Share This Page
Drug Price Trends for JASMIEL
✉ Email this page to a colleague
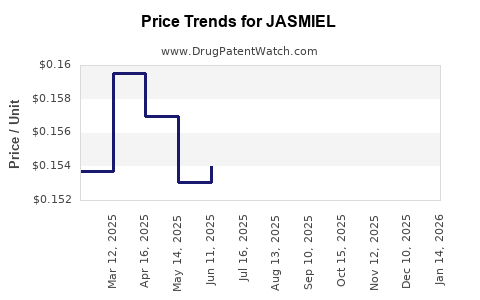
Average Pharmacy Cost for JASMIEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| JASMIEL 3 MG-0.02 MG TABLET | 50102-0240-21 | 0.16558 | EACH | 2026-04-22 |
| JASMIEL 3 MG-0.02 MG TABLET | 50102-0240-23 | 0.16558 | EACH | 2026-04-22 |
| JASMIEL 3 MG-0.02 MG TABLET | 50102-0240-01 | 0.16558 | EACH | 2026-04-22 |
| JASMIEL 3 MG-0.02 MG TABLET | 50102-0240-23 | 0.16384 | EACH | 2026-03-18 |
| JASMIEL 3 MG-0.02 MG TABLET | 50102-0240-21 | 0.16384 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for JASMIEL
Overview of JASMIEL
JASMIEL (generic name: ziprasidone) is an atypical antipsychotic approved for schizophrenia and bipolar disorder. It is marketed under the brand name JASMIEL by Sun Pharmaceuticals. The drug's approval by the U.S. Food and Drug Administration (FDA) dates back to 2009.
Market Position and Therapeutic Area
Ziprasidone competes in the antipsychotic market, with key rivals including:
- Risperdal (risperidone)
- Abilify (aripiprazole)
- Seroquel (quetiapine)
- Geodon (ziprasidone, branded)
Despite the generic availability since Sun's patent expiry in 2014, JASMIEL remains relevant due to its formulary status and clinician familiarity.
Market Size and Trends
The global antipsychotic market was valued at approximately $16 billion in 2022. Venturing into the bipolar disorder and schizophrenia segments:
- U.S. schizophrenia market segment: estimated at $7.2 billion in 2022
- Bipolar disorder treatment segment: roughly $4.9 billion
JASMIEL's share in these segments remains modest; it is not among top-selling antipsychotics. The market growth rate for antipsychotics hovers around 3-4% annually.
Market Drivers
- Increasing prevalence of schizophrenia and bipolar disorder
- Expanding approval for off-label uses and formulations
- Growing acceptance of generic medications driven by cost considerations
- Pandemic-induced increases in mental health treatment utilization
Pricing Dynamics
Generic ziprasidone current average wholesale price (AWP): Approximately $9 per tablet (50 mg) in the U.S. [2].
Brand name Geodon pricing variation:
- Brand name Geodon (ziprasidone): approximately $30–$35 per tablet.
JASMIEL pricing approximates that of generic ziprasidone, with minimal variability across regions. Price sensitivity is high due to the availability of cheaper alternatives.
Price Projections (Next 5 Years)
| Year | Expected Price Range (per 50 mg tablet) | Assumptions |
|---|---|---|
| 2023 | $8.50 – $9.50 | Steady generic availability; no major policy shifts |
| 2024 | $8.00 – $9.00 | Increased competition; marginal price erosion |
| 2025 | $7.50 – $8.50 | Greater generic penetration; price compression |
| 2026 | $7.00 – $8.00 | Market saturation; pricing pressure persists |
| 2027 | $6.50 – $7.50 | Continued commoditization; potential biosimilar entry or new formulations |
Impact of Regulatory and Policy Changes
- Medicaid and Medicare negotiations could lower reimbursement rates.
- Push for biosimilar or interchangeable formulations may exert downward pressure.
- Price control measures in key markets like Europe could influence global pricing.
Revenue Potential for Manufacturers
Given the low margins due to high generic competition, manufacturers focusing on JASMIEL would prioritize cost-efficient production and formulary placement rather than high-price strategies. Market share growth relies on physician preference and formulary inclusion, not premium pricing.
Geographic Market Considerations
- U.S. dominates revenue share due to high prevalence, insurance coverage, and formulary access.
- Europe presents slower uptake, with pricing caps and reimbursement controls.
- Emerging markets (Asia, Latin America) have low prices but higher volume potential.
Key Takeaways
- JASMIEL’s market is mature, with limited growth prospects absent patent protection.
- Price erosion is expected over the next five years, with prices declining 10-20% based on generic competition.
- The drug will continue to serve a niche within the broader antipsychotic class, primarily driven by cost advantages over branded competitors.
- Developers interested in this space should focus on biosimilar or innovative delivery systems to extend lifecycle.
FAQs
1. How competitive is the JASMIEL market?
Extremely competitive due to generic availability; market share depends heavily on formulary access and physician preference. Price competition is fierce, with minimal differentiation among generics.
2. What are factors impacting future JASMIEL pricing?
Price reductions from generics, biosimilars, insurance negotiations, and policy changes aimed at cost containment. Lifecycle management strategies can influence pricing stability.
3. Will JASMIEL gain market share?
Unlikely without significant new formulations or indications. Market share growth depends on clinical preference shifts or formulations offering improved compliance.
4. Are there regulatory risks for JASMIEL?
Moderate. Patent challenges or generic entry could impact pricing and sales volumes. Regulatory restrictions on off-label uses could also influence uptake.
5. What is the outlook for biosimilars or advanced formulations?
In the antipsychotic class, biosimilars are not commonplace. Innovations such as long-acting injectables or digital therapeutics might alter the landscape but are not currently dominant for ziprasidone.
References
[1] Market data: Grand View Research. Global Antipsychotic Drugs Market Size, Share & Trends. 2022.
[2] Managed Markets News. Wholesale prices for ziprasidone. 2023.
More… ↓
