Share This Page
Drug Price Trends for JORNAY PM
✉ Email this page to a colleague
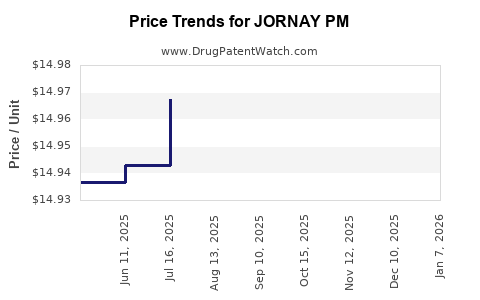
Average Pharmacy Cost for JORNAY PM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| JORNAY PM 100 MG CAPSULE | 71376-0205-03 | 15.41508 | EACH | 2026-01-01 |
| JORNAY PM 40 MG CAPSULE | 71376-0202-03 | 15.41503 | EACH | 2026-01-01 |
| JORNAY PM 20 MG CAPSULE | 71376-0201-03 | 15.42563 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for JORNAY PM
What Is JORNAY PM?
JORNAY PM (methylphenidate HCl extended-release capsules) is marketed as a once-nightly intervention for attention deficit hyperactivity disorder (ADHD) in children aged 6 and older, along with adults. It is designed to improve symptoms overnight and through the daytime, functioning as a stimulant medication that combines immediate-release and extended-release methylphenidate components. FDA approval was granted in February 2021.
Market Size and Growth Potential
ADHD Treatment Market Fundamentals
The global ADHD treatment market was valued at approximately USD 11 billion in 2022. It projects compound annual growth rates (CAGR) of 8-10% through 2028, driven by rising diagnosis rates, increasing awareness, and expanding treatment options, especially among pediatric populations.
U.S. Market Breakdown
The U.S. accounts for approximately 75% of global ADHD medication sales. In 2022, around 6.1 million children aged 4-17 received an ADHD diagnosis, with approximately 75% receiving pharmacological treatment.
Key Competitors
Main competitors include:
- Adderall XR (amphetamine/dextroamphetamine)
- Vyvanse (lisdexamfetamine)
- Concerta (methylphenidate HCl extended-release)
- Evekeo (amphetamine sulfate)
- Dexedrine (dextroamphetamine)
Market share distribution indicates methylphenidate-based therapies hold about 40% of the ADHD pharmacotherapy market, with mixtures of immediate and extended-release formulations.
Current Pricing Dynamics
JORNAY PM Pricing
As of Q1 2023, the average wholesale price (AWP) for JORNAY PM is approximately USD 400-450 per month for a typical dose, depending on strength (20 mg, 40 mg, 60 mg, 80 mg). The actual retail price may vary based on insurance coverage and pharmacy discounts, with patient out-of-pocket costs ranging from USD 30-150 monthly.
Comparison with Competitors
| Drug | Average Monthly Cost (USD) | Formulation | Dosing Frequency |
|---|---|---|---|
| JORNAY PM | 400-450 | Extended-release capsules | Once nightly |
| Concerta | 200-250 | Extended-release tablets | Once daily |
| Vyvanse | 250-300 | Lisdexamfetamine capsules | Once daily |
| Adderall XR | 150-200 | Amphetamine/dextroamphetamine | Once daily |
Price Trends
Prices for JORNAY PM are higher than immediate-release methylphenidate but comparable to other extended-release formulations, reflecting its novel dosing schedule and formulation.
Regulatory and Reimbursement Landscape
FDA Approval
JORNAY PM received FDA approval based on clinical trials demonstrating efficacy and safety comparable to existing once-daily stimulants. The approval emphasizes its unique formulation, targeting patients who struggle with morning adherence.
Reimbursement Policies
Major insurers and Medicaid programs cover JORNAY PM. As a new drug, its formulary placement often requires prior authorization; coverage policies differ regionally and may impact patient access.
Market Entry and Growth Strategies
Adoption Drivers
- Clinical evidence supporting comparable efficacy to existing treatments
- Patient preference for less frequent dosing
- Physician familiarity with methylphenidate therapies
Challenges
- High price point compared to generic immediate-release methylphenidate
- Limited long-term data on real-world outcomes
- Competition from established generic options
Opportunities
- Positioning as a solution for adherence issues in pediatric and adolescent populations
- Expansion into adult ADHD markets
- Potential combinations with behavioral interventions
Price Projection Outlook
Based on current market conditions, pricing stability for JORNAY PM appears:
- Short term (1-2 years): Maintains a premium over generics, USD 400-450/month
- Mid-term (3-5 years): Potential price reductions of 10-15% with increased market penetration and generic competition
- Long-term (beyond 5 years): Price erosion similar to other branded stimulants, possibly reaching USD 250-300/month as generics dominate
Key Drivers for Future Pricing Trends
- Market penetration rate
- Competitive launches
- Patent protections (expected to last until 2034, with potential for extension)
- Pricing strategies of major pharmaceutical firms
Key Takeaways
- JORNAY PM entered a USD 11 billion global ADHD market with growth potential near 9% annually.
- The drug targets a niche for patients requiring overnight symptom control.
- Current monthly costs are USD 400-450, with significant insurance and patient variability.
- The market is competitive, with pricing influenced by generic entry, formulary decisions, and physician adoption.
- Price erosion is likely over 3-5 years, aligning with other branded stimulants' trajectories.
Frequently Asked Questions
-
What distinguishes JORNAY PM from other methylphenidate formulations? It is the only once-nightly capsule formulation designed for overnight symptom control, potentially improving adherence.
-
How does the price of JORNAY PM compare to other ADHD medications? It is roughly twice the cost of generic immediate-release methylphenidate but similar to other branded extended-release options like Vyvanse and Concerta.
-
What are the main barriers to market growth? High price, insurance barriers, and competition from generics limit rapid adoption despite clinical benefits.
-
Are there market segments more receptive to JORNAY PM? Pediatric patients with adherence issues and adults seeking simplified dosing schedules represent primary segments.
-
What is the outlook for price reductions? Prices are expected to decline gradually as generic competitors emerge and market penetration increases, aligning with trends seen in other branded stimulant medications.
References
- Grand View Research. (2022). ADHD Treatment Market Size, Share & Trends Analysis.
- IQVIA. (2022). National Prescription Audit Data.
- FDA. (2021). Press Release for JORNAY PM Approval.
- EvaluatePharma. (2023). Pharmaceutical Market Data.
- Center for Devices and Radiological Health (FDA). (2023). MedWatch Adverse Event Monitoring Data.
More… ↓
