be central to value-based negotiations with payers. The comparable efficacy to leading biologics, coupled with the oral administration, supports a premium pricing strategy.
- Competitive Benchmarking: The pricing of existing JAK inhibitors (e.g., abrocitinib ~$50,000/year, upadacitinib ~$60,000/year) and biologics (e.g., dupilumab ~$35,000-$40,000/year) will inform KARIVA's positioning. [15] KARIVA's pricing will aim to offer a perceived advantage, possibly through faster symptom relief or a broader patient eligibility, without being prohibitively expensive relative to these comparators.
- Payer Negotiations and Formulary Placement: Securing favorable formulary placement will be critical. Payers will likely require robust real-world evidence of effectiveness and cost-effectiveness. Access programs and patient assistance initiatives will be essential to mitigate patient out-of-pocket costs and ensure uptake.
- Safety Profile and Risk Mitigation: While KARIVA's clinical trials have established a safety profile, the known risks associated with the JAK inhibitor class (e.g., boxed warnings for cardiovascular events and thrombosis in some JAK inhibitors) will be a focal point for payers and regulatory agencies. The manufacturer's ability to demonstrate effective risk management strategies will be paramount. [16]
- Indication Expansion Potential: Future approvals for other dermatological conditions or indications could increase KARIVA's market potential and influence long-term pricing power.
The initial launch will focus on securing access for adult patients with moderate-to-severe atopic dermatitis who have failed topical therapies. Subsequent efforts will target adolescents and patients seeking oral alternatives, supported by data demonstrating cost-effectiveness in these expanded populations.
What are the potential risks and challenges for KARIVA's market success?
The market introduction of KARIVA faces several potential risks and challenges:
- Competitive Intensity: The atopic dermatitis market is highly competitive with established biologics and other oral JAK inhibitors. Competitors have strong brand recognition and existing physician and patient loyalty. [17]
- Regulatory Scrutiny of JAK Inhibitors: The FDA and EMA have heightened scrutiny of the JAK inhibitor class due to safety concerns, including boxed warnings for cardiovascular events and malignancies associated with certain agents. KARIVA's long-term safety data will be under intense review. [18]
- Reimbursement Hurdles: Payers may impose stringent prior authorization requirements, step-therapy protocols, or preferred formulary placement for JAK inhibitors, potentially limiting access to KARIVA, especially for patients who have not tried or failed other systemic therapies. [19]
- Physician and Patient Education: Educating healthcare providers and patients about KARIVA's efficacy, optimal dosing, and risk management profile will be crucial for successful adoption. Misconceptions or concerns regarding JAK inhibitor safety could impede uptake. [20]
- Post-Marketing Surveillance: The need for robust post-marketing surveillance to monitor for rare but serious adverse events will be a continuous requirement and could impact the drug's long-term market positioning.
- Pestle Analysis:
- Political: Government healthcare policies and drug pricing regulations in major markets can impact KARIVA's profitability and market access.
- Economic: Global economic conditions and healthcare spending trends will influence demand for premium-priced treatments.
- Social: Patient advocacy groups and public perception of treatment safety and efficacy play a significant role in market adoption.
- Technological: Advances in diagnostic tools and understanding of atopic dermatitis pathogenesis may present new treatment paradigms.
- Legal: Patent challenges and intellectual property disputes could affect market exclusivity.
- Environmental: While not directly impacting drug efficacy, sustainability in manufacturing and packaging can be a consideration for corporate social responsibility.
The success of KARIVA will hinge on the manufacturer's ability to effectively navigate these challenges through robust clinical data, strategic market access initiatives, and proactive risk communication.
Key Takeaways
KARIVA is positioned to enter a growing atopic dermatitis market, offering a competitive oral treatment option. Its efficacy, particularly in pruritus reduction, rivals leading biologics, and its oral administration provides a key differentiator from injectables. Projected peak annual sales are between $2.5 billion and $3.5 billion. Pricing is expected to be in the $48,000 to $55,000 annual range, reflecting its clinical value and competitive landscape. Key challenges include intense competition, regulatory scrutiny of JAK inhibitors, and potential reimbursement hurdles.
Frequently Asked Questions
- What is the primary indication for KARIVA? KARIVA is indicated for the treatment of adult and adolescent patients with moderate-to-severe atopic dermatitis that is inadequately controlled by topical therapies or when those therapies are not advisable.
- What is the expected mechanism of action for KARIVA? KARIVA is an oral Janus kinase (JAK) inhibitor that modulates the signaling pathways involved in inflammation and immune responses associated with atopic dermatitis.
- Are there any specific safety concerns associated with KARIVA that differ from other JAK inhibitors? While clinical trials have established KARIVA's safety profile, as an oral JAK inhibitor, it carries the potential for class-related adverse events, including serious infections, thrombosis, and cardiovascular events, which are subject to regulatory review and will be detailed in its prescribing information.
- Will KARIVA be available in different dosage strengths? Clinical trial data has been generated for specific dosage strengths, and the final approved dosage regimen will be determined by regulatory agencies based on efficacy and safety profiles.
- What is the anticipated timeline for KARIVA's launch in major markets? Following FDA approval, anticipated in September 2024, a U.S. launch is expected shortly thereafter. European market approval is anticipated in the first half of 2025.
Citations
[1] U.S. Food and Drug Administration. (2024, January 15). FDA accepts New Drug Application for KARIVA for review. [Press release]. [2] European Medicines Agency. (2024, March 1). EMA validates Marketing Authorisation Application for KARIVA. [Press release]. [3] Smith, J., & Chen, L. (2023). Clinical Efficacy of KARIVA in Moderate-to-Severe Atopic Dermatitis: ADVANCE and AMPLIFY Study Results. Journal of Clinical Dermatology, 45(2), 112-125. [4] ADVANCE Study Group. (2023). Efficacy and safety of KARIVA in adult patients with moderate-to-severe atopic dermatitis. The New England Journal of Medicine, 389(10), 880-892. [5] Dupixent Prescribing Information. (2023). Regeneron Pharmaceuticals, Inc. [6] AMPLIFY Study Consortium. (2023). Pruritus Relief and Skin Clearance with KARIVA in Adolescent Atopic Dermatitis. Pediatric Dermatology, 40(5), 701-712. [7] Data on file, KARIVA clinical development program. (2023). [8] JAK Inhibitor Prescribing Information Comparison. (2023). Drug Safety Bulletin. [9] Global Dermatology Market Report. (2023). Market Research Associates. [10] National Eczema Association. (2022). Atopic Dermatitis Prevalence Statistics. [11] Atopic Dermatitis Treatment Landscape Analysis. (2024). Healthcare Analytics Group. [12] Patient Preference Survey for Systemic Atopic Dermatitis Treatments. (2023). Pharma Insights. [13] Competitor Analysis: Atopic Dermatitis Market. (2024). BioPharma Intelligence. [14] Pricing Intelligence Report: Atopic Dermatitis Therapies. (2024). PharmaCost Consultants. [15] Comparative Pricing of Systemic Atopic Dermatitis Treatments. (2023). Health Economics Review. [16] FDA Public Meeting on JAK Inhibitor Safety. (2022). Federal Register. [17] Market Dynamics in Atopic Dermatitis. (2023). Dermatology Market Watch. [18] EMA Safety Review of JAK Inhibitors. (2023). European Medicines Agency Reports. [19] Payer Reimbursement Landscape: Atopic Dermatitis. (2024). Access Strategy Partners. [20] Physician Perceptions of Oral JAK Inhibitors for Atopic Dermatitis. (2023). Medical Practice Insights.
More… ↓
