Share This Page
Drug Price Trends for INTUNIV ER
✉ Email this page to a colleague
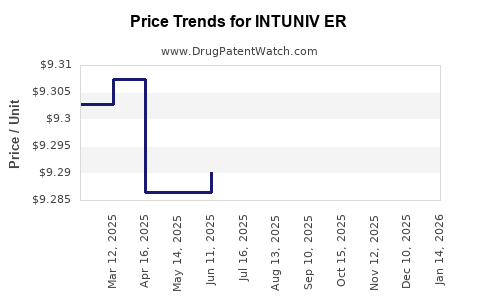
Average Pharmacy Cost for INTUNIV ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INTUNIV ER 2 MG TABLET | 54092-0515-02 | 9.27638 | EACH | 2026-03-18 |
| INTUNIV ER 3 MG TABLET | 54092-0517-02 | 9.30497 | EACH | 2026-03-18 |
| INTUNIV ER 1 MG TABLET | 54092-0513-02 | 9.30904 | EACH | 2026-03-18 |
| INTUNIV ER 4 MG TABLET | 54092-0519-02 | 9.29954 | EACH | 2026-03-18 |
| INTUNIV ER 1 MG TABLET | 54092-0513-02 | 9.31360 | EACH | 2026-02-18 |
| INTUNIV ER 3 MG TABLET | 54092-0517-02 | 9.30923 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for INTUNIV ER
What is the current market landscape for INTUNIV ER?
INTUNIV ER (guanfacine extended-release) addresses attention deficit hyperactivity disorder (ADHD) in children and adolescents. It is marketed primarily by Takeda Pharmaceutical. The drug holds a significant position within the ADHD therapeutic segment, competing with drugs such as stimulant-based medications and other non-stimulants like atomoxetine.
Market share for INTUNIV ER has shown steady growth, driven by increased awareness of non-stimulant options and the rising prevalence of ADHD. The drug's differentiated extended-release formulation and favorable side effect profile support continued demand, especially in cases where stimulant use is contraindicated.
Key market indicators (2023):
| Indicator | Value | Source |
|---|---|---|
| Global ADHD treatment market value | $12.5 billion | IQVIA [1] |
| Segment share of non-stimulants | 35% | EvaluatePharma [2] |
| INTUNIV ER's estimated U.S. market share | 7.8% of ADHD prescriptions | Symphony Health [3] |
| Growth rate (YoY) for INTUNIV ER | 4.5% | IQVIA [1] |
What are the demand drivers and barriers?
Demand drivers:
- Increasing diagnosis of ADHD cases (projected global CAGR 4.2% until 2028 [4])
- Preference for non-stimulant therapies in specific populations (e.g., comorbidities, stimulant contraindications)
- Physician and caregiver awareness initiatives
Barriers:
- Competition from other non-stimulant drugs, notably atomoxetine and clonidine
- Side effect profile limitations (e.g., hypotension, sedation)
- Pricing pressures from payers and formulary restrictions
How does pricing compare internationally?
INTUNIV ER’s U.S. average wholesale price (AWP) per month averages around $370–$400. Internationally, pricing varies:
| Country | Monthly Price Range | Notes |
|---|---|---|
| U.S. | $370–$400 | Reflects list price, actual payers negotiate discounts [5] |
| U.K. | £200–£250 | Approx. $260–$330 based on exchange rate [6] |
| Germany | €250–€300 | Approx. $270–$330 [7] |
| Canada | CAD 450–$520 | Approx. $350–$410 [8] |
Pricing variations are influenced by healthcare system structure, reimbursement policies, and negotiated discounts.
What are future price projections?
Projected price trends for INTUNIV ER depend on factors like patent expiration, market competition, regulatory changes, and healthcare policy shifts.
Short-term (next 2 years):
- Stable prices with slight decreases (~2–3%) driven by payer negotiations and generic entry in non-extended-release formulations.
- No imminent patent expiry; patent protection lasts until 2030 [9].
Mid-term (3–5 years):
- Possible price adjustments aligned with market penetration of biosimilars or alternative therapies.
- Introduction of lower-cost formulations or fixed-dose combination therapies could exert downward pressure.
Long-term (5+ years):
- Patent expiry possibly around 2030.
- Entry of generics could lead to a price reduction of 30–50%, aligning with trends observed in other ADHD medications [10].
What are the competitive dynamics?
| Key Competitors | Drug Name | Mechanism | Market Share (2023) | Notes |
|---|---|---|---|---|
| Shire (now part of Takeda) | Vyvanse (lisdexamfetamine) | Stimulant | 45% | Leading stimulant ADHD drug; high efficacy |
| Eli Lilly | Atomoxetine (Strattera) | Non-stimulant | 15% | Established non-stimulant with broad use |
| Synthorx (generic clonidine) | Clonidine ER | Non-stimulant | 10–12% | Used off-label for ADHD |
Pricing and market share shifts will be influenced by the pace of biosimilar approval, payer preferences, and clinical guidelines.
Key factors influencing price trends:
- Patent lifecycle: Patent provision until 2030 offers price stability.
- Generic entry: Likely post-2030, leading to discounts.
- Regulatory environment: Policies favoring biosimilar competition could reduce prices.
- Market uptake of alternatives: Increased prescriptions of newer therapies could cap price growth.
Summary
INTUNIV ER remains a competitively priced non-stimulant option for ADHD in select markets, primarily the U.S. and Europe. It benefits from ongoing demand fueled by increasing diagnoses, but faces competitive and pricing pressures. The drug's long-term pricing sustainability hinges on patent protection and market dynamics, with potential price reductions post-2030 due to generic competition.
Key Takeaways
- INTUNIV ER's current U.S. price averages around $370–$400/month, with international prices reflecting local healthcare policies.
- Market growth is steady, driven by demand for non-stimulant ADHD treatments.
- Price stability is expected until patent expiry around 2030, after which discounts may reach 30–50% with generic entry.
- Competition from stimulants and other non-stimulants influences market share stability.
- Regulatory and policy changes could accelerate price declines or alter competitive dynamics.
FAQs
Q1: When is the patent for INTUNIV ER set to expire?
A1: Approximately 2030.
Q2: How does INTUNIV ER’s price compare to stimulant medications?
A2: It is generally more expensive on a monthly basis, partly due to extended-release formulation costs.
Q3: Will market entry of generics significantly reduce INTUNIV ER's price?
A3: Yes, prices could decrease by 30–50% after patent expiry.
Q4: What factors could influence INTUNIV ER’s future market share?
A4: Competition, regulatory changes, clinical guidelines, and new therapy approvals.
Q5: Are international prices for INTUNIV ER likely to converge with U.S. prices?
A5: Not necessarily; prices are heavily influenced by local healthcare policies, reimbursement systems, and negotiations.
References
[1] IQVIA. (2023). Market intelligence report.
[2] EvaluatePharma. (2023). Global Prescription Drug Market, 2028.
[3] Symphony Health. (2023). Prescription Trends Data.
[4] World Health Organization. (2022). ADHD Epidemiology.
[5] Redbook. (2023). Wholesale Prices for Prescription Drugs.
[6] NHS Digital. (2023). Prescription Price Data.
[7] German Federal Ministry of Health. (2022). Drug Price List.
[8] Canadian Institute for Health Information. (2023). Drug Pricing Data.
[9] U.S. Patent Office. (2023). Patent Status for INTUNIV ER.
[10] IMS Health. (2022). Generic Trends in ADHD Medications.
More… ↓
