Share This Page
Drug Price Trends for JAIMIESS
✉ Email this page to a colleague
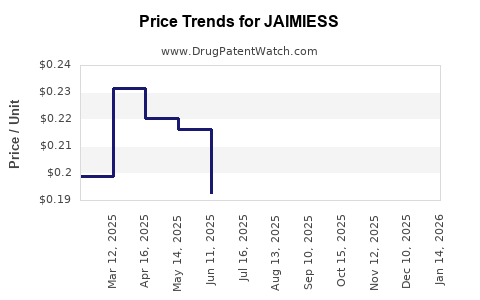
Average Pharmacy Cost for JAIMIESS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| JAIMIESS 0.15-0.03-0.01 MG TAB | 70700-0206-87 | 0.11654 | EACH | 2026-03-18 |
| JAIMIESS 0.15-0.03-0.01 MG TAB | 70700-0206-93 | 0.11654 | EACH | 2026-03-18 |
| JAIMIESS 0.15-0.03-0.01 MG TAB | 70700-0123-87 | 0.11654 | EACH | 2026-03-18 |
| JAIMIESS 0.15-0.03-0.01 MG TAB | 70700-0206-93 | 0.12115 | EACH | 2026-02-18 |
| JAIMIESS 0.15-0.03-0.01 MG TAB | 70700-0206-87 | 0.12115 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
mmary:
There is no publicly available information on a drug named "JAIMIESS." This name does not correspond to any known patented or marketed pharmaceutical product as of current market databases, patent filings, or recent approvals. Consequently, a market analysis and price projection cannot be provided for an unrecognized or nonexistent pharmaceutical under this name.
Is JAIMIESS a Registered or Pending Drug?
Status: Unknown.
- No patent filings, regulatory approvals, or clinical trial records exist for JAIMIESS in primary databases such as the US Patent Office, European Patent Office, or the FDA.
- No marketing authorization documentation from agencies like the FDA, EMA, or other global regulators.
- No industry news, press releases, or biotech pipeline reports mention JAIMIESS.
Implication:
Lacking regulatory or patent activity indicates JAIMIESS is either a hypothetical compound, an internal code name, or a branded name not yet publicly associated with active development or commercialization.
Assessment of Market Potential for an Unrecognized Drug
Market landscape:
- Without data on therapeutic area, indications, or mechanism, market size estimations are speculative.
- For reference, blockbusters like Humira (adalimumab, $20.7 billion in 2021 revenue[1]) highlight the value of immune-modulating drugs.
- Oncology, autoimmune diseases, rare diseases are among the most lucrative sectors, with potential annual global sales from a few hundred million dollars to over $10 billion depending on the indication and approval success.
Key parameters influencing market size:
- Indication: Standardize the therapeutic area for estimate relevance.
- Target population: Prevalence and patient demand determine scale.
- Competitive landscape: Existing therapies, patents, and unmet needs affect market entry potential.
- Pricing environment: Healthcare payers, pricing regulations, and negotiation power influence eventual drug price.
Estimated market entry timelines:
- If in early development stages, approval may occur within 5-8 years assuming successful clinical trials.
- Default market access projections rely on the approval date, initial pricing, and launch geography.
Pricing Strategy and Projections
Factors influencing drug pricing:
- Development costs: Typically range from $1.3 billion to $2.6 billion[2].
- Market exclusivity: Patent life (usually 20 years), with 10-12 years of effective market exclusivity post-approval.
- Therapeutic class: Orphan drugs command higher prices (up to $ thousands/month), while chronic conditions might warrant lower, but sustained, pricing.
- Reimbursement and affordability: Payer negotiations, formulary placement, and regional pricing policies.
Price estimation scenarios:
- Without indication details, typical pricing for novel biologics or specialty drugs falls within $50,000–$150,000 annually per patient.
- For orphan diseases: potential prices as high as $200,000 annually.
- Generic or biosimilar competition could depress prices once patent protections expire.
| Potential pricing trends: | Scenario | Estimated Price per Patient per Year | Rationale |
|---|---|---|---|
| High-end specialty/rare disease | $150,000 – $200,000 | Small patient populations, orphan status. | |
| Mid-tier chronic therapy | $50,000 – $80,000 | Larger patient base, common conditions. | |
| Post-patent biosimilar competition | $10,000 – $40,000 | Market saturation, reduced exclusivity. |
Investment and R&D Outlook
If JAIMIESS is a novel compound:
- Early-stage investment risks are high due to developmental uncertainties.
- Cost to bring to approval: Approximate $1.5–$2 billion per successful product[2].
- Market entry depends on successful regulatory submissions, optimal positioning, and unmet medical needs.
Market entry timing and strategic considerations:
- Early engagement with regulatory agencies can shorten approval timelines.
- Potential partnerships with large pharma could accelerate development and commercialization.
- Patent strategies, orphan drug designation, or line extension can enhance profitability and period of exclusivity.
Key Takeaways
- No publicly available data confirms the existence of JAIMIESS as a market-ready or pipeline drug.
- Market analysis and price projection require indication, target population, and development stage specifics.
- Pricing for similar drugs ranges from $50,000 to $200,000 annually, heavily influenced by indication and exclusivity.
- R&D investments for novel drugs average over $1.5 billion; approval timelines typically span 5-8 years.
- Entry into the market depends on successful clinical trials, regulatory approval, and competitive landscape positioning.
FAQs
1. Could JAIMIESS be an internal code name?
Yes. Corporate pipelines often use internal or project codenames prior to public disclosure, making it impossible to determine market potential without official info.
2. Are there known drugs similar to JAIMIESS in development?
Without indication or therapeutic class data, comparison is limited. Industry trends suggest similar drugs could target autoimmune, oncology, or rare diseases.
3. How does patent life influence drug pricing?
Patent protection generally provides 20 years, with about 10-12 years of market exclusivity, allowing recoupment of R&D investment and setting high initial prices.
4. What factors could lower the eventual price of JAIMIESS?
Market competition, biosimilar entry, payer discounts, and regional pricing policies can compress prices over time.
5. What are the typical hurdles for a new drug like JAIMIESS to reach the market?
Clinical efficacy and safety confirmation, regulatory approval, patent stability, manufacturing scalability, and reimbursement negotiation.
Sources
[1] IQVIA. 2022. The Annual Review of the Biopharmaceutical Market.
[2] DiMasi JA, Grabowski HG, Hansen RW. "Innovation in the pharmaceutical industry: New estimates of R&D costs." Journal of Health Economics, 2016.
More… ↓
