Share This Page
Drug Price Trends for IPRATROPIUM-ALBUTEROL
✉ Email this page to a colleague
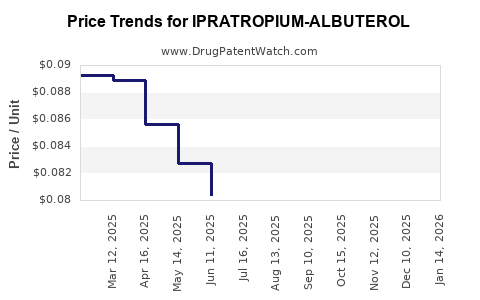
Average Pharmacy Cost for IPRATROPIUM-ALBUTEROL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IPRATROPIUM-ALBUTEROL 0.5-3(2.5) MG/3 ML | 00378-9671-60 | 0.08390 | ML | 2026-05-20 |
| IPRATROPIUM-ALBUTEROL 0.5-3(2.5) MG/3 ML | 00378-9671-93 | 0.08739 | ML | 2026-05-20 |
| IPRATROPIUM-ALBUTEROL 0.5-3(2.5) MG/3 ML | 00487-0201-01 | 0.08739 | ML | 2026-05-20 |
| IPRATROPIUM-ALBUTEROL 0.5-3(2.5) MG/3 ML | 00378-9671-64 | 0.08739 | ML | 2026-05-20 |
| IPRATROPIUM-ALBUTEROL 0.5-3(2.5) MG/3 ML | 76204-0600-60 | 0.08390 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Ipratropium-Albuterol
What is the current market landscape for Ipratropium-Albuterol?
Ipratropium-Albuterol is a combination inhaler used primarily for chronic obstructive pulmonary disease (COPD) and asthma. It combines two active ingredients: ipratropium bromide, an anticholinergic, and albuterol sulfate, a beta-2 adrenergic agonist. The combination offers rapid bronchodilation and improved symptom control.
The global respiratory drugs market reached approximately $41 billion in 2022. Ipratropium-Albuterol inhalers represent a significant segment due to the high prevalence of COPD and asthma, especially in North America and Europe. The drug is available via both brand-name and generic formulations, with the former characterized by higher prices.
Major manufacturers include Boehringer Ingelheim, Teva Pharmaceuticals, Cipla, and Mylan. The availability of generics has increased price competition, impacting market dynamics.
How is the market expected to evolve over the next five years?
Market growth projections
The global respiratory drugs market is projected to grow at a Compound Annual Growth Rate (CAGR) of 4.5% from 2023 to 2028. Ipratropium-Albuterol will benefit from increased COPD prevalence, aging populations, and rising respiratory disease awareness.
Key factors influencing growth
- Prevalence increase: COPD affects over 200 million globally, with a rising trend in low- and middle-income countries.
- Regulatory developments: Approval of new inhaler devices and formulations expands market options.
- Generic penetration: A surge in generic versions lowers prices and increases accessibility, but may narrow profit margins for branded products.
Emerging markets
Growth in Asia-Pacific, Latin America, and Africa driven by rising urban pollution and tobacco use. These markets' demand for affordable inhalers will shape supply chains and pricing strategies.
What are the price trends and projections?
Current pricing overview
- Brand-name Ipratropium-Albuterol inhalers: Range between $50 and $80 per inhaler (approximately 200 doses).
- Generic alternatives: Range between $20 and $40 per inhaler.
- Insurance impact: Reimbursements vary, with higher coverage in developed countries, influencing out-of-pocket costs.
Price evolution forecast
- Next 3 years: Expect a plateau or slight decline (1-3%) owing to increased generic competition.
- By 2028: Prices for branded inhalers could decrease by up to 15%, while generics stabilize at current levels or decrease further.
Regional price disparities
- North America maintains the highest prices due to regulatory and patent protections.
- Asia-Pacific and similar regions will see the most significant price reductions due to local manufacturing and increased generic choices.
What are the competitive and regulatory factors affecting pricing?
- Patent expirations: Several branded inhalers face patent cliffs between 2023 and 2026, leading to increased generic availability.
- Regulatory approval processes: Faster pathways for generics, especially in the U.S. (via ANDA), expedite market entry and price reductions.
- Pricing policies: Countries such as Canada and the UK employ price caps and reimbursement controls, impacting retail prices.
What are the R&D trends and potential innovations?
Pharmaceutical companies invest in inhaler technology improvements, targeting enhanced drug delivery and reduced manufacturing costs. Next-generation inhalers with smart technology or better pharmacokinetics could command premium pricing but are currently in early development phases.
Summary of key metrics
| Parameter | Data Point |
|---|---|
| 2023 global market size | $3.9 billion (estimated for Ipratropium-Albuterol segment) |
| 2028 projected market size | $5.4 billion (assuming growth aligned with respiratory drugs CAGR) |
| Price range (brand-name) | $50–$80 per inhaler |
| Price range (generic) | $20–$40 per inhaler |
| CAGR (2023–2028) | 4.5% |
| Patent expiry window | 2023–2026 |
Key Takeaways
- The Ipratropium-Albuterol market will grow gradually, driven by disease prevalence and demographic shifts.
- Pricing will decrease, especially in markets with high generic penetration.
- Industry innovation focuses on improved delivery devices and formulations.
- Patent expirations will accelerate generic competition and price reductions.
FAQs
1. How will patent expirations impact Ipratropium-Albuterol prices?
Patent expirations between 2023 and 2026 will allow generic manufacturers to enter the market, leading to lower prices and increased competition.
2. Are there new formulations or delivery systems in development?
Yes. Companies are developing inhalers with enhanced drug delivery, digital dose tracking, and easier administration, which may command premium prices.
3. Which regions will see the fastest growth?
The Asia-Pacific region poses the fastest growth opportunity due to increasing COPD prevalence and demand for affordable inhalers.
4. How do regulatory schemes influence market pricing?
Price caps, reimbursement policies, and accelerated approval pathways in different jurisdictions significantly impact retail and wholesale pricing.
5. Will branded inhalers maintain high prices?
Branded inhalers will likely decline due to competition but may retain premium pricing where patent protection remains or for advanced delivery devices.
References
[1] GlobalData. (2022). Respiratory drugs market overview.
[2] IQVIA. (2022). World drug price and reimbursement report.
[3] U.S. Food and Drug Administration (FDA). (2022). ANDA approvals and patent expirations.
[4] World Health Organization (WHO). (2022). COPD prevalence report.
[5] EvaluatePharma. (2023). Prescription drug price trends.
More… ↓
