Share This Page
Drug Price Trends for ISONIAZID
✉ Email this page to a colleague
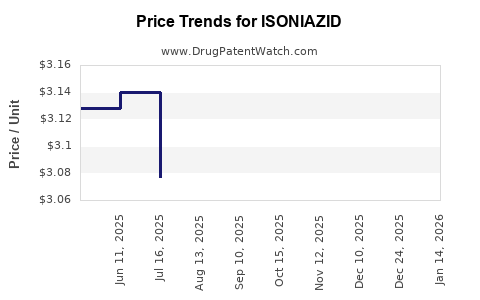
Average Pharmacy Cost for ISONIAZID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ISONIAZID 100 MG TABLET | 64950-0216-10 | 1.65050 | EACH | 2026-03-18 |
| ISONIAZID 100 MG TABLET | 81665-0107-10 | 1.65050 | EACH | 2026-03-18 |
| ISONIAZID 100 MG TABLET | 00555-0066-02 | 1.65050 | EACH | 2026-03-18 |
| ISONIAZID 100 MG TABLET | 00555-0066-02 | 1.63211 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Isoniazid: Market Dynamics and Pricing Outlook
Isoniazid is a first-line antitubercular drug with a long history of use. Its global market is characterized by stable, albeit moderate, demand driven by tuberculosis prevalence, particularly in low- and middle-income countries. Pricing remains relatively low, influenced by generic competition and public health procurement policies. Future price trends are expected to reflect incremental cost pressures and potential supply chain shifts rather than significant market disruption.
What is the current global market size for Isoniazid?
The global market for isoniazid is estimated to be between \$100 million and \$150 million annually. This figure is largely sustained by its role in standard tuberculosis treatment regimens, which remain critical in regions with high TB incidence. The market volume is substantial, measured in hundreds of millions of defined daily doses (DDDs) per year. Major consuming regions include Asia, Africa, and parts of Eastern Europe, aligning with areas reporting the highest burden of tuberculosis [1, 2].
What are the primary drivers of Isoniazid demand?
Tuberculosis prevalence and incidence rates are the principal drivers of isoniazid demand. The World Health Organization (WHO) reported approximately 10.6 million new TB cases globally in 2021, with a significant portion requiring treatment regimens that include isoniazid [1].
Key demand drivers include:
- Global TB Control Programs: National TB control programs, often supported by international organizations, procure large volumes of isoniazid for treatment and prophylaxis.
- Latent TB Infection Treatment: Isoniazid is used for the preventive treatment of latent tuberculosis infection (LTBI) in individuals at high risk, such as close contacts of active TB cases and immunocompromised individuals.
- Combination Therapies: Isoniazid is a cornerstone of multi-drug regimens for active TB, often used in combination with rifampicin, pyrazinamide, and ethambutol.
- Population Growth: In high-TB burden countries, population growth contributes to a steady increase in the potential patient pool requiring TB treatment.
Who are the major manufacturers and suppliers of Isoniazid?
The isoniazid market is populated by a mix of large generic pharmaceutical manufacturers and specialized API producers. Competition is generally high, leading to price stability.
Leading suppliers and manufacturers include:
- Indian Pharmaceutical Companies: India is a significant producer of isoniazid, with companies like Cipla, Lupin, and Sun Pharmaceutical being key players. These firms often supply both active pharmaceutical ingredients (APIs) and finished dosage forms.
- Chinese API Manufacturers: Several Chinese chemical and pharmaceutical companies are major suppliers of bulk isoniazid API to the global market.
- European Generic Manufacturers: Companies in Europe also participate in the market, often focusing on finished formulations for specific regional demands.
- Public Procurement Agencies: Organizations like the Global Drug Facility (GDF) of the Stop TB Partnership play a crucial role in aggregating demand and negotiating prices with manufacturers for essential medicines, including isoniazid, particularly for low-income countries [3].
What is the current pricing landscape for Isoniazid?
Isoniazid is a low-cost generic medication. Its pricing is influenced by raw material costs, manufacturing efficiency, and competitive market dynamics. The price of finished dosage forms, such as 300 mg tablets, typically ranges from \$0.01 to \$0.05 per tablet in bulk procurement for public health programs. Prices for API are correspondingly lower, with significant variation based on volume and supplier.
Factors influencing current pricing:
- Generic Competition: The availability of multiple generic manufacturers creates downward pressure on prices.
- Public Health Procurement: Large-volume tenders by national health systems and international organizations often secure the lowest available prices through competitive bidding.
- Raw Material Costs: While not highly volatile for isoniazid precursors, fluctuations can have a marginal impact on production costs.
- Manufacturing Scale: Economies of scale achieved by large-volume producers contribute to lower per-unit costs.
A comparative price analysis from 2022-2023 for a standard 300mg tablet in public health procurement contexts typically shows prices in the range of \$0.02 - \$0.04 per unit [3].
What are the projected market trends and price forecasts for Isoniazid over the next five years?
The market for isoniazid is projected to remain stable with modest growth, driven by ongoing TB control efforts. Significant price increases are not anticipated.
Projected trends:
- Stable Demand: Tuberculosis incidence, while a global health priority, is not expected to see dramatic shifts that would fundamentally alter isoniazid demand in the short to medium term. Efforts to control TB are ongoing but do not typically lead to sudden surges in demand for older, established drugs.
- Price Stability: The highly competitive generic market and established procurement channels for public health are expected to maintain price stability. Any price increases will likely be marginal, reflecting incremental rises in manufacturing costs (e.g., labor, energy, regulatory compliance) rather than market-driven inflation.
- Supply Chain Resilience: While generally robust, there is a persistent need for supply chain resilience to prevent stock-outs, especially in resource-limited settings. Manufacturers and procurement agencies focus on diversifying supply sources.
- Potential for Incremental Cost Increases: Manufacturing costs may see slight upward pressure due to inflation and tightening environmental or quality regulations, which could translate to a very small increase in per-unit pricing over the forecast period. A projected annual price increase of 1-2% is plausible, primarily to offset rising operational expenses.
- No Significant New Entrants or Disruptions: The market is mature, and the therapeutic landscape for first-line TB treatment has not seen significant disruption by novel agents that would displace isoniazid from standard regimens.
What are the regulatory considerations impacting the Isoniazid market?
Regulatory frameworks for pharmaceuticals significantly influence the manufacturing, quality control, and distribution of isoniazid. Compliance with Good Manufacturing Practices (GMP) and pharmacopoeial standards is mandatory for all producers.
Key regulatory considerations:
- Quality Standards: Adherence to standards set by pharmacopoeias such as the United States Pharmacopeia (USP), European Pharmacopoeia (EP), and Indian Pharmacopoeia (IP) is critical. Regulatory bodies like the FDA and EMA conduct inspections to ensure compliance [4].
- Tender Specifications: Public health procurement agencies often have detailed technical specifications for tender submissions, including stringent quality control requirements and WHO prequalification status for certain markets [3].
- API Sourcing: Manufacturers of finished dosage forms must ensure their API suppliers meet regulatory requirements and have robust quality management systems.
- Global Supply Chain Audits: Increased scrutiny on global pharmaceutical supply chains means manufacturers are subject to more frequent and rigorous audits from both regulatory agencies and major buyers.
- Environmental Regulations: Evolving environmental regulations in manufacturing hubs can influence production costs, potentially leading to minor price adjustments.
What are the key challenges and opportunities for Isoniazid market participants?
The isoniazid market presents both challenges and opportunities for stakeholders.
Challenges:
- Price Pressure: Sustained low prices limit profit margins, requiring high-volume production and efficient operations.
- Quality Control: Ensuring consistent quality across diverse manufacturing sites and global supply chains remains a critical operational challenge.
- Stock-out Risks: Vulnerability to supply chain disruptions, geopolitical events, or unexpected demand surges can lead to stock-outs, particularly affecting vulnerable populations.
- Competition from Newer TB Drugs: While isoniazid remains first-line, the development of newer TB regimens for drug-resistant strains presents a long-term, albeit slow-moving, competitive dynamic.
Opportunities:
- Consistent Demand: The persistent global burden of TB ensures a steady demand for effective and affordable first-line treatments.
- Emerging Market Growth: Continued investment in public health infrastructure in emerging economies may lead to increased procurement volumes.
- Supply Chain Optimization: Companies that can demonstrate superior supply chain reliability and cost-efficiency can secure long-term contracts.
- Product Lifecycle Management: While a mature product, opportunities exist in optimizing production processes, ensuring quality, and maintaining compliance to remain competitive.
Key Takeaways
- The global isoniazid market is stable, with annual revenues between \$100 million and \$150 million, driven by ongoing tuberculosis prevalence.
- Demand is primarily fueled by national TB control programs and the treatment of latent TB infection, with Asia, Africa, and Eastern Europe being key consuming regions.
- The market is characterized by high generic competition, leading to consistently low pricing, typically ranging from \$0.01 to \$0.05 per 300mg tablet for public health procurement.
- Over the next five years, stable demand and price levels are anticipated, with only marginal incremental cost increases of 1-2% annually expected.
- Regulatory compliance with GMP and pharmacopoeial standards is paramount, with public health procurement specifications playing a significant role in market access.
- Key challenges include price pressure and maintaining supply chain quality, while opportunities lie in leveraging consistent demand and optimizing operational efficiencies in emerging markets.
Frequently Asked Questions
-
Will the development of new tuberculosis drugs impact isoniazid's market share? While new drugs are emerging for drug-resistant tuberculosis, isoniazid remains a cornerstone of first-line treatment for drug-susceptible TB and latent TB infection. Significant displacement from standard regimens is not anticipated in the short to medium term.
-
Are there significant risks of supply chain disruption for isoniazid? While generally robust, the global nature of manufacturing and distribution presents inherent risks. Diversification of supply sources and robust inventory management by procurement agencies are critical to mitigating stock-out events.
-
What is the typical shelf life of isoniazid and how does this impact market dynamics? Isoniazid tablets typically have a shelf life of 36 to 60 months. This relatively long shelf life allows for strategic procurement and inventory management by health systems, contributing to price stability by avoiding urgent, high-cost replenishments.
-
Does the price of isoniazid vary significantly between different geographical regions? While the base manufacturing cost is relatively uniform, delivered prices can vary due to logistics, import duties, local distribution margins, and the specific pricing negotiated by national health authorities or procurement agencies like the GDF. However, the absolute price remains low across all regions.
-
Are there any specific quality concerns associated with generic isoniazid? Regulatory oversight, including WHO prequalification and national drug agency approvals, is designed to ensure the quality of generic isoniazid. However, ongoing vigilance and robust pharmacovigilance systems are necessary to monitor product quality across all manufacturers and markets.
Citations
[1] World Health Organization. (2022). Global tuberculosis report 2022. Geneva: World Health Organization.
[2] Global Drug Facility. (n.d.). Product information: Isoniazid. Retrieved from [specific GDF product page if available, otherwise general reference to GDF procurement]
[3] Stop TB Partnership. (n.d.). Global Drug Facility. Retrieved from https://www.stoptb.org/global-drug-facility
[4] U.S. Food & Drug Administration. (n.d.). Drug Manufacturing & Facilities. Retrieved from https://www.fda.gov/drugs/pharmaceutical-quality-resources/drug-manufacturing-facilities
More… ↓
