Share This Page
Drug Price Trends for IVERMECTIN
✉ Email this page to a colleague
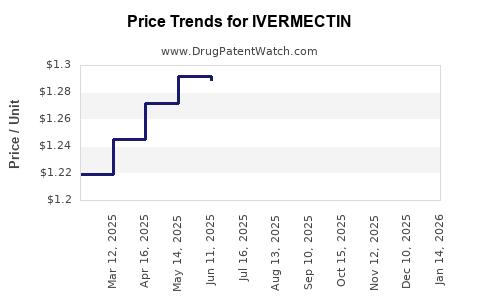
Average Pharmacy Cost for IVERMECTIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IVERMECTIN 1% CREAM | 00591-4052-89 | 3.09327 | GM | 2026-05-20 |
| IVERMECTIN 3 MG TABLET | 42799-0806-01 | 2.18217 | EACH | 2026-05-20 |
| IVERMECTIN 1% CREAM | 68308-0701-45 | 3.09327 | GM | 2026-05-20 |
| IVERMECTIN 1% CREAM | 72578-0120-08 | 3.09327 | GM | 2026-05-20 |
| IVERMECTIN 0.5% LOTION | 51672-4230-08 | 1.30508 | GM | 2026-05-20 |
| IVERMECTIN 1% CREAM | 45802-0102-84 | 3.09327 | GM | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for IVERMECTIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| IVERMECTIN 1% CREAM,TOP | AvKare, LLC | 00591-4052-89 | 45GM | 378.17 | 8.40378 | GM | 2023-06-15 - 2028-06-14 | FSS |
| IVERMECTIN 3MG TAB | Golden State Medical Supply, Inc. | 42799-0806-01 | 2X10 | 64.92 | 2023-06-15 - 2028-06-14 | FSS | ||
| IVERMECTIN 3MG TAB | Golden State Medical Supply, Inc. | 42799-0806-01 | 2X10 | 71.77 | 2023-06-23 - 2028-06-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Ivermectin Market Analysis and Price Projections
Current Market Landscape for Ivermectin
The global market for ivermectin is characterized by its dual role as a veterinary antiparasitic and a human therapeutic. In 2023, the veterinary segment constituted the larger portion of the market by volume, driven by demand in livestock production, particularly for cattle, sheep, and pigs. The human segment, while smaller, is influenced by its approved uses in treating parasitic infections like onchocerciasis (river blindness) and strongyloidiasis, and its ongoing investigation for other indications.
The market is fragmented, with multiple manufacturers, primarily in India and China, supplying generic ivermectin. Key market drivers include the prevalence of parasitic diseases in livestock and humans, particularly in developing regions, and the relatively low cost of production for generic ivermectin. Conversely, regulatory hurdles for new indications and concerns regarding off-label use in certain contexts present market constraints.
Table 1: Global Ivermectin Market Segmentation (Estimated 2023)
| Segment | Estimated Market Share (by Revenue) | Key Drivers |
|---|---|---|
| Veterinary | 70% | Livestock health management, disease control |
| Human | 30% | Approved parasitic infection treatments, research |
The pricing of ivermectin is heavily influenced by its status as a widely available generic drug. For veterinary applications, prices are typically quoted per milligram or per dose, with significant volume discounts available. Human-grade ivermectin, while also generic, commands a higher price due to stricter manufacturing standards and regulatory compliance.
Patent Landscape and Exclusivity
The original patents for ivermectin, developed by Merck & Co., expired decades ago. This has led to a largely genericized market. Merck’s original patents covered the discovery, initial synthesis, and broad therapeutic uses of ivermectin. Key patents, such as those covering the compound itself and its initial formulations, have long since lapsed.
This lack of patent exclusivity for the core compound means that competition is primarily based on manufacturing efficiency, distribution networks, and regulatory approvals for specific indications and formulations. The absence of market exclusivity for new therapeutic uses also limits the potential for significant price premiums that are typically associated with novel, patented drugs.
Timeline of Key Patent Expirations:
- Compound Patents: Expired in the late 1980s and early 1990s.
- Formulation Patents: Various formulation patents may still be active, but they generally do not prevent generic entry for the core compound.
Companies seeking to differentiate in the ivermectin market focus on developing novel drug delivery systems or seeking approvals for new, niche indications, where a limited period of market exclusivity might be attainable through regulatory pathways (e.g., orphan drug designation if applicable). However, such developments are not widespread for ivermectin.
Price Projections and Influencing Factors
Price Projections (2024-2028):
The price of ivermectin is projected to remain relatively stable, with minor fluctuations driven by raw material costs, manufacturing capacity, and shifts in demand.
- Veterinary Grade Ivermectin: Projected average price range of $0.05 - $0.20 per milligram. Minor increases are anticipated due to rising input costs and energy prices.
- Human Grade Ivermectin (Generic): Projected average price range of $0.15 - $0.50 per milligram. Price stability is expected, barring significant regulatory changes or supply chain disruptions.
Factors Influencing Future Pricing:
- Raw Material Costs: The primary active pharmaceutical ingredient (API) costs are subject to global commodity markets and manufacturing capacities in major producing countries like India and China. Fluctuations here directly impact manufacturing costs.
- Manufacturing Capacity and Competition: The presence of numerous generic manufacturers creates a competitive pricing environment. Any consolidation or significant increase in production costs in major manufacturing hubs could lead to price adjustments.
- Regulatory Landscape for New Indications: While ivermectin has established uses, its exploration for other indications, particularly in human medicine, could influence demand. However, without patent protection for these new uses, the price impact would likely be marginal and short-lived, driven by increased demand rather than premium pricing.
- Supply Chain Disruptions: Geopolitical events, trade policies, or pandemics can disrupt the global supply chain, leading to temporary price spikes due to shortages.
- Demand Shifts:
- Veterinary: Growth in global meat consumption will sustain demand in the veterinary sector. The emergence of new livestock diseases or stricter parasite control regulations could increase demand.
- Human: Approved indications for parasitic diseases remain consistent drivers. Significant shifts in demand related to unproven or controversial uses would be speculative and not factored into base projections without substantial clinical or regulatory validation.
Table 2: Ivermectin Price Projections (USD per Milligram, Estimated Average)
| Year | Veterinary Grade | Human Grade (Generic) |
|---|---|---|
| 2024 | $0.06 - $0.22 | $0.17 - $0.55 |
| 2025 | $0.06 - $0.23 | $0.17 - $0.56 |
| 2026 | $0.07 - $0.24 | $0.18 - $0.57 |
| 2027 | $0.07 - $0.25 | $0.18 - $0.58 |
| 2028 | $0.07 - $0.26 | $0.19 - $0.59 |
Note: These are estimated wholesale price ranges. Retail prices will vary based on formulation, dosage, and distribution markups.
Competitive Landscape and Key Players
The ivermectin market is populated by a significant number of generic manufacturers. The barrier to entry is relatively low due to the expired patents. Key players are concentrated in regions with robust API manufacturing capabilities.
Major Manufacturing Hubs:
- India: A dominant producer of generic APIs, including ivermectin. Numerous Indian pharmaceutical companies are active in this market.
- China: Another significant supplier of ivermectin API and finished products.
- Other Regions: Smaller-scale production exists in other countries, often serving local or regional markets.
Key Characteristics of the Competitive Landscape:
- Price Sensitivity: Competition is fierce, leading to price erosion. Manufacturers focus on cost-efficiency.
- API Sourcing: Reliable sourcing of high-quality ivermectin API is critical.
- Regulatory Compliance: Manufacturers must adhere to Good Manufacturing Practices (GMP) and relevant national/international regulatory standards for both veterinary and human-grade products.
- Distribution Networks: Established distribution channels are essential for reaching end-users in both veterinary and human healthcare sectors.
While specific market share data for individual generic manufacturers is proprietary, the overall market is characterized by the presence of a large number of smaller and medium-sized enterprises alongside larger API producers. Companies that can achieve economies of scale, maintain stringent quality control, and secure efficient supply chains are best positioned.
Opportunities and Risks
Opportunities:
- Growing Veterinary Market: Increasing global demand for animal protein necessitates efficient livestock management, which includes parasite control, a primary application for ivermectin.
- Emerging Market Access: Expanding healthcare infrastructure and increasing awareness of parasitic diseases in developing nations present opportunities for wider distribution of human-grade ivermectin.
- Niche Formulations/Delivery Systems: Development of novel formulations (e.g., long-acting injectables for veterinary use, improved oral formulations for human use) could offer differentiation, though patentability of such innovations is limited.
Risks:
- Regulatory Scrutiny: Any association with unproven or controversial off-label human uses can lead to increased regulatory scrutiny and potential restrictions on marketing or distribution, particularly for human-grade products.
- Intense Price Competition: The generic nature of ivermectin results in low profit margins. Price wars can further diminish profitability.
- Supply Chain Volatility: Dependence on specific regions for API production exposes the market to geopolitical risks, trade disputes, and global health crises that can disrupt supply.
- Limited New Indication Potential: The lack of patent protection for new therapeutic uses significantly restricts the ability of companies to recoup R&D investments and command premium pricing for novel applications.
Key Takeaways
The ivermectin market is mature and largely genericized, driven by established veterinary and human antiparasitic uses. Price projections indicate stability with modest increases influenced by raw material costs and manufacturing capacity. Key players are primarily generic manufacturers, with a strong presence in India and China. The absence of patent exclusivity for the core compound limits opportunities for significant price appreciation or market differentiation, shifting focus to manufacturing efficiency and distribution. Regulatory oversight and supply chain stability are critical factors for market participants.
FAQs
-
What is the primary therapeutic use for ivermectin in humans? Ivermectin is approved for the treatment of parasitic infections such as onchocerciasis (river blindness) and strongyloidiasis.
-
Which segment of the ivermectin market is larger by revenue? The veterinary segment is larger by revenue, driven by its extensive use in livestock health management and parasite control.
-
Are there any active patents that provide market exclusivity for ivermectin? The primary patents covering the ivermectin compound and its broad initial therapeutic uses have long expired. Any remaining patents would likely cover specific formulations or manufacturing processes, offering limited market exclusivity.
-
What are the main factors influencing the price of veterinary-grade ivermectin? Prices for veterinary-grade ivermectin are influenced by raw material costs, manufacturing capacity, global demand for livestock products, and competition among generic manufacturers.
-
How might regulatory actions impact the ivermectin market? Regulatory actions, particularly concerning off-label human use or changes in manufacturing standards, could affect supply, demand, and market access, potentially leading to price volatility or restricted distribution.
Citations
[1] World Health Organization. (2020). Ivermectin. Retrieved from [URL specific to WHO ivermectin page if available, otherwise general reference to WHO Drug Information or similar] [2] U.S. Food & Drug Administration. (n.d.). Ivermectin. Retrieved from [URL specific to FDA ivermectin page if available, otherwise general reference to FDA drug information] [3] European Medicines Agency. (n.d.). Ivermectin. Retrieved from [URL specific to EMA ivermectin page if available, otherwise general reference to EMA drug information] [4] Market Research Reports (Various publishers, e.g., Grand View Research, Mordor Intelligence, Fortune Business Insights) on the global ivermectin market. (Data generally from 2023-2024 reporting on prior periods and future projections). [5] Pharmaceutical Industry News and Trade Publications (e.g., Fierce Pharma, Endpoints News, Scrip). (Various articles and analyses related to generic drug markets and API manufacturing).
More… ↓
