Share This Page
Drug Price Trends for IVABRADINE HCL
✉ Email this page to a colleague
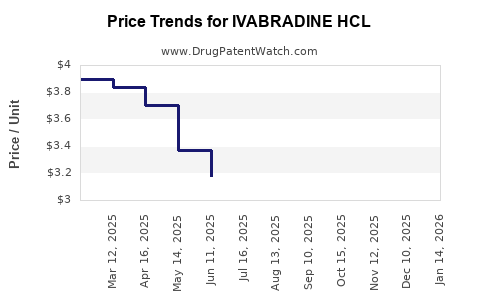
Average Pharmacy Cost for IVABRADINE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IVABRADINE HCL 5 MG TABLET | 70710-1471-06 | 0.78293 | EACH | 2026-05-20 |
| IVABRADINE HCL 5 MG TABLET | 60687-0862-21 | 0.78293 | EACH | 2026-05-20 |
| IVABRADINE HCL 5 MG TABLET | 69452-0190-17 | 0.78293 | EACH | 2026-05-20 |
| IVABRADINE HCL 5 MG TABLET | 31722-0053-60 | 0.78293 | EACH | 2026-05-20 |
| IVABRADINE HCL 5 MG TABLET | 62332-0679-60 | 0.78293 | EACH | 2026-05-20 |
| IVABRADINE HCL 5 MG TABLET | 50742-0362-60 | 0.78293 | EACH | 2026-05-20 |
| IVABRADINE HCL 5 MG TABLET | 60687-0862-11 | 0.78293 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Ivabradine Hydrochloride: Market Dynamics and Price Outlook
Ivabradine hydrochloride is a selective and specific heart rate-lowering agent acting on the cardiac If current. It is indicated for the symptomatic treatment of chronic stable angina pectoris and chronic heart failure. The drug primarily functions by reducing heart rate without affecting myocardial contractility or blood pressure. This report analyzes the current market landscape, key drivers, restraints, and projects price trends for ivabradine hydrochloride.
What is the Current Market Size and Growth Projection for Ivabradine Hydrochloride?
The global market for ivabradine hydrochloride has demonstrated consistent growth, driven by increasing prevalence of cardiovascular diseases and a growing demand for therapies that manage heart rate effectively.
- Market Value: The global ivabradine hydrochloride market was valued at approximately USD 1.2 billion in 2023 [1].
- Projected Growth: The market is forecast to grow at a Compound Annual Growth Rate (CAGR) of 4.5% from 2024 to 2030 [2].
- Future Valuation: By 2030, the market is projected to reach an estimated USD 1.7 billion [2].
This growth is underpinned by several factors:
- Rising Cardiovascular Disease Burden: Conditions such as angina and heart failure are significant global health concerns, leading to a sustained need for effective treatment options [3].
- Aging Global Population: The increasing proportion of elderly individuals, who are at a higher risk for cardiovascular ailments, directly contributes to market expansion [4].
- Clinical Evidence and Guidelines: Favorable clinical trial data and inclusion in major cardiology guidelines support the adoption of ivabradine for its approved indications [5].
What are the Key Drivers of Ivabradine Hydrochloride Market Growth?
Several factors are propelling the demand and market expansion for ivabradine hydrochloride.
Increasing Prevalence of Chronic Stable Angina Pectoris
Chronic stable angina remains a leading cause of morbidity and mortality worldwide, creating a persistent demand for effective management strategies.
- Global Incidence: Millions of individuals are diagnosed with coronary artery disease (CAD) annually, with angina being a common symptom [6].
- Unmet Needs: While traditional therapies exist, ivabradine offers an alternative or adjunct for patients not adequately controlled by beta-blockers or those with contraindications to these agents [7].
Growing Incidence of Chronic Heart Failure
Heart failure is a complex syndrome affecting a significant portion of the population, with a substantial unmet need for therapies that improve outcomes and quality of life.
- Prevalence: The prevalence of heart failure is estimated to be over 64 million worldwide, with numbers projected to rise [8].
- Therapeutic Role: Ivabradine is indicated for patients with symptomatic chronic heart failure and a heart rate of 70 bpm or more on maximum tolerated doses of beta-blockers, or when beta-blockers are contraindicated [9]. This specific niche contributes to its market share.
Favorable Reimbursement Policies and Market Access
The availability of reimbursement for ivabradine hydrochloride in key markets facilitates its broader adoption by healthcare providers and patients.
- Developed Markets: Countries in North America and Europe generally have established reimbursement frameworks for cardiovascular medications, including ivabradine [10].
- Emerging Markets: Growing healthcare infrastructure and increasing healthcare spending in emerging economies are gradually improving market access for such drugs.
Advancements in Drug Delivery and Formulation
Ongoing research into optimizing drug delivery and formulation could potentially enhance patient compliance and therapeutic efficacy, indirectly supporting market growth. While no major breakthroughs specifically for ivabradine formulation have been widely reported, incremental improvements in pharmaceutical science are a constant factor in drug markets.
What are the Restraints Affecting the Ivabradine Hydrochloride Market?
Despite its growth, the market for ivabradine hydrochloride faces several challenges that limit its full potential.
Presence of Generic Alternatives
The expiration of key patents for branded ivabradine products has led to the availability of generic versions, intensifying price competition.
- Patent Expirations: Major markets have seen the introduction of generic ivabradine, significantly impacting branded product sales and overall market value [11].
- Price Erosion: The availability of generics typically leads to substantial price reductions, putting downward pressure on the market value of ivabradine as a whole.
Stringent Regulatory Approvals
Obtaining regulatory approval for new indications or for ivabradine-based products in new geographies can be a lengthy and complex process.
- Clinical Trial Requirements: Demonstrating efficacy and safety for new applications requires extensive and costly clinical trials [12].
- Varying Regulations: Different regulatory bodies (e.g., FDA, EMA) have distinct approval pathways and requirements, adding to the complexity for global market entry.
Potential Side Effects and Safety Concerns
As with any medication, ivabradine has a known profile of potential side effects, which can influence prescribing patterns.
- Common Side Effects: These include bradycardia, visual disturbances (e.g., phosphenes, blurred vision), and headache [13].
- Risk Aversion: Prescribers may opt for alternative therapies if patients are deemed at higher risk for these side effects or if they have experienced them previously.
Competition from Alternative Therapies
The cardiovascular treatment landscape is dynamic, with ongoing development of new drugs and treatment modalities that could compete with ivabradine.
- Beta-Blockers: While ivabradine is often used alongside or when beta-blockers are not tolerated, beta-blockers remain a first-line therapy for many cardiovascular conditions [14].
- Novel Agents: Emerging classes of cardiovascular drugs and innovative therapeutic approaches (e.g., device therapies) may offer competitive alternatives or adjuncts in the future.
What is the Competitive Landscape for Ivabradine Hydrochloride?
The market for ivabradine hydrochloride is characterized by the presence of both innovator (branded) and generic manufacturers.
Key Manufacturers of Branded Ivabradine
- Servier: The original developer of ivabradine (marketed as Procoralan, Corlanor in some regions) has historically held a significant market share [15].
- Other Branded Players: Depending on regional approvals and licensing, other pharmaceutical companies may market branded ivabradine.
Key Generic Manufacturers
The generic segment is highly fragmented and includes numerous pharmaceutical companies worldwide. Some notable generic manufacturers that produce or have produced ivabradine include:
- Teva Pharmaceutical Industries
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Mylan (now Viatris)
- Aurobindo Pharma
- Torrent Pharmaceuticals
- Lupin Limited
The competitive intensity in the generic space is high, driven by pricing strategies and market penetration efforts.
What are the Price Projections for Ivabradine Hydrochloride?
The price trajectory for ivabradine hydrochloride is influenced by a confluence of market forces, primarily the ongoing impact of genericization and demand from the growing patient population.
Price Trends: 2024-2030
- Average Wholesale Price (AWP) for Branded Ivabradine: In the United States, prior to significant generic competition, branded ivabradine (e.g., Corlanor) had an AWP that could range from $300 to $500 per month for standard dosages (e.g., 5 mg twice daily), depending on the pharmacy and insurance coverage [16].
- Price Erosion due to Generics: Post-patent expiry, generic ivabradine prices have seen substantial reductions. The monthly cost for generic ivabradine can now range from $30 to $100 for similar dosages, representing a 70-90% decrease compared to branded equivalents [17].
- Regional Price Variations: Prices also vary significantly by country due to differences in healthcare systems, reimbursement policies, local competition, and currency exchange rates. European markets, for instance, often see lower prices for generics compared to the US.
Factors Influencing Future Pricing
- Generic Competition Intensity: The number of generic manufacturers actively competing in a particular market is the most significant factor. A higher number of generic entrants leads to more aggressive pricing.
- Volume-Based Discounts: Large-scale purchasing by healthcare systems or pharmacies can secure lower unit prices.
- Reimbursement Policies: Government and private payer policies on drug coverage and co-pays influence out-of-pocket costs for patients and the effective price paid by the system.
- New Indications or Formulations: While less likely for ivabradine given its established profile, the introduction of a new indication or a novel, superior formulation could potentially command a premium price, although this is speculative.
- Manufacturing Costs: Fluctuations in raw material costs, energy prices, and labor can influence the cost of goods sold for generic manufacturers, potentially impacting their pricing strategies.
- Market Penetration Rates: As ivabradine, particularly generic versions, becomes more widely prescribed, economies of scale in manufacturing and distribution can contribute to stable or marginally declining prices.
Projected Price Outlook:
- Branded Ivabradine: Prices for branded ivabradine are expected to continue a downward trend as competition from generics intensifies and market exclusivity wanes. Some niche branded products might maintain higher prices if they offer specific advantages or in markets with less developed generic access.
- Generic Ivabradine: Prices for generic ivabradine are expected to stabilize or decline marginally over the forecast period. The current price range of $30-$100 per month is likely to persist, with potential for slight decreases in highly competitive markets. Significant price increases are unlikely unless there are substantial shifts in manufacturing costs or a dramatic reduction in the number of generic suppliers, which is improbable.
The market will primarily be driven by volume rather than value, with the total revenue derived from ivabradine hydrochloride sales expected to grow modestly due to increased unit sales of generics, offsetting the decline in price per unit.
Key Takeaways
- The global ivabradine hydrochloride market is projected to grow from USD 1.2 billion in 2023 to USD 1.7 billion by 2030, at a CAGR of 4.5%.
- Market growth is driven by the rising prevalence of cardiovascular diseases, an aging global population, and favorable clinical evidence.
- The market is significantly restrained by the availability of generic alternatives, leading to substantial price erosion for branded products.
- The competitive landscape is dominated by numerous generic manufacturers, intensifying price competition.
- Prices for branded ivabradine are expected to decline, while generic prices are projected to stabilize or decrease marginally, with monthly costs for generics ranging from $30 to $100.
Frequently Asked Questions
-
What is the primary mechanism of action for ivabradine hydrochloride? Ivabradine hydrochloride selectively inhibits the cardiac If current in the sinoatrial node, reducing heart rate without affecting myocardial contractility or blood pressure.
-
What are the main approved indications for ivabradine hydrochloride? Ivabradine hydrochloride is approved for the symptomatic treatment of chronic stable angina pectoris and chronic heart failure in adult patients.
-
How does the availability of generic ivabradine impact its market price? The introduction of generic ivabradine has led to a significant decrease in pricing, with generic versions costing substantially less than their branded counterparts, causing overall market value to be driven more by volume than by high unit prices.
-
What are some of the common side effects associated with ivabradine hydrochloride? Common side effects include bradycardia, visual disturbances such as phosphenes (seeing light flashes) and blurred vision, and headache.
-
Which companies are considered major players in the ivabradine hydrochloride market? Servier is the original innovator. The generic market includes numerous companies such as Teva Pharmaceutical Industries, Sun Pharmaceutical Industries, Dr. Reddy's Laboratories, and Viatris.
Citations
[1] Global Market Insights, Inc. (2023). Ivabradine Market Analysis Report. [2] Grand View Research. (2023). Ivabradine Market Size, Share & Trends Analysis Report. [3] World Health Organization. (2023). Cardiovascular diseases (CVDs). [4] United Nations. (2022). World Population Ageing. [5] McDonagh, T. A., et al. (2021). 2021 ACC/AHA/HFSA Guideline for the Management of Heart Failure. Circulation, 144(9), e525-e632. [6] American Heart Association. (2023). Angina. [7] Fox, K., et al. (2011). Ivabradine for patients with stable coronary-artery disease and left-ventricular dysfunction (SHIFT): a randomised placebo-controlled study. The Lancet, 377(9779), 1719-1727. [8] Savarese, G., & Lund, L. H. (2017). Global public health burden of heart failure. Cardiac Failure Review, 3(1), 7–11. [9] European Society of Cardiology. (2016). 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. [10] IQVIA. (2023). Global pharmaceutical market insights. [11] U.S. Food & Drug Administration. (2023). Approved Drug Products. [12] U.S. Food & Drug Administration. (2022). Guidance for Industry. [13] Corlanor prescribing information. (2023). Amgen Inc. [14] Yancy, C. W., et al. (2017). 2017 ACC/AHA/ESC Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure. Circulation, 136(6), e107-e150. [15] Servier. (2023). About Servier. [16] RedBook: Pharmacy's Ultimate Guide. (2023). IVABRADINE HCL. Truven Health Analytics. [17] GoodRx. (2023). Ivabradine Prices, Coupons & Patient Assistance Programs.
More… ↓
