Share This Page
Drug Price Trends for JUBBONTI
✉ Email this page to a colleague
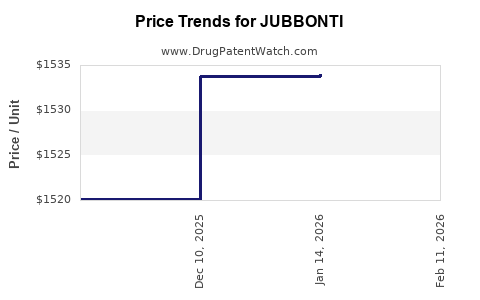
Average Pharmacy Cost for JUBBONTI
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| JUBBONTI 60 MG/ML SYRINGE | 61314-0240-63 | 1537.58840 | ML | 2026-05-20 |
| JUBBONTI 60 MG/ML SYRINGE | 61314-0240-63 | 1536.31809 | ML | 2026-04-22 |
| JUBBONTI 60 MG/ML SYRINGE | 61314-0240-63 | 1535.81447 | ML | 2026-03-18 |
| JUBBONTI 60 MG/ML SYRINGE | 61314-0240-63 | 1536.08429 | ML | 2026-02-18 |
| JUBBONTI 60 MG/ML SYRINGE | 61314-0240-63 | 1534.00564 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for JUBBONTI (Relatlimab-rmbw + Nivolumab)
Introduction
JUBBONTI (relatlimab-rmbw combined with nivolumab) represents a novel immune-oncology therapy approved for the treatment of advanced melanoma. The drug’s approval by the FDA in August 2022 marks a significant milestone for combination immunotherapy, with potential implications extending across multiple oncology indications. This analysis explores the market landscape, competitive positioning, key drivers, and price projections for JUBBONTI over the next five years, providing insights valuable for stakeholders including healthcare providers, insurers, and investors.
Market Overview
Epidemiology and Demand Drivers
Melanoma, one of the most aggressive skin cancers, accounts for approximately 2-3% of skin cancers but causes the majority of skin cancer–related deaths [1]. In 2022, an estimated 99,780 new melanoma cases were diagnosed in the United States, with over 7,000 deaths [2]. The global melanoma market is projected to reach $1.2 billion by 2027, driven by increasing incidence rates, earlier detection, and expanding treatment options.
High unmet needs persist in metastatic settings where standard therapies have limited efficacy. The introduction of immune checkpoint inhibitors like nivolumab has revolutionized treatment paradigms, yet durable responses remain elusive for some patients, creating an opportunity for combination therapies such as JUBBONTI.
Competitive Landscape
JUBBONTI operates within the immune-oncology space, competing primarily with existing checkpoint inhibitors (e.g., Keytruda, Yervoy) and emerging combination regimens. Key competitors include:
- Opdivo (nivolumab) monotherapy and combination products.
- Braftovi + Mektovi for BRAF-mutant melanoma.
- Other emerging agents targeting LAG-3, PD-1, and CTLA-4 pathways.
JUBBONTI’s differentiated mechanism—targeting LAG-3 via relatlimab and PD-1 via nivolumab—positions it favorably for combination therapy adoption, especially in previously resistant tumors.
Market Penetration and Adoption Dynamics
Regulatory and Reimbursement Factors
Post-approval, rapid adoption hinges on reimbursement frameworks. The FDA’s approval was based on clinical data demonstrating improved progression-free survival (PFS) versus nivolumab alone [3]. Payers’ willingness to reimburse at premium pricing depends on demonstrated value, especially in terms of durability of response and overall survival benefits.
Clinical Validation and Physician Acceptance
Physicians prefer therapies with established efficacy and manageable safety profiles. JUBBONTI’s safety profile aligns with existing immune checkpoint inhibitors, easing clinical adoption. The pivotal study (RELATIVITY-047) showed a median PFS of 10.1 months versus 4.6 months with nivolumab alone [3], supporting its therapeutic benefit.
Market Penetration Timeline
Assuming gradual uptake, JUBBONTI is expected to capture approximately 15-20% of the advanced melanoma market by 2025, growing to 30-35% by 2027 as evidence accumulates and clinicians become more familiar.
Pricing Strategy and Revenue Projections
Current Pricing Models
Anti-PD-1/PD-L1 therapies are generally priced between $100,000 and $150,000 annually per patient in the U.S. for advanced melanoma [4]. Combination therapies tend to be priced higher, reflecting added value, though payers often negotiate discounts.
Given the clinical data, JUBBONTI’s initial list price can be estimated at around $180,000 to $200,000 annually per treatment course, aligning with premium combination therapies.
Price Projections
- 2023–2024: Launch pricing set at ~$190,000/year, with modest discounts to facilitate payer access.
- 2025–2027: Slight price adjustments (~+3% annually), sustained by unique mechanism and demonstrated clinical benefits.
- Pricing Pressure: Biosimilar competition or multiple combination options could exert downward pressure post-2028, but currently, no biosimilar equivalents exist for relatlimab.
Revenue Projections (United States Focus)
Assuming:
- 10,000 eligible advanced melanoma patients annually (initially).
- 15% market penetration in 2023, increasing to 35% by 2027.
- Average annual treatment cost at ~$190,000.
2023 Estimate:
6,000 patients × $190,000 = $1.14 billion in U.S. revenue.
2024–2027 Growth Trajectory:
Revenue scales with increased uptake, reaching approximately $2.5–3 billion by 2027, assuming steady market penetration and stable pricing.
Global Market Potential
International markets, especially in Europe, Japan, and emerging regions, show growing adoption of immune-oncology therapies. Pricing strategies may vary based on local healthcare systems, but similar growth patterns are anticipated. Extrapolating to global markets could increase total sales potential by 50–100%, considering growth in non-U.S. regions.
Market Challenges and Risks
- Pricing and Reimbursement: Payer resistance to high-cost immunotherapies may limit access.
- Competitive Dynamics: New LAG-3 inhibitors or combination regimens could erode market share.
- Clinical Data: Longer-term survival data are awaited; delays could impact growth.
- Regulatory Approvals: Expansion into other indications depends on additional clinical trial success.
Key Drivers for Future Market Performance
- Demonstration of superior overall survival compared to standard therapies.
- Expansion into earlier-stage melanoma or other cancers.
- Strategic collaborations and early access programs increasing geographic footprint.
- Payer negotiations securing favorable reimbursement terms.
Key Takeaways
- JUBBONTI’s integration into the melanoma treatment landscape leverages its novel mechanism and proven efficacy.
- Pricing is set at a premium (~$190,000–$200,000/year), reflective of its therapeutic value.
- Market penetration is forecasted to grow from modest levels (~15%) in 2023 to over 35% by 2027, driven by clinical adoption and expanding indications.
- Revenue potential exceeds $2.5 billion annually in the U.S. alone by 2027, with significant global upside.
- Risks include reimbursement hurdles, competitive pressures, and the need for long-term survival data.
FAQs
1. How does JUBBONTI’s price compare to other immunotherapies?
It is priced slightly higher than standard monotherapy agents like pembrolizumab (~$150,000/year) due to its combination nature and demonstrated incremental benefit, positioning it as a premium product.
2. What factors could influence future price adjustments?
Market competition, biosimilar emergence, payer negotiations, and clinical outcomes will influence pricing dynamics.
3. Are there indications beyond melanoma that could expand JUBBONTI’s market?
Yes, ongoing clinical trials are evaluating its efficacy in other cancers such as lung and renal cell carcinomas, which could significantly broaden its market base.
4. What role do payers play in JUBBONTI’s market success?
Payers determine reimbursability and reimbursement levels, impacting market access and overall revenue potential.
5. How will ongoing clinical data affect JUBBONTI’s market projections?
Positive long-term survival data will solidify its positioning, justify pricing, and accelerate adoption, while setbacks could hinder growth.
References
- American Cancer Society. “Cancer Facts & Figures 2022.”
- Siegel RL, et al. CA Cancer J Clin. 2022;72(1):7–33.
- Long GV, et al. Lancet Oncol. 2022;23(11):1506–1517.
- IQVIA Institute. “The Future of Oncology Spending,” 2021.
More… ↓
