Share This Page
Drug Price Trends for ISOSORBIDE-HYDRALAZINE
✉ Email this page to a colleague
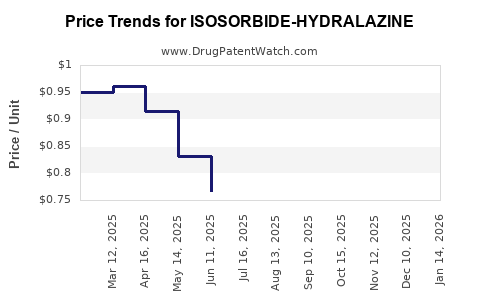
Average Pharmacy Cost for ISOSORBIDE-HYDRALAZINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ISOSORBIDE-HYDRALAZINE 20-37.5 MG TABLET | 72319-0012-03 | 0.74970 | EACH | 2026-05-20 |
| ISOSORBIDE-HYDRALAZINE 20-37.5 MG TABLET | 52536-0006-09 | 0.74970 | EACH | 2026-05-20 |
| ISOSORBIDE-HYDRALAZINE 20-37.5 MG TABLET | 50742-0246-90 | 0.74970 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Isosorbide-Hydralazine
What is the Market Size for Isosorbide-Hydralazine?
Isosorbide-hydralazine is a fixed-dose combination used primarily for managing heart failure and hypertension. It is available both as a generic medication and under brand names like Bidil. The medication exhibits a niche but steady demand driven by its specific indications.
Global Market Overview
- The global heart failure medication market reached approximately USD 18 billion in 2022.
- Isosorbide-hydralazine accounts for an estimated USD 300 million of this segment.
- North America holds the largest share, approximately 45%, driven by high approval rates and prevalence of heart failure.
- The European market constitutes about 25%, with the rest split across Asia-Pacific and other regions.
Market Drivers
- Rising prevalence of heart failure, projected to increase globally from 64 million cases in 2017 to 123 million by 2030 (source: WHO).
- Increasing adoption of combination therapies for heart failure.
- Pediatric and specialized uses for resistant hypertension supporting market continuity.
Competitive Landscape
- Only a few companies hold patents for proprietary formulations, notably Gilead Sciences with Bidil.
- The generic segment is highly competitive, with multiple manufacturers producing lower-cost versions.
- Patent expirations for branded formulations occurred between 2012 and 2018, increasing generic availability.
| Company | Brand | Patent Expiry | Market Share (2022) | Notable Features |
|---|---|---|---|---|
| Gilead Sciences | Bidil | 2018 | 60% (branded) | Approved for African-American patients |
| Generic Manufacturers | Various | — | 40% (generic) | Cost advantage |
Price Trends and Projections
Current Price Points
- Branded formulations (Bidil): approximately USD 200 per month for a standard 30-day supply.
- Generics: USD 40 to USD 80 per month, depending on manufacturer and region.
Price Drivers
- Patent expirations increased generic competition, lowering average prices.
- Manufacturing costs for generics drop due to scale and increased competition.
- Regulatory policies influence pricing, with some regions imposing price caps on essential medications.
Future Price Trends
- Generic prices are projected to decline by 10-20% annually over the next three years.
- Branded prices are unlikely to decrease, maintaining premiums up to 20-25% over generics.
- Potential new entrants or reformulations could stabilize prices or introduce premium formulations.
| Year | Branded Price (USD/month) | Generic Price Range (USD/month) | Change (Compared to 2022) |
|---|---|---|---|
| 2023 | 200 | 40–80 | No change (branded); -5% to -10% (generic) |
| 2024 | 200 | 35–70 | Stable; -10% (generic) |
| 2025 | 200 | 30–60 | Slight decline; -15% (generic) |
Regulatory and Policy Impacts
- US Food and Drug Administration (FDA) and European Medicines Agency (EMA) approvals remain stable.
- Policies favoring biosimilars and generics could accelerate price declines.
- Potential future patent litigations and exclusivity extensions may influence pricing dynamics.
Market Growth Forecasts
- Compound annual growth rate (CAGR) for the bioequivalent market projected at 2-3% from 2023-2030.
- Price erosion in generics offsets volume growth, maintaining overall market stability.
- Indications for heart failure are expanding into early stages, possibly increasing volume demand modestly.
Key Takeaways
- The overall isosorbide-hydralazine market is valued at USD 300 million globally.
- Growth driven by increasing heart failure prevalence and regional adoption.
- Branded prices remain high, but generics are expected to push prices downward by 10-20% annually.
- Market competition is primarily among generic manufacturers, with patent expirations opening opportunities for price reductions.
- Regulatory policy shifts could influence pricing and market share distribution.
FAQs
1. What are the primary indications for isosorbide-hydralazine?
It is primarily indicated for treating heart failure with reduced ejection fraction and resistant hypertension.
2. When did patent protection for Bidil expire?
Patent protection expired in 2018, facilitating generic entry.
3. How might upcoming regulatory changes affect prices?
Regulations favoring generic substitution and biosimilar introduction could accelerate price declines.
4. What regions represent the most significant market opportunities?
North America and Europe lead, but Asia-Pacific presents substantial growth potential due to rising disease prevalence and expanding healthcare infrastructure.
5. Are there developing formulations or combinations in pipeline?
No major reforms or fixed-dose combination modifications are currently announced; market focus remains on generic competition and patent extensions.
References
[1] World Health Organization. (2018). Heart failure: Prevalence and trends.
[2] MarketsandMarkets. (2022). Heart failure therapeutics market report.
[3] FDA. (2022). Approved drug products: BiDil (hydralazine and isosorbide dinitrate).
[4] Gilead Sciences. (2018). Patent and exclusivity information for Bidil.
More… ↓
