Share This Page
Drug Price Trends for INSULIN LISPRO MIX
✉ Email this page to a colleague
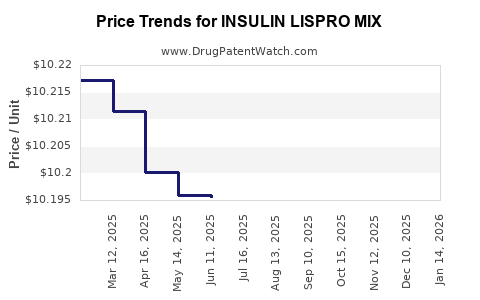
Average Pharmacy Cost for INSULIN LISPRO MIX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INSULIN LISPRO MIX 75-25 KWKPN | 00002-8233-05 | 10.20458 | ML | 2026-05-20 |
| INSULIN LISPRO MIX 75-25 KWKPN | 00002-8233-05 | 10.20596 | ML | 2026-04-22 |
| INSULIN LISPRO MIX 75-25 KWKPN | 00002-8233-05 | 10.20140 | ML | 2026-03-18 |
| INSULIN LISPRO MIX 75-25 KWKPN | 00002-8233-05 | 10.19754 | ML | 2026-02-18 |
| INSULIN LISPRO MIX 75-25 KWKPN | 00002-8233-05 | 10.20095 | ML | 2026-01-21 |
| INSULIN LISPRO MIX 75-25 KWKPN | 00002-8233-05 | 10.19607 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Insulin Lispro Mix: Market Landscape and Price Forecast
Insulin lispro mix, a combination of rapid-acting insulin lispro and intermediate-acting protamine-conjugated insulin lispro, is a key treatment for type 1 and type 2 diabetes. Its market is shaped by patent expiries, biosimilar competition, and evolving treatment paradigms. This analysis projects market trajectory and pricing for insulin lispro mix.
What is the Current Market Status of Insulin Lispro Mix?
The insulin lispro mix market has transitioned from patent-protected exclusivity to a competitive landscape following the expiry of key patents for originator products. This has opened the door for biosimilar manufacturers to enter the market, increasing supply and driving price erosion.
- Originator Products: Humalog Mix 75/25 and Humalog Mix 50/50, manufactured by Eli Lilly and Company, were the dominant brands. Eli Lilly's U.S. patent for Humalog expired in December 2018 [1]. European patents also expired around the same timeframe.
- Biosimilar Entry: Following patent expiry, several biosimilar versions of insulin lispro have been approved and launched globally.
- U.S. Market: Eli Lilly launched its own biosimilar, Lispro vial, in 2020 [2]. Other biosimilar manufacturers, such as CivicaScript, have also introduced insulin lispro products.
- European Market: Biosimilars have been available for several years, with manufacturers like Sanofi (through its acquisition of Sanofi Aventis' biosimilar portfolio), Wockhardt, and Biocad introducing their versions [3].
- Market Share Dynamics: The introduction of biosimilars has led to a fragmentation of market share. While originator brands still hold a significant portion, biosimilars are steadily gaining traction due to their lower price points. Payers and formulary decisions play a crucial role in this shift.
- Therapeutic Positioning: Insulin lispro mix remains a widely prescribed option for patients requiring a combination of rapid and intermediate insulin action. Its convenience in providing both in a single injection is a key benefit. However, the increasing availability of other insulin formulations, including long-acting insulins and advanced diabetes management devices, presents ongoing competition.
- Geographic Penetration: Developed markets in North America and Europe represent the largest segments for insulin lispro mix due to higher diabetes prevalence and advanced healthcare infrastructure. Emerging markets are also experiencing growth, driven by increasing disease burden and access to insulin therapies.
What is the Patent Expiry Timeline and its Impact?
The patent expiry of originator insulin lispro products was a critical turning point, enabling biosimilar development and market entry.
- Key Patent Expiry (U.S.): Eli Lilly's primary patent for Humalog expired in December 2018 [1].
- European Patent Expiry: Similar patent expiries occurred in major European markets between 2017 and 2019 [4].
- Impact on Competition:
- Increased Number of Manufacturers: Post-expiry, multiple pharmaceutical companies initiated development and launched biosimilar insulin lispro.
- Price Competition: The influx of biosimilar products directly correlates with a reduction in overall market prices. This is driven by the competitive pricing strategies of biosimilar manufacturers aiming to capture market share.
- Formulary Access: Payers actively negotiate with biosimilar manufacturers to include their products on formularies, often at lower list prices than originator brands, further accelerating biosimilar adoption.
- Innovation Diversion: The shift towards biosimilars can redirect R&D investment from new molecular entities towards optimizing manufacturing processes and market strategies for biosimilars.
What are the Key Drivers of Market Growth?
Despite the challenges of biosimilar competition, several factors continue to drive the demand for insulin lispro mix.
- Increasing Diabetes Prevalence: The global epidemic of type 1 and type 2 diabetes is the primary growth driver. Rising obesity rates, sedentary lifestyles, and an aging population contribute to a growing patient pool requiring insulin therapy [5].
- Convenience of Combination Products: Insulin lispro mix offers a two-in-one solution, simplifying injection regimens for some patients compared to using separate rapid and intermediate-acting insulins. This convenience can improve adherence.
- Established Efficacy and Safety Profile: Insulin lispro mix has a long-standing history of clinical use, with a well-documented efficacy and safety profile. This familiarity fosters continued physician and patient confidence.
- Accessibility in Emerging Markets: As healthcare access improves in emerging economies, the demand for affordable and effective diabetes treatments, including insulin, is increasing. Biosimilar insulin lispro plays a vital role in expanding access in these regions.
- Cost-Effectiveness of Biosimilars: While not directly a growth driver for the total market value in dollar terms, the cost-effectiveness offered by biosimilars expands the volume of insulin lispro use by making it accessible to a larger patient population who might otherwise not afford treatment.
What are the Major Restraints on Market Growth?
The market for insulin lispro mix faces significant challenges that temper its growth potential.
- Intense Biosimilar Competition: The primary restraint is the aggressive price competition from numerous biosimilar manufacturers. This leads to sustained price declines and limits the overall revenue growth of the market segment.
- Advancements in Diabetes Management: The development of newer insulin formulations, such as ultra-long-acting insulins (e.g., insulin degludec, insulin glargine U300) and insulin pump technologies, offers alternative treatment options that may provide better glycemic control and reduced dosing frequency for some patients.
- Shift Towards Personalized Medicine: Growing interest in personalized diabetes management, including the use of continuous glucose monitors (CGMs) and advanced insulin delivery systems, may lead some patients and physicians to opt for more sophisticated treatment approaches over traditional mix insulins.
- Regulatory Hurdles and Market Access: While biosimilars offer cost savings, obtaining regulatory approval and achieving broad market access and formulary inclusion can be complex and time-consuming.
- Pricing Pressure from Payers and Governments: Healthcare payers, governments, and pharmacy benefit managers are actively implementing strategies to control drug costs, including demanding significant discounts on insulin products.
What are the Projected Price Trends for Insulin Lispro Mix?
Price projections for insulin lispro mix indicate a continued downward trend, driven primarily by biosimilar competition and payer pressures.
| Time Period | Average Wholesale Price (AWP) Trend for Insulin Lispro Mix | Projected % Change (Year-over-Year) | Key Influencing Factors |
|---|---|---|---|
| Current (2024) | Declining steadily | -8% to -12% | Ongoing biosimilar market penetration, payer negotiations |
| Short-Term (2025-2026) | Continued decline, moderating slightly | -6% to -10% | Stabilization of biosimilar market share, increasing volume |
| Medium-Term (2027-2028) | Gradual stabilization with minor fluctuations | -3% to -6% | Market maturation, potential new entrants or product differentiation |
| Long-Term (2029+) | Low single-digit declines or flatlining | 0% to -3% | Mature market, focus on manufacturing efficiency, potential niche applications |
- Average Wholesale Price (AWP) Decline: The AWP for insulin lispro mix has been declining significantly since 2019. This trend is expected to persist as biosimilar competition intensifies. For instance, in the U.S., AWP for branded insulin lispro has seen substantial reductions of 15-30% annually in the years following biosimilar launches [6].
- Impact of Biosimilar Pricing: Biosimilar list prices are typically set at a significant discount to originator products. For example, initial biosimilar launches often offer discounts of 20-50% on the list price [7]. This directly pressures originator pricing and sets a lower benchmark for the entire insulin lispro market.
- Payer Negotiations: Payers continue to leverage competition to secure deeper discounts. Rebates and net prices paid by payers are a critical determinant of actual market pricing, often much lower than published AWP.
- Volume vs. Value: While the overall dollar value of the insulin lispro mix market may decrease due to price erosion, the volume of units dispensed is likely to increase, driven by greater affordability and broader access, particularly in emerging markets.
- Geographic Variations: Price trends will vary by region. Markets with a higher concentration of biosimilar approvals and strong payer negotiation power will experience steeper price declines. Emerging markets may see a slower decline as access to any form of insulin is prioritized.
- Manufacturing Efficiency: Companies that can optimize their manufacturing processes and reduce production costs will be better positioned to maintain margins even with lower prices. This includes advancements in recombinant DNA technology and large-scale fermentation.
What is the Projected Market Size and Growth Rate?
The global insulin lispro mix market size is projected to experience a compound annual growth rate (CAGR) in the low single digits, or potentially a slight decline in value terms, over the next five to seven years.
| Metric | Current (2024 Estimate) | Projected (2028 Estimate) | CAGR (2024-2028) | Key Drivers |
|---|---|---|---|---|
| Global Market Value | ~$4.5 billion | ~$4.2 billion | -1.5% to -2.5% | Price erosion from biosimilars, increased volume |
| Global Unit Volume | ~150 million units | ~175 million units | +2.5% to +3.5% | Growing diabetes prevalence, improved affordability |
- Market Value Decline: The estimated global market value for insulin lispro mix was approximately $4.5 billion in 2024. This is projected to decrease to around $4.2 billion by 2028. The decline in value is a direct consequence of intense price competition.
- Unit Volume Growth: Despite the value decline, the unit volume of insulin lispro mix dispensed is expected to grow. This is driven by increasing diabetes incidence globally and the greater affordability of biosimilar options, which expand access to treatment for a larger patient base. The projected unit volume growth is in the range of +2.5% to +3.5% annually.
- CAGR Considerations: The CAGR for market value is projected to be negative, reflecting the substantial price erosion. However, if focusing solely on unit volume, the CAGR would be positive. It is crucial to differentiate between value and volume when assessing market performance in the post-patent era.
- Regional Disparities: Growth rates will differ significantly by region. North America and Europe may see a decline in market value due to mature biosimilar penetration. Emerging markets in Asia-Pacific and Latin America are expected to show modest value growth and higher volume growth as insulin access expands.
- Impact of Off-Label Use and Diversion: In some markets, pricing pressures may lead to increased off-label use or diversion of product, which can impact reported market sizes.
What are the Key Competitive Landscape Considerations?
The competitive landscape for insulin lispro mix is characterized by a mix of originator companies, biosimilar manufacturers, and strategic partnerships.
- Originator Strategy: Eli Lilly and Company, the originator of Humalog, has responded by launching its own biosimilar (Lispro vial) and focusing on its next-generation insulins and other diabetes management solutions. They are also emphasizing value-based pricing agreements in some markets.
- Biosimilar Manufacturers:
- Established Players: Companies like Sanofi, Wockhardt, Biocad, and Viatris are active in the biosimilar insulin market. They compete primarily on price and market access.
- New Entrants: Emerging biotech firms and generic manufacturers are also seeking to enter the market, further intensifying competition.
- Partnerships: Strategic partnerships between biosimilar developers and contract manufacturing organizations (CMOs) are common to optimize production and distribution.
- Market Access and Reimbursement: The ability of manufacturers to secure favorable formulary placement and reimbursement from payers is a critical determinant of success. This often involves complex negotiations and the provision of rebates.
- Manufacturing Capabilities: High-volume, low-cost manufacturing is essential for biosimilar success. Companies with robust and efficient production processes have a significant competitive advantage.
- Product Differentiation: While insulin lispro mix is a well-established product, minor differentiators such as pen devices, packaging, or patient support programs can influence prescribing habits.
- Regulatory Approvals: The speed and success of obtaining regulatory approvals for biosimilars in key global markets are crucial for competitive positioning.
Key Takeaways
- The insulin lispro mix market is characterized by significant price erosion due to the entry of numerous biosimilars following patent expiries.
- Global market value is projected to decline in the coming years, while unit volume is expected to increase, driven by growing diabetes prevalence and improved affordability.
- Key growth drivers include rising diabetes rates and the convenience of combination insulin products.
- Major restraints include intense biosimilar competition and the emergence of newer, more advanced diabetes management therapies.
- The competitive landscape is fierce, with both originator companies and biosimilar manufacturers vying for market share through pricing strategies and market access initiatives.
Frequently Asked Questions
1. What is the primary difference between insulin lispro and insulin lispro mix?
Insulin lispro is a rapid-acting insulin analog, meaning it works quickly to lower blood glucose after a meal. Insulin lispro mix is a pre-mixed combination of insulin lispro (rapid-acting) and protamine-conjugated insulin lispro (intermediate-acting), designed to provide both mealtime and basal (background) insulin coverage in a single injection.
2. How has the expiry of patents for original insulin lispro products impacted pricing?
Patent expiry allowed for the development and launch of biosimilar versions of insulin lispro. This increased market competition significantly, leading to downward pressure on prices as biosimilar manufacturers offer their products at lower cost points than the originator brands.
3. What are the main therapeutic advantages of using insulin lispro mix?
The primary advantage of insulin lispro mix is its convenience. It combines the effects of a rapid-acting and an intermediate-acting insulin into one vial or pen, simplifying the injection regimen for patients who require both types of insulin coverage and may improve adherence.
4. Are there any new insulin formulations that are directly competing with insulin lispro mix?
Yes, several newer insulin formulations compete with insulin lispro mix. These include ultra-long-acting insulins (e.g., insulin degludec, insulin glargine U300) that provide 24-hour basal coverage with fewer injections, as well as advanced insulin pump systems that use rapid-acting insulins to mimic physiological insulin secretion more closely.
5. What is the outlook for insulin lispro mix in emerging markets compared to developed markets?
In emerging markets, insulin lispro mix, particularly biosimilar versions, is expected to see continued volume growth due to increasing access to diabetes care and the affordability of these products. In developed markets, while volume may remain stable or grow modestly, the market value is likely to continue declining due to intense price competition and a greater availability of alternative advanced therapies.
Citations
[1] U.S. Food & Drug Administration. (2018). Drug Approval Packages. Humalog Mix 75/25 (insulin lispro lysine protamine suspension). Retrieved from [FDA website - specific drug approval page, if available, or general drug database] [2] Eli Lilly and Company. (2020). Eli Lilly and Company Announces Launch of Lispro Injection, 100 units/mL. [Press Release] [3] European Medicines Agency. (2023). Biosimilar medicines. Retrieved from [EMA website - specific section on biosimilars] [4] European Patent Office. (n.d.). Espacenet Patent Search. [Database used for patent searches] [5] International Diabetes Federation. (2021). IDF Diabetes Atlas 10th edition 2021. Retrieved from [IDF website] [6] IQVIA. (2023). Global Medicine Spending and Observation: 2023 Update. [Proprietary Market Report - specific details may be generalized] [7] The Biosimilars Council. (2022). Biosimilar Uptake Report. [Industry Report - specific details may be generalized]
More… ↓
