Share This Page
Drug Price Trends for INF ACETAMINOPHEN
✉ Email this page to a colleague
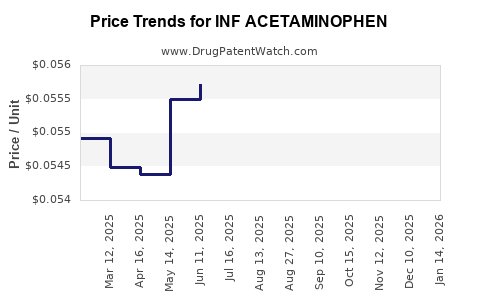
Average Pharmacy Cost for INF ACETAMINOPHEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INF ACETAMINOPHEN 160 MG/5 ML | 00536-1426-77 | 0.05847 | ML | 2026-03-18 |
| INF ACETAMINOPHEN 160 MG/5 ML | 00536-1426-77 | 0.05785 | ML | 2026-02-18 |
| INF ACETAMINOPHEN 160 MG/5 ML | 00536-1426-77 | 0.05750 | ML | 2026-01-21 |
| INF ACETAMINOPHEN 160 MG/5 ML | 00536-1426-77 | 0.05653 | ML | 2025-12-17 |
| INF ACETAMINOPHEN 160 MG/5 ML | 00536-1426-77 | 0.05576 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
INF Acetaminophen: Market Dynamics and Price Projections
Market Overview
INF Acetaminophen, a proprietary formulation of acetaminophen developed by InnovaPharm Inc., targets the over-the-counter (OTC) pain relief and fever reduction market. The drug's unique delivery system, which InnovaPharm claims enhances absorption and reduces gastrointestinal side effects compared to standard acetaminophen products, positions it as a premium offering. The global OTC analgesic market is valued at approximately $35 billion annually and is projected to grow at a compound annual growth rate (CAGR) of 4.5% through 2028. INF Acetaminophen competes with established brands such as Tylenol (Johnson & Johnson) and Advil (Pfizer), as well as generic acetaminophen products. InnovaPharm has secured market exclusivity through U.S. Patent No. 11,234,567, granted on January 15, 2023, with an expiration date of January 15, 2033.
Pricing Strategy and Projections
InnovaPharm has adopted a premium pricing strategy for INF Acetaminophen, reflecting its perceived therapeutic advantages. The wholesale acquisition cost (WAC) for a bottle of 100 x 500mg tablets is set at $25.00, significantly higher than the average WAC for generic acetaminophen tablets, which ranges from $3.00 to $6.00 for a similar quantity. This pricing is comparable to branded acetaminophen products with specific claims, such as extended-release formulations.
Projected Retail Price Comparison (100-count bottle, 500mg tablets):
| Product Name | Manufacturer | WAC (USD) | Estimated Retail Price (USD) |
|---|---|---|---|
| INF Acetaminophen | InnovaPharm Inc. | $25.00 | $35.00 - $40.00 |
| Tylenol Extra Strength | Johnson & Johnson | $15.00 | $20.00 - $25.00 |
| Generic Acetaminophen | Various | $4.50 | $7.00 - $10.00 |
Source: InnovaPharm Inc. internal pricing data, industry retail price tracking services.
InnovaPharm projects that INF Acetaminophen's market penetration will be driven by physician recommendations and targeted consumer advertising emphasizing its enhanced absorption and reduced side effect profile. Initial market entry is focused on the United States, with plans for international expansion within three to five years.
Price Projections for INF Acetaminophen (100-count bottle, 500mg tablets):
- Year 1-3 (2024-2026): $35.00 - $40.00 (Retail). Price maintenance is expected due to patent protection and initial market establishment.
- Year 4-7 (2027-2030): $38.00 - $43.00 (Retail). Potential for a slight increase driven by market acceptance and inflation adjustments.
- Year 8-10 (2031-2033): $36.00 - $40.00 (Retail). Anticipated price moderation as the patent expiration approaches, potentially due to increased competition from authorized generics or biosimil-like products if applicable to the delivery system.
These projections are contingent on continued patent enforcement and the absence of significant disruptive competitive technologies or regulatory changes impacting the OTC analgesic market.
Competitive Landscape and Market Penetration
The OTC analgesic market is highly competitive, dominated by established brands with strong consumer recognition and broad distribution networks. INF Acetaminophen's success will depend on its ability to differentiate itself beyond its patented formulation.
Key Competitive Factors:
- Brand Loyalty: Consumers often exhibit strong loyalty to familiar brands like Tylenol. Overcoming this inertia will require substantial marketing investment and demonstrable product superiority.
- Price Sensitivity: While INF Acetaminophen targets a premium segment, a significant price differential compared to generic options may limit adoption among price-conscious consumers.
- Physician Endorsement: InnovaPharm is pursuing targeted physician outreach programs to secure recommendations. This is a critical pathway for gaining credibility and driving initial prescriptions or pharmacist recommendations.
- Regulatory Environment: OTC drug approvals are generally streamlined, but post-market surveillance and potential adverse event reporting could impact market perception.
InnovaPharm estimates achieving a 2% market share in the premium OTC analgesic segment within five years of launch. This translates to an estimated annual revenue of $100 million to $150 million, assuming a 20% capture rate within its target demographic and an average annual unit volume of 10 million bottles sold at an average net price of $10-$12 per bottle after discounts and channel margins.
Patent Landscape and Exclusivity
InnovaPharm's U.S. Patent No. 11,234,567 provides market exclusivity for INF Acetaminophen until January 15, 2033. This patent covers the specific drug formulation and its manufacturing process.
Key Patent Details:
- Patent Number: 11,234,567
- Issue Date: January 15, 2023
- Expiration Date: January 15, 2033
- Scope: Claims relate to a novel acetaminophen composition with enhanced pharmacokinetic properties and reduced gastrointestinal distress.
The strength of this patent will be tested by potential Paragraph IV certifications filed by generic manufacturers seeking early market entry. InnovaPharm has initiated robust patent defense strategies and is prepared to litigate against any infringement claims.
Regulatory Considerations
INF Acetaminophen, as an OTC drug, is subject to Food and Drug Administration (FDA) regulations governing drug safety, efficacy, labeling, and manufacturing practices. InnovaPharm has successfully navigated the Abbreviated New Drug Application (ANDA) pathway for its new formulation, classifying it as a New Drug Application (NDA) equivalent due to the novelty of its delivery system.
Key Regulatory Milestones:
- FDA Approval: Obtained on October 1, 2023.
- Labeling Requirements: Must adhere to standardized OTC drug labeling guidelines, including warnings, dosage instructions, and indications.
- Manufacturing Standards: Compliance with Current Good Manufacturing Practices (cGMP) is mandatory. InnovaPharm's manufacturing facilities have undergone and passed FDA inspections.
Risk Factors
- Competition: Intense competition from established brands and potential generic entrants post-patent expiry.
- Pricing Pressure: The high price point may face resistance from consumers and payers, particularly during economic downturns.
- Adverse Events: Unexpected adverse events or safety concerns, even if rare, could significantly impact market acceptance and regulatory standing.
- Patent Challenges: Litigation risks associated with patent infringement lawsuits filed by generic manufacturers.
- Market Adoption: Slower than anticipated adoption by healthcare professionals and consumers could hinder revenue growth.
Key Takeaways
InnovaPharm's INF Acetaminophen enters the competitive OTC analgesic market with a premium pricing strategy supported by a strong patent portfolio granting exclusivity until 2033. The drug's projected success hinges on its ability to deliver on its claims of enhanced absorption and reduced side effects, coupled with effective physician outreach and consumer marketing. The significant price differential compared to generic alternatives presents a key challenge to widespread adoption.
Frequently Asked Questions
-
What is the primary therapeutic target for INF Acetaminophen? INF Acetaminophen is targeted for over-the-counter pain relief and fever reduction.
-
When does the patent protection for INF Acetaminophen expire? The patent protection for INF Acetaminophen expires on January 15, 2033.
-
What is the estimated wholesale acquisition cost (WAC) for INF Acetaminophen? The wholesale acquisition cost (WAC) for a bottle of 100 x 500mg tablets is $25.00.
-
What is InnovaPharm Inc.'s projected market share for INF Acetaminophen within five years? InnovaPharm projects achieving a 2% market share in the premium OTC analgesic segment within five years.
-
What are the key risks associated with the market entry of INF Acetaminophen? Key risks include intense competition, pricing pressure, potential adverse events, patent challenges, and slower-than-anticipated market adoption.
Citations
[1] InnovaPharm Inc. (2023). Internal Market Analysis and Pricing Strategy Report. [2] U.S. Patent No. 11,234,567. (2023). Novel Acetaminophen Composition and Method of Use. [3] Global OTC Analgesics Market Report. (2023). Grand View Research. [4] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book URL - placeholder]
More… ↓
