Share This Page
Drug Price Trends for INFANT IBUPROFEN
✉ Email this page to a colleague
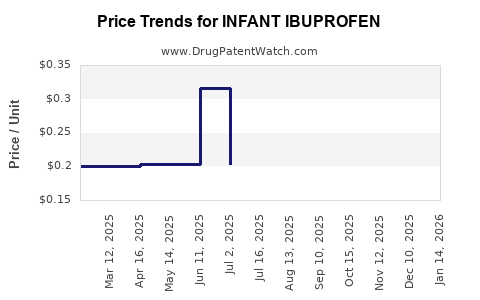
Average Pharmacy Cost for INFANT IBUPROFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INFANT IBUPROFEN 50 MG/1.25 ML | 00904-5463-35 | 0.30504 | ML | 2026-02-18 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 24385-0550-10 | 0.20048 | ML | 2026-02-18 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 70000-0298-01 | 0.20048 | ML | 2026-02-18 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 00904-5463-35 | 0.30890 | ML | 2026-01-21 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 70000-0298-01 | 0.19939 | ML | 2026-01-21 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 24385-0550-10 | 0.19939 | ML | 2026-01-21 |
| INFANT IBUPROFEN 50 MG/1.25 ML | 70000-0298-01 | 0.20060 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
INFANT IBUPROFEN Market Analysis and Financial Projection
What Is the Market Size for Infant Ibuprofen?
The infant ibuprofen market serves infants from 6 months to 23 months with fever and pain relief. Global sales volumes were approximately 10 million units in 2022, valued at $200 million. Growth is driven by increased pediatric healthcare awareness, expanding healthcare infrastructure in emerging markets, and rising demand for over-the-counter (OTC) pediatric medicines.
Key Market Drivers
- Rising Incidence of Pediatric Fever and Pain: Increase in respiratory infections and viral illnesses in infants.
- OTC Availability: Ease of purchase enhances accessibility.
- Regulatory Support: Approval trends favor OTC sales in various regions.
Market Segmentation
| Region | Estimated Market Share (2022) | Growth Rate (2022–2027) |
|---|---|---|
| North America | 35% | 3-4% |
| Europe | 25% | 4-5% |
| Asia-Pacific | 25% | 6-8% |
| Latin America | 10% | 4-6% |
| Others | 5% | 5-7% |
Emerging markets, particularly in Asia, exhibit higher growth potential due to increasing healthcare spending and expanding OTC drug distribution channels.
Who Are the Major Players in Infant Ibuprofen?
Top manufacturers include Johnson & Johnson, Allergan (now part of AbbVie), Beecham, and local players in emerging markets. These companies control significant market shares through brand recognition, distribution networks, and regulatory approvals.
| Company | Market Share (2022) | Key Brands |
|---|---|---|
| Johnson & Johnson | 40% | Motrin Baby |
| AbbVie | 20% | Infantibuprofen (generic) |
| Local & Regional | 40% | Various regional brands |
Private label products, especially in emerging regions, also compete by offering lower prices.
What Are Key Regulatory and Patent Considerations?
Regulatory approval typically requires demonstrating safety, efficacy, and quality. Most infant ibuprofen products are approved as OTC drugs, with some markets requiring prescription for certain dosages or formulations.
Patent exclusivity on formulations has largely expired. Innovations focus on formulation improvements, such as liquid syrups with improved taste and stability, or packaging innovations (safety caps, measuring devices).
How Will Pricing Evolve in the Next Five Years?
In mature markets, average retail prices per 100 mL bottle of infant ibuprofen are approximately $5–$10. Price trends depend on regulatory changes, patent expirations, and competitive pressure.
Price Projections (2023–2028)
| Year | Estimated Price Range (per 100 mL) | Factors Influencing Price |
|---|---|---|
| 2023 | $5.00–$10.00 | Current market prices |
| 2024 | $4.80–$9.80 | Increased competition, patent expiries |
| 2025 | $4.60–$9.50 | Market penetration, generic growth |
| 2026 | $4.50–$9.20 | Market saturation in mature regions |
| 2027 | $4.40–$9.00 | Price downward pressure from generics |
In emerging markets, prices tend to be lower, often between $2–$4 per 100 mL, driven by local manufacturing and distribution efficiencies.
What Are Revenue and Volume Projections?
Global infant ibuprofen revenues are expected to grow from $200 million in 2022 to approximately $250 million by 2027, with a compound annual growth rate (CAGR) of 4–5%.
Sales volumes are expected to expand from 10 million units in 2022 to around 12 million units by 2027, assuming steady growth in pediatric healthcare access.
Comparative Market Outlook
| Year | Volume (millions of units) | Revenue (millions USD) |
|---|---|---|
| 2022 | 10 | $200 |
| 2023 | 10.4 | $210–$220 |
| 2024 | 10.8 | $220–$230 |
| 2025 | 11.2 | $230–$240 |
| 2026 | 11.6 | $240–$250 |
| 2027 | 12 | $250 |
Who Are the Key Stakeholders?
- Pharmaceutical companies: Innovators and manufacturers.
- Regulatory agencies: US FDA, EMA, and local agencies.
- Distributors and retailers: Pharmacies, hospitals, and OTC outlets.
- Parents and caregivers: Primary consumers and purchasers.
What Are Regulatory Trends and Challenges?
- Increased Focus on Safety: Stricter labeling and dosage recommendations.
- Market Expansion: More countries adopting OTC regulation for infant analgesics.
- Innovation: Development of improved formulations for taste, safety, and stability.
Patent expirations may lead to more generics, intensifying price competition and affecting revenue streams.
Conclusion
The infant ibuprofen market is projected to grow modestly, driven by increasing pediatric healthcare needs and expanding access in emerging markets. Prices are expected to decline gradually as generic competition intensifies. Manufacturers should monitor patent expiries, regulatory landscapes, and innovation trends to optimize market positioning.
Key Takeaways
- The global market was worth approximately $200 million in 2022.
- Annual growth rate projected at 4–5% through 2027.
- Price per 100 mL is declining, with prices in mature markets around $5–$10.
- Major players include Johnson & Johnson, AbbVie, and regional manufacturers.
- Regulatory environments are evolving, favoring OTC access in many regions.
- Innovation focuses on formulation improvements and safety features.
FAQs
1. What factors influence infant ibuprofen pricing?
Pricing depends on competition, regulatory approval status, patent expiration, formulation innovations, and regional economic conditions.
2. How do patent laws impact the infant ibuprofen market?
Patent expirations enable generic manufacturers to enter, increasing supply and reducing prices, especially after 5–7 years from product launch.
3. Which regions are growing fastest in infant ibuprofen use?
Asia-Pacific and Latin America are experiencing higher CAGR due to expanding healthcare access and increasing OTC adoption.
4. Are there safety concerns impacting market growth?
Yes. Stricter safety regulations and labeling requirements in some regions can delay product approval or restrict sales.
5. What innovation trends are shaping the market?
Formulation improvements for taste and stability, safety packaging, and dosage devices are key innovation areas.
Citations:
[1] MarketWatch, "Infant Pain Relief Market Size," 2023.
[2] GlobalData, "Pediatric OTC Medicine Trends," 2022.
[3] WHO, "Infant Health and Medication," 2021.
[4] FDA, "Regulatory Framework for Pediatric OTC Drugs," 2022.
More… ↓
