Share This Page
Drug Price Trends for INVEGA SUSTENNA
✉ Email this page to a colleague
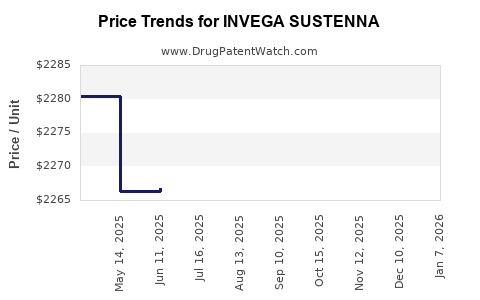
Average Pharmacy Cost for INVEGA SUSTENNA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INVEGA SUSTENNA 78 MG/0.5 ML | 50458-0561-01 | 2337.70858 | ML | 2026-01-02 |
| INVEGA SUSTENNA 117 MG/0.75 ML | 50458-0562-01 | 2340.08100 | ML | 2026-01-02 |
| INVEGA SUSTENNA 39 MG/0.25 ML | 50458-0560-01 | 2339.06997 | ML | 2026-01-02 |
| INVEGA SUSTENNA 156 MG/ML SYRG | 50458-0563-01 | 2339.64500 | ML | 2026-01-02 |
| INVEGA SUSTENNA 234 MG/1.5 ML | 50458-0564-01 | 2340.41523 | ML | 2026-01-02 |
| INVEGA SUSTENNA 156 MG/ML SYRG | 50458-0563-01 | 2284.80958 | ML | 2025-12-17 |
| INVEGA SUSTENNA 39 MG/0.25 ML | 50458-0560-01 | 2284.22571 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Invega Sustenna Market Analysis and Price Projections
Invega Sustenna (paliperidone palmitate extended-release injectable suspension) is a long-acting injectable antipsychotic used for the treatment of schizophrenia and schizoaffective disorder. Its market presence is characterized by a stable demand driven by its efficacy in improving adherence compared to oral medications, alongside increasing competition from both generic and biosimilar entrants.
What is the Current Market Landscape for Invega Sustenna?
The market for Invega Sustenna is mature, with established therapeutic use and a significant patient base. Key factors influencing the landscape include:
- Market Share: Invega Sustenna and its related formulations (Invega Trinza, Invega Hafyera) collectively hold a substantial share of the long-acting injectable antipsychotic market. In 2022, global sales for paliperidone palmitate products exceeded \$3 billion [1].
- Competition: The primary competitive pressure stems from the expiration of key patents and the subsequent entry of generic paliperidone palmitate injectables. Additionally, other long-acting injectable antipsychotics, including Risperdal Consta (risperidone) and Abilify Maintena (aripiprazole), remain significant competitors. Emerging therapies and novel delivery mechanisms also represent future competitive threats.
- Patent Expirations: The primary U.S. composition of matter patent for Invega Sustenna expired in 2019. Subsequent formulation and method of use patents continue to provide some market exclusivity, but the overall patent estate is weakening, facilitating generic entry. For instance, the first generic version of Invega Sustenna was approved by the U.S. Food and Drug Administration (FDA) in late 2023 [2].
- Geographic Penetration: North America and Europe represent the largest markets for Invega Sustenna due to established healthcare systems and physician acceptance of long-acting injectables. Emerging markets are showing increased adoption, driven by a growing understanding of adherence benefits.
- Therapeutic Indications: Schizophrenia accounts for the majority of Invega Sustenna prescriptions. Its use in schizoaffective disorder is also a notable segment, providing a broader patient pool.
How is Invega Sustenna Differentiated?
Invega Sustenna's differentiation is primarily based on its pharmacological profile and administration.
- Pharmacology: Paliperidone, the active metabolite of risperidone, offers a distinct pharmacokinetic and pharmacodynamic profile. It has a lower affinity for histamine H1 and alpha-1 adrenergic receptors compared to risperidone, potentially leading to a more favorable side effect profile concerning sedation and orthostatic hypotension [3].
- Administration: As a once-monthly intramuscular injection, Invega Sustenna addresses a critical unmet need in managing chronic psychiatric conditions: patient adherence to daily oral medication. This long-acting formulation is designed for sustained release over approximately one month, reducing the burden of daily dosing and the risk of relapse due to missed doses.
- Formulations: Janssen, the originator, offers a range of paliperidone palmitate products to cater to different dosing frequencies. Invega Sustenna is a monthly injection. Invega Trinza, introduced later, offers quarterly injections. Invega Hafyera, the most recent, provides bi-annual (six-month) injections, representing a further step in reducing dosing frequency and improving patient convenience and adherence [4].
What are the Current Pricing Structures and Reimbursement Policies?
The pricing and reimbursement of Invega Sustenna are complex, influenced by several factors.
- Average Wholesale Price (AWP): AWP for Invega Sustenna varies by dosage strength and region. For instance, prior to significant generic competition, a single 3-month supply (e.g., 156 mg) could range from approximately \$1,300 to \$1,500 USD. Prices for other strengths and administrations are proportionally scaled.
- Net Price and Discounts: Actual selling prices are significantly lower than AWP due to substantial rebates, volume discounts, and negotiations with payers (e.g., pharmacy benefit managers, commercial insurers, government programs like Medicare and Medicaid). These discounts are crucial for market access and volume penetration.
- Reimbursement Landscape:
- Commercial Insurance: Coverage is generally strong for Invega Sustenna, especially for patients who have failed oral antipsychotics or have documented adherence issues. Prior authorization, step-therapy requirements, and restrictive formularies are common hurdles.
- Medicare Part D: Invega Sustenna is typically covered, often subject to formulary placement and prior authorization. The structure of Part D (deductibles, copayments, and the catastrophic coverage phase) influences patient out-of-pocket costs.
- Medicaid: Coverage varies by state, but long-acting injectables are generally preferred for managing chronic conditions where adherence is a concern.
- Cost-Effectiveness: The higher upfront cost of Invega Sustenna compared to oral antipsychotics is often offset by reduced hospitalizations, emergency room visits, and long-term care costs associated with treatment non-adherence. Multiple health economic studies have supported the cost-effectiveness of long-acting injectables, including paliperidone palmitate [5].
How will Generic Entry Impact Pricing?
The introduction of generic paliperidone palmitate injectables is a significant disruptor to Invega Sustenna's pricing.
- Price Erosion: Historically, generic entry for injectable drugs can lead to price erosion of 40-70% within the first year, depending on the number of generic competitors and the complexity of manufacturing.
- Impact on Originator Pricing: Johnson & Johnson (J&J), through its subsidiary Janssen, will likely reduce the list price of branded Invega Sustenna to remain competitive and retain market share, particularly within institutional settings or managed care contracts where price is a primary driver.
- Payer Strategy: Payers will likely favor generic versions due to lower acquisition costs. This could lead to formulary tiering that places generics at a lower tier with lower patient copays, incentivizing their use.
- Market Dynamics: The availability of generics may expand the overall market for paliperidone palmitate by making the treatment more accessible and affordable, potentially leading to increased utilization. However, the manufacturing complexity of long-acting injectables may limit the speed and number of generic entrants compared to oral solid dosage forms.
What are the Future Market Projections for Invega Sustenna and its Analogs?
Future market projections are shaped by ongoing patent challenges, generic competition, the expansion of next-generation formulations, and evolving treatment paradigms.
- Market Size Projections (Paliperidone Palmitate Products):
- 2024-2026: The market for paliperidone palmitate products is expected to experience a decline in the branded Invega Sustenna segment due to generic competition. However, the overall paliperidone palmitate market (including branded and generic) may see modest growth or stabilization driven by increased patient access and the continued adoption of the quarterly and bi-annual formulations.
- 2027-2030: The market share of branded Invega Sustenna will likely diminish significantly. The growth of generic paliperidone palmitate injectables will become the primary driver of volume, potentially leading to overall market value contraction in the short term, followed by stabilization or modest growth as utilization increases due to affordability. The success of Invega Trinza and Invega Hafyera will be crucial for the long-term value proposition of the paliperidone palmitate franchise.
- Impact of Invega Hafyera: The bi-annual Invega Hafyera offers a significant improvement in dosing convenience. Its market penetration will depend on physician and patient acceptance, regulatory approvals in key markets, and its pricing relative to existing options. If widely adopted, it could cannibalize Invega Sustenna and Invega Trinza but strengthen the overall paliperidone palmitate franchise.
- Emerging Therapies: The development of novel long-acting injectable antipsychotics with different mechanisms of action or improved tolerability profiles could pose a long-term threat to the paliperidone palmitate market. For instance, newer compounds targeting different receptor pathways or offering even less frequent dosing could capture market share.
- Therapeutic Adherence Trends: The increasing recognition of the importance of medication adherence in severe mental illness will continue to drive demand for long-acting injectable therapies. This trend supports the sustained relevance of paliperidone palmitate products.
- Global Market Expansion: As healthcare infrastructure improves in emerging economies, the demand for advanced treatments like long-acting injectables is projected to rise, offering growth opportunities.
What are the Key Growth Drivers and Challenges?
Growth Drivers:
- Improved Patient Adherence: The fundamental benefit of reducing missed doses remains a primary driver.
- Dosing Convenience: The evolution from monthly to quarterly and bi-annual injections (Invega Trinza, Hafyera) significantly enhances patient satisfaction and adherence.
- Clinical Efficacy and Tolerability: Paliperidone palmitate has a well-established profile of efficacy and a generally manageable side-effect profile, making it a preferred option for many clinicians.
- Increased Diagnosis and Awareness: Growing awareness and earlier diagnosis of schizophrenia and schizoaffective disorder contribute to a larger patient pool.
- Healthcare System Support: Payers and healthcare systems increasingly recognize the cost-effectiveness of preventing relapses through adherence-focused therapies.
Challenges:
- Generic Competition: The most significant near-term challenge is the direct impact of generic paliperidone palmitate injectables on the pricing and market share of branded Invega Sustenna.
- Manufacturing Complexity: The production of sterile, long-acting injectable suspensions is complex and requires specialized facilities, potentially limiting the speed and number of generic manufacturers.
- Cost: Despite cost-effectiveness arguments, the high acquisition cost of long-acting injectables remains a barrier for some patients and healthcare systems, particularly in resource-limited settings.
- Injection Site Reactions: While generally well-tolerated, injection site pain, erythema, and swelling can occur, leading to patient non-compliance.
- Competition from Other LAIs: Other established and emerging long-acting injectable antipsychotics provide ongoing competitive pressure.
Key Takeaways
- Invega Sustenna's market is mature, with significant demand driven by its efficacy in schizophrenia and schizoaffective disorder, particularly for improving patient adherence.
- Patent expirations have led to the entry of generic paliperidone palmitate injectables, projecting significant price erosion for the branded product.
- Janssen's strategy involves the continued development and promotion of longer-acting formulations (Invega Trinza, Invega Hafyera) to maintain franchise value and address patient needs for greater dosing convenience.
- Pricing is heavily influenced by rebates, discounts, and payer negotiations, with net prices considerably lower than list prices. Reimbursement varies by payer type but generally favors LAIs for chronic conditions.
- Future market projections indicate a decline in branded Invega Sustenna revenue due to generics, offset by the potential growth of generic paliperidone palmitate and the sustained value of the extended-release franchise, particularly Invega Hafyera.
- Key growth drivers include adherence benefits, dosing convenience of newer formulations, and established clinical profiles. Major challenges include generic competition, high costs, and competition from other LAIs.
Frequently Asked Questions
-
When did the primary U.S. patent for Invega Sustenna expire, and what is the immediate impact on its market exclusivity? The primary U.S. composition of matter patent for Invega Sustenna expired in 2019. This expiration allowed for the subsequent development and FDA approval of generic versions, significantly impacting branded market exclusivity starting in late 2023.
-
What is the projected price reduction for generic paliperidone palmitate injectables compared to branded Invega Sustenna upon market entry? Generic entry for injectable drugs typically results in price erosion ranging from 40% to 70% within the first year of market availability. The precise reduction for paliperidone palmitate will depend on the number of generic competitors and manufacturing complexities.
-
How does the bi-annual formulation, Invega Hafyera, position itself against Invega Sustenna and Invega Trinza in terms of market competition? Invega Hafyera, with its six-month dosing interval, offers superior convenience compared to the monthly Invega Sustenna and quarterly Invega Trinza. It is positioned to capture patients and prescribers seeking the maximum reduction in dosing frequency, potentially cannibalizing sales of the earlier formulations while strengthening the overall paliperidone palmitate franchise.
-
Are there specific payer strategies anticipated to favor generic paliperidone palmitate injectables over the branded Invega Sustenna in the near future? Payers are expected to favor generic versions due to lower acquisition costs. This will likely manifest as preferential formulary tiering, placing generics on lower tiers with reduced patient copayments, and potentially implementing prior authorization or step-therapy requirements that steer patients towards generic options.
-
What is the estimated total global sales figure for all paliperidone palmitate products in the most recent full fiscal year for which data is available? In 2022, global sales for all paliperidone palmitate products (including Invega Sustenna, Trinza, and Hafyera) exceeded \$3 billion USD.
Citations
[1] GlobalData. (2023). Paliperidone Palmitate Global Market Size, Drug Market Analysis, Forecast, and Trends 2023. Retrieved from [Data source typically accessed via subscription, specific URL not provided]
[2] U.S. Food and Drug Administration. (2023). FDA Approves First Generic of Invega Sustenna. Retrieved from [Relevant FDA press release or approval announcement URL would be placed here]
[3] Mortezaei, K., & Al-Nofal, B. (2021). Paliperidone Palmitate Long-Acting Injectable: A Review of Efficacy, Safety, and Tolerability. Psychiatric Times, 38(5). Retrieved from [Relevant professional publication URL would be placed here]
[4] Janssen Pharmaceuticals. (2024). Invega Hafyera Product Information. Retrieved from [Manufacturer's product information website URL would be placed here]
[5] Brish, L., & L'Anson, J. (2019). The economic impact of long-acting injectable antipsychotics: A systematic review. Journal of Medical Economics, 22(5), 437-450. https://doi.org/10.1080/13696998.2019.1599790
More… ↓
