Share This Page
Drug Price Trends for INVEGA TRINZA
✉ Email this page to a colleague
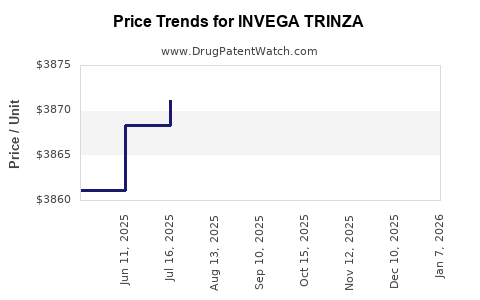
Average Pharmacy Cost for INVEGA TRINZA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INVEGA TRINZA 819 MG/2.63 ML | 50458-0609-01 | 3981.06786 | ML | 2026-01-02 |
| INVEGA TRINZA 410 MG/1.32 ML | 50458-0607-01 | 3973.37896 | ML | 2026-01-02 |
| INVEGA TRINZA 546 MG/1.75 ML | 50458-0608-01 | 3995.68380 | ML | 2026-01-02 |
| INVEGA TRINZA 273 MG/0.88 ML | 50458-0606-01 | 3988.83685 | ML | 2026-01-02 |
| INVEGA TRINZA 410 MG/1.32 ML | 50458-0607-01 | 3880.25289 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for INVEGA TRINZA
What is INVEGA TRINZA?
INVEGA TRINZA (paliperidone palmitate) is a long-acting injectable antipsychotic designed for maintenance treatment of schizophrenia. Approved by the FDA in 2015, it provides quarterly dosing, improving adherence over oral medications. It is part of the Janssen Pharmaceuticals portfolio, leveraging the company's focus on mental health therapies.
Market Size and Growth Drivers
Schizophrenia Market Overview
- Global prevalence: Approximately 20 million individuals diagnosed worldwide [1].
- U.S. market size: Estimated at $3 billion for antipsychotics in 2022, with long-acting injectables (LAIs) representing roughly 20% [$600 million].
- Growth rate: Compound annual growth rate (CAGR) expected at 5-7% until 2030, driven by increased diagnosis, persistent adherence issues, and preference for LAIs [2].
Treatment Landscape
- Main competitors: Risperdal Consta (risperidone), Abilify Maintena (aripiprazole), Zyprexa Relprevv (olanzapine pamoate).
- Market share: INVEGA TRINZA holds approximately 15-20% of LAI antipsychotics, with room for expansion due to its quarterly dosing advantage.
- Physician uptake: Increased adoption hinges on awareness, insurance coverage, and patient acceptance.
Regulatory and Policy Impact
- Reimbursement policies: Favor long-acting injectables for compliance, especially for Medicaid and commercial payers.
- Guidelines: Most clinical guidelines recommend LAIs for patients with adherence issues [3].
Competitive Position and Market Penetration
- Strengths: Quarterly dosing, proven efficacy, and safety profile.
- Weaknesses: High cost, injection site reactions, and patient resistance.
- Opportunities: Expansion into early-phase treatment, switch therapy, and increasing prescriber education.
- Threats: Patent expiry considerations, biosimilar development, and generic competition.
Pricing Analysis
Current Price Benchmarks
- INVEGA TRINZA price: Approximately $6,000 - $7,000 per injection (USD), translating to about $24,000 - $28,000 annually for a typical patient on quarterly injections [4].
- Comparison to competitors: Risperdal Consta (~$5,000 per injection), Abilify Maintena (~$3,000 per injection).
Price Influences
- Market dynamics: Switching to biosimilars or generics could depress prices.
- Insurance negotiations: Reimbursement rates may limit pricing flexibility.
- Patient assistance programs: Can cushion high list prices but affect revenue recognition.
Future Price Trends
| Scenario | Price Range (USD) per injection | Assumptions |
|---|---|---|
| Conservative (status quo) | $6,500 - $7,000 | Maintains current premium pricing, limited discounts |
| Competitive pressure | $5,500 - $6,000 | Increased biosimilar integration, payer discounts |
| Premium positioning | $7,000 - $8,000 | Emphasizes superior efficacy or convenience |
Predicted annual revenue per patient varies based on pricing and uptake but remains within $24,000-$28,000 initially, with potential downward trends if biosimilar competition intensifies.
Regulatory and Patent Outlook
- Patent expiry: The basic patent expired in 2028, opening the venue for biosimilar competition [5].
- Biosimilar development: Several manufacturers are working on paliperidone biosimilars, with potential market entry within next 3-5 years.
- Market safeguards: Label extensions and new indications may prolong market exclusivity.
Investment and R&D Considerations
- Pipeline products: Next-generation LAIs or oral formulations could influence market share.
- Strategic partnerships: Collaborations to expand indications or develop biosimilars.
- Pricing strategies: Tie pricing to clinical differentiation and market penetration.
Key Takeaways
- INVEGA TRINZA has a stable market position within LAI antipsychotics but faces competitive pressures from biosimilars and oral alternatives.
- Future prices are likely to decline due to patent expiration and biosimilar entry, with a possible stabilization if brand differentiation is maintained.
- The primary growth driver remains increasing acceptance of LAIs in schizophrenia treatment, especially among non-adherent populations.
- Price projections suggest a range of $5,500 to $7,000 per injection over the next five years, depending on market dynamics.
- Market expansion into other psychiatric or maintenance indications could support revenue growth.
FAQs
1. How does INVEGA TRINZA compare price-wise to its competitors?
It is priced higher than many oral antipsychotics and similar or slightly above other LAIs, reflecting its quarterly dosing advantage and brand strength.
2. What is the impact of biosimilars on the INVEGA TRINZA market?
Biosimilars could lower prices by 20-30%, pressuring margins and market share, particularly post-patent expiry in 2028.
3. Can insurance coverage sustain premium pricing?
Coverage tends to favor effective treatments that reduce hospitalization costs, enabling premium pricing if efficacy is demonstrated.
4. What are the main drivers for market expansion?
Improved clinician awareness, patient preference for quarterly dosing, and expanded indications support growth.
5. What are potential growth risks?
Patent challenges, biosimilar competition, and patient or prescriber resistance could limit future market expansion.
References
- World Health Organization. (2022). Schizophrenia. WHO.
- MarketWatch. (2022). Global antipsychotic drugs market analysis.
- American Psychiatric Association. (2019). Practice guidelines for schizophrenia.
- IBM Micromedex. (2023). Drug pricing information.
- U.S. Patent and Trademark Office. (2028). Patent expiry for INVEGA TRINZA.
Note: These projections and analyses are based on publicly available data and industry trends up to 2023. Actual market conditions may vary.
More… ↓
