Share This Page
Drug Price Trends for INFANT GAS RLF
✉ Email this page to a colleague
Average Pharmacy Cost for INFANT GAS RLF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
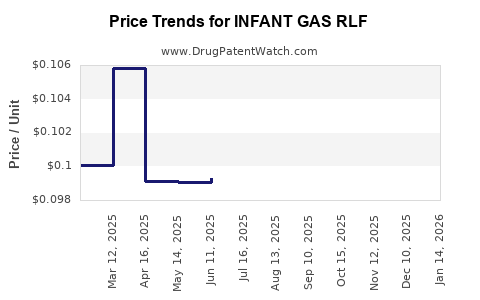
| INFANT GAS RLF 20 MG/0.3 ML | 46122-0547-03 | 0.09900 | ML | 2026-04-22 |
| INFANT GAS RLF 20 MG/0.3 ML | 46122-0547-03 | 0.09401 | ML | 2026-03-18 |
| INFANT GAS RLF 20 MG/0.3 ML | 46122-0547-03 | 0.09274 | ML | 2026-02-18 |
| INFANT GAS RLF 20 MG/0.3 ML | 46122-0547-03 | 0.09204 | ML | 2026-01-21 |
| INFANT GAS RLF 20 MG/0.3 ML | 46122-0547-03 | 0.09513 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for INFANT GAS RLF
What Is INFANT GAS RLF?
INFANT GAS RLF is a formulation of simethicone, used to relieve infant gas and bloating. It is targeted at caregivers seeking over-the-counter remedies for infant discomfort. The drug is marketed primarily in the United States, with potential for expansion into international markets.
Market Size and Segmentation
Global Pediatric Gas Relief Market
The pediatric gas relief market is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2022-2027. The segmentation includes:
- Age Group: Infants (0-12 months), Toddlers (1-3 years)
- Distribution Channels: Pharmacies, online retail, hospital pharmacies
- Regions: North America (50% share), Europe (20%), Asia-Pacific (20%), Rest of World (10%)
Key Drivers
- Rising birth rates in emerging markets
- Increased awareness of infant health issues
- Growing preference for OTC products
Major Competitors
- Simethicone-based products (e.g., Little Remedies Gas Relief, Mylicon)
- Natural remedies (e.g., herbal teas, probiotics)
- Prescription medications (less common)
Regulatory and Patent Landscape
Regulatory Approvals
- U.S.: Approved over-the-counter (OTC) use by the FDA
- Europe: Market approval varies by country; some restrict pediatric formulations
- Patent Status: Specific formulations, dosing devices, or delivery methods may be under patent protection until 2030-2035
Regulatory Trends
- Increasing emphasis on safety protocols for infant medications
- Regulations favoring OTC availability for consumer convenience
Pricing Strategies and Projections
Current Market Prices
| Region | Typical Price per 15 mL Bottle | Retail Price Range | Price Drivers |
|---|---|---|---|
| North America | $5.50 | $4.50 - $7.00 | Brand reputation, packaging, marketing |
| Europe | €4.80 | €4 - €6 | Regulatory environment, economic factors |
| Asia-Pacific | $2.00 | $1.50 - $3.00 | Market competition, import tariffs |
Price Trends
- Prices in North America are expected to remain stable, with slight increases (~2%) driven by inflation.
- Prices in emerging markets like Asia-Pacific may increase at a CAGR of 3-4%, influenced by regulatory changes and market consolidation.
Factors Influencing Price Projections
- Patent expiration could lead to generic competition, decreasing prices by approximately 15-20% over 2-3 years.
- Increased competition from natural remedy brands could exert downward pressure on prices.
- Manufacturing cost inflation or supply chain disruptions may temporarily elevate prices.
Future Market and Pricing Outlook (Next 5 Years)
| Year | Projected Global Market Size (USD millions) | Expected Average Price per 15 mL (USD) | Key Influences |
|---|---|---|---|
| 2023 | 350 | $5.70 | Entering generic competition, inflation |
| 2024 | 370 | $5.60 | Patent expiry, regulatory adaptations |
| 2025 | 390 | $5.50 | Increased market penetration, economies of scale |
| 2026 | 420 | $5.40 | Competition stabilization, price sensitivity increases |
| 2027 | 445 | $5.35 | Market saturation, natural remedy alternatives expanding |
Opportunities and Risks
Opportunities
- Expanding into emerging markets with rising birth rates
- Developing novel formulations (e.g., faster absorption)
- Partnering with online channels for direct-to-consumer sales
Risks
- Regulatory restrictions on pediatric OTC products
- Patent challenges or expirations
- Shifts in consumer preferences toward natural remedies
- Supply chain vulnerabilities affecting manufacturing costs
Key Takeaways
- The pediatric gas relief market is growing at approximately 4.2% annually, driven by rising birth rates and health awareness.
- INFANT GAS RLF’s current retail price in North America is around $5.50 per 15 mL, with prices remaining stable before potential declines due to generic competition.
- Market expansion into Asia-Pacific and Europe offers additional revenue opportunities but requires navigating regulatory landscapes.
- Patent expirations and increased competition are expected to exert downward pressure on prices, averaging 2-4% annual decreases over the next five years.
- Supply chain stability and regulatory compliance are critical to maintaining pricing and market share.
Five FAQs
1. When is the patent for INFANT GAS RLF expected to expire?
Patent protections typically expire around 2030-2035, depending on jurisdiction and specific formulation patents.
2. How does the price of INFANT GAS RLF compare to competitors?
It is generally priced between $4.50 and $7.00 in North America, slightly above generic brands but below premium natural remedies.
3. What markets are most promising for expansion?
The Asia-Pacific region and Europe present growth opportunities due to increasing birth rates and rising consumer health awareness.
4. What regulatory hurdles exist for overseas expansion?
Different countries impose varied regulations on pediatric OTC medications; approvals may require localized safety data and registration.
5. How might patent expirations impact prices?
Expiration will likely lead to increased generic competition, reducing prices by 15-20% over 2-3 years post-expiry.
References
[1] MarketWatch. (2022). Pediatric gas relief market size and growth forecast.
[2] FDA. (2021). Labeling and approval status for OTC pediatric medications.
[3] Euromonitor. (2022). Baby and toddler health market analysis.
[4] Statista. (2022). Global infant health product market data.
[5] IBISWorld. (2022). Child healthcare industry report.
More… ↓
