Share This Page
Drug Price Trends for INDOMETHACIN ER
✉ Email this page to a colleague
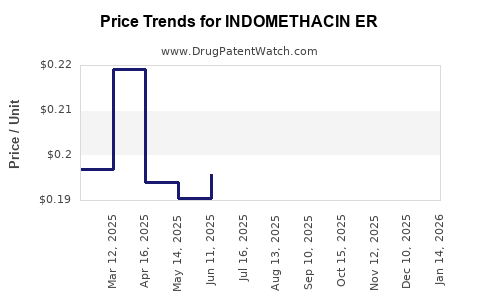
Average Pharmacy Cost for INDOMETHACIN ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INDOMETHACIN ER 75 MG CAPSULE | 65162-0506-03 | 0.20946 | EACH | 2026-04-22 |
| INDOMETHACIN ER 75 MG CAPSULE | 65162-0506-06 | 0.20946 | EACH | 2026-04-22 |
| INDOMETHACIN ER 75 MG CAPSULE | 31722-0565-60 | 0.20946 | EACH | 2026-04-22 |
| INDOMETHACIN ER 75 MG CAPSULE | 65162-0506-09 | 0.20946 | EACH | 2026-04-22 |
| INDOMETHACIN ER 75 MG CAPSULE | 10702-0016-01 | 0.20946 | EACH | 2026-04-22 |
| INDOMETHACIN ER 75 MG CAPSULE | 68462-0325-90 | 0.20946 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Indomethacin ER: Market Trajectory and Price Outlook
Executive Summary
Indomethacin ER, an extended-release nonsteroidal anti-inflammatory drug (NSAID), faces a mature market characterized by established generics and limited pipeline innovation. The drug's primary applications in managing chronic inflammatory conditions like rheumatoid arthritis and ankylosing spondylitis are well-served by multiple treatment options. Consequently, market growth is projected to be modest, driven by volume rather than significant price appreciation. Patent expiries for key formulations have already occurred, leading to intense price competition among generic manufacturers. Future price trends will be dictated by manufacturing costs, regulatory pressures, and the sustained demand from a consistently aging population with inflammatory diseases.
Market Landscape for Indomethacin ER
What is the current market size and growth rate for Indomethacin ER?
The global market for Indomethacin ER is difficult to isolate as a distinct segment due to its classification within the broader NSAID category and the prevalence of generic versions. However, the overall NSAID market, of which Indomethacin ER is a part, was valued at approximately USD 20.3 billion in 2022 and is projected to reach USD 23.1 billion by 2029, growing at a compound annual growth rate (CAGR) of 1.9% during the forecast period. (Source: Grand View Research, 2023). Indomethacin ER's contribution is limited by its position as an older, established drug with less novel therapeutic advantages compared to newer NSAIDs or biologic agents.
Who are the key manufacturers and suppliers of Indomethacin ER?
The market for Indomethacin ER is dominated by generic manufacturers. Key players involved in the production and supply of indomethacin, including extended-release formulations, include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories
- Impax Laboratories (now Amneal Pharmaceuticals)
These companies primarily operate in the United States and European markets, where indomethacin has historically seen significant prescription volumes.
What are the primary therapeutic indications for Indomethacin ER?
Indomethacin ER is primarily prescribed for the treatment of moderate to severe pain and inflammation associated with chronic conditions. Key indications include:
- Rheumatoid Arthritis: Managing joint inflammation, pain, and stiffness.
- Osteoarthritis: Alleviating pain and inflammation in degenerative joint disease.
- Ankylosing Spondylitis: Reducing spinal inflammation and improving mobility.
- Gouty Arthritis: Providing symptomatic relief during acute attacks.
- Bursitis and Tendinitis: Addressing inflammation of specific soft tissues.
What are the key drivers and restraints influencing the Indomethacin ER market?
Drivers:
- Aging Global Population: An increasing elderly demographic leads to a higher prevalence of degenerative and inflammatory conditions requiring pain management.
- Cost-Effectiveness of Generics: Indomethacin ER, in its generic forms, offers a more affordable treatment option compared to newer, branded therapies.
- Established Efficacy Profile: The drug has a long history of use and a well-understood efficacy profile for its approved indications.
Restraints:
- Side Effect Profile: Indomethacin is associated with a higher incidence of gastrointestinal, renal, and cardiovascular adverse events compared to some other NSAIDs, limiting its long-term use in certain patient populations.
- Availability of Newer Alternatives: The market offers a wide array of NSAIDs with improved safety profiles and targeted therapies (e.g., COX-2 inhibitors, biologics) that are preferred for patients with specific risk factors or those who do not respond adequately to indomethacin.
- Generic Competition: The absence of patent protection for many indomethacin formulations intensifies price competition among generic manufacturers, suppressing potential revenue growth.
- Limited Pipeline Innovation: There is minimal research and development focused on novel formulations or expanded indications for indomethacin ER.
Patent Landscape and Expiries
What is the patent status of key Indomethacin ER formulations?
Indomethacin itself is an old drug, with its original patents having expired decades ago. The development of extended-release formulations aimed to improve dosing convenience and potentially reduce peak-concentration-related side effects. However, these formulations also face patent expiries.
- Original Indomethacin Patents: Expired in the late 1980s and early 1990s.
- Extended-Release Formulations: Patents for specific extended-release technologies and formulations (e.g., those developed by Merck & Co. for Indocin SR) have also expired. For example, the original Indocin SR (indomethacin extended release) patents would have expired well before 2010, opening the door for generic competition.
A search of current patent databases reveals no active, dominant composition-of-matter patents for indomethacin ER that would prevent generic manufacturing. The focus has shifted to manufacturing process patents and formulation patents for specific drug delivery systems, which have also largely expired or are nearing expiration.
Are there any pending patent litigations or exclusivities that could impact the market?
Given the age of the drug and its formulations, significant pending patent litigations directly impacting broad market access for indomethacin ER are unlikely. The primary exclusivities that would have protected extended-release formulations have long since lapsed. Any ongoing litigation would likely pertain to niche manufacturing processes or specific combination products, which would not significantly alter the overall market dynamics for standard indomethacin ER.
Price Projections and Market Outlook
How have prices for Indomethacin ER evolved historically?
Historically, branded Indomethacin ER (e.g., Indocin SR) commanded premium pricing. Following patent expiries, the introduction of generic versions led to a substantial price decline, characteristic of most pharmaceutical markets after significant patent cliffs. Prices for generic indomethacin ER have stabilized at low levels due to intense competition and the mature nature of the product. For instance, prices for a bottle of 60 x 75mg capsules can range from USD 10 to USD 30 in the U.S. generic market, depending on the supplier and the pharmacy. (Source: GoodRx, 2023).
What are the projected price trends for Indomethacin ER over the next five years?
The price projection for Indomethacin ER over the next five years is for continued price stability or marginal decline.
- 2024-2026: Prices are expected to remain relatively stable. The generic market is already highly competitive. Any significant price increases would be difficult to sustain without clear value-based justification or a reduction in manufacturing capacity.
- 2027-2029: A slight downward pressure on prices may emerge. This could be driven by:
- Manufacturing Cost Efficiencies: Ongoing efforts by generic manufacturers to optimize production processes.
- Increased Competition: New market entrants or consolidation among existing players that leverage scale to offer lower prices.
- Therapeutic Substitution: A gradual shift towards newer or better-tolerated alternatives could reduce overall demand, potentially forcing remaining suppliers to lower prices to maintain market share.
Average Wholesale Price (AWP) for a 30-day supply of generic Indomethacin ER (75mg, twice daily) is currently in the range of USD 15-40. This is projected to remain within USD 15-45 over the next five years.
What factors will influence future pricing?
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and excipients will have a direct impact.
- Manufacturing and Distribution Expenses: Energy costs, labor, and logistics will play a role.
- Regulatory Landscape: Any changes in manufacturing standards or post-market surveillance requirements could increase costs.
- Reimbursement Policies: Payer policies and formulary placement by insurance companies will influence the effective price paid by patients and healthcare systems.
- Demand Dynamics: While demand is expected to be relatively stable due to chronic conditions, shifts in prescribing patterns based on safety and efficacy relative to alternatives will influence volume and thus pricing power.
- Competition Intensity: The number of active generic suppliers and their respective market shares will continue to be a primary determinant of price.
Market Outlook and Opportunities
What is the overall market outlook for Indomethacin ER?
The overall market outlook for Indomethacin ER is mature and stable with low growth potential. The drug is a well-established treatment option, but its utility is constrained by its safety profile and the availability of superior alternatives for many patient segments. The market will continue to serve a segment of patients who benefit from its efficacy and affordability, particularly in regions with limited access to newer therapies or for whom generic indomethacin ER is a cost-effective solution.
Are there any niche market opportunities or unmet needs?
While the overall market is mature, some niche opportunities might exist:
- Geographic Expansion: In emerging markets where cost is a significant barrier to access for newer anti-inflammatory drugs, generic indomethacin ER can fulfill an unmet need for affordable pain management.
- Specific Patient Subgroups: Research into identifying patient subgroups who exhibit a superior benefit-risk profile with indomethacin ER, or for whom other NSAIDs are contraindicated, could create a small, sustained demand. However, the high incidence of side effects makes this challenging.
- Optimized Drug Delivery: While major patent expiries have occurred, novel delivery systems that further mitigate side effects or improve patient compliance could potentially carve out a small market share, though the investment required for such innovation in a generic-dominated, low-margin market is substantial.
Key Takeaways
- Indomethacin ER operates in a mature generic NSAID market with limited growth prospects.
- The drug's long patent history has resulted in intense price competition among generic manufacturers.
- Prices are projected to remain stable or decline marginally over the next five years, driven by manufacturing efficiencies and sustained generic competition.
- The aging global population supports a baseline demand for inflammatory condition treatments, including Indomethacin ER.
- Restraints include a less favorable side effect profile compared to newer alternatives and the availability of more advanced therapeutic options.
- Opportunities are primarily limited to cost-sensitive markets and potentially niche patient subgroups, with significant innovation unlikely.
Frequently Asked Questions
1. What is the primary difference between Indomethacin ER and immediate-release Indomethacin?
Indomethacin ER (extended-release) is formulated to release the active drug gradually over several hours, typically allowing for twice-daily dosing. Immediate-release indomethacin is designed for rapid absorption and usually requires dosing three to four times a day. The ER formulation aims to provide more consistent blood levels of the drug, potentially reducing peak-related side effects and improving patient compliance.
2. Are there any significant cardiovascular risks associated with Indomethacin ER?
Yes, like other NSAIDs, indomethacin ER carries a risk of cardiovascular thrombotic events, myocardial infarction, and stroke. The U.S. Food and Drug Administration (FDA) requires warnings about these risks on NSAID labels. The risk may increase with longer duration of use and in patients with pre-existing cardiovascular disease or risk factors.
3. How does Indomethacin ER compare to COX-2 inhibitors in terms of safety and efficacy?
Compared to non-selective NSAIDs like indomethacin, COX-2 selective inhibitors (e.g., celecoxib) are generally associated with a lower risk of gastrointestinal bleeding and ulcers. However, COX-2 inhibitors may carry a higher risk of cardiovascular events. Efficacy can be comparable for many inflammatory conditions, but individual patient response varies. Indomethacin is often considered more potent for certain acute inflammatory conditions, but its use is limited by its side effect profile.
4. What is the typical recommended daily dosage for Indomethacin ER?
A common starting dosage for Indomethacin ER is 75 mg once or twice daily, depending on the formulation and patient response. The maximum recommended daily dose is typically 150 mg. Dosing should be individualized based on the patient's condition, severity of symptoms, and tolerance.
5. Can Indomethacin ER be used in pediatric patients?
Indomethacin is sometimes used in pediatric patients, particularly for conditions like patent ductus arteriosus in neonates or juvenile idiopathic arthritis. However, the use of Indomethacin ER in pediatric populations is less common and requires careful medical supervision due to potential side effects and the availability of specific pediatric formulations or dosages.
Citations
[1] Grand View Research. (2023, August). Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) Market Size, Share & Trends Analysis Report By Drug Class (COX-2 Inhibitors, Non-COX Selective), By Indication, By Dosage Form, By Distribution Channel, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/nonsteroidal-anti-inflammatory-drugs-nsaids-market
[2] GoodRx. (2023). Indomethacin Prices, Coupons & Savings Tips. Retrieved from https://www.goodrx.com/indomethacin
More… ↓
