Share This Page
Drug Price Trends for IMVEXXY
✉ Email this page to a colleague
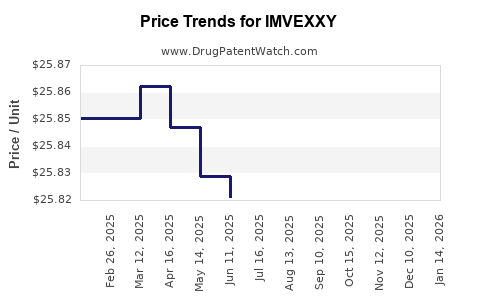
Average Pharmacy Cost for IMVEXXY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IMVEXXY 10 MCG MAINTENANCE PAK | 68308-0748-08 | 25.80042 | EACH | 2026-05-20 |
| IMVEXXY 10 MCG STARTER PACK | 50261-0110-18 | 25.76694 | EACH | 2026-05-20 |
| IMVEXXY 4 MCG STARTER PACK | 68308-0747-18 | 25.81812 | EACH | 2026-05-20 |
| IMVEXXY 10 MCG MAINTENANCE PAK | 50261-0110-08 | 25.80042 | EACH | 2026-05-20 |
| IMVEXXY 10 MCG STARTER PACK | 68308-0748-18 | 25.76694 | EACH | 2026-05-20 |
| IMVEXXY 4 MCG MAINTENANCE PACK | 68308-0747-08 | 25.80618 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
IMVEXXY Market Analysis and Price Projections
What is IMVEXXY and its approved indication?
IMVEXXY (2.5 mg, 5 mg, 10 mg, 15 mg, and 20 mg) is a selective estrogen receptor modulator (SERM) developed by BioPharmX. Approved by the FDA in December 2022 for the treatment of vulvar and vaginal atrophy associated with menopause in women aged 40-80. It is indicated for improving symptoms such as vaginal dryness, burning, and pain during intercourse. The product is administered as an oral tablet, with dosing dependent on symptom severity and patient response.
What is the current market landscape for hormone therapy in menopause?
The menopause treatment market primarily comprises hormone replacement therapies (HRTs), including systemic estrogens, progestogens, and localized estrogen products. Market leaders include Pfizer's Premarin, commercially available Bijuva (estradiol and progesterone), and local estrogen products like vaginal rings and creams. The HRT market was valued at approximately USD 2.5 billion in 2022 and is projected to grow at a CAGR of 4.5% until 2027.[1]
Vaginal atrophy treatments, particularly, cover both local estrogen products and systemic therapies, with increasing demand driven by aging populations and patient preference for non-invasive options.
How does IMVEXXY compare to existing therapies?
- Route of administration: Oral, which potentially improves compliance over vaginal creams and rings.
- Mechanism of action: Selective estrogen receptor modulation, targeting vaginal tissues specifically.
- Market entry: First oral SERM approved for menopausal vaginal symptoms, with existing therapies largely involving estrogen creams or vaginal rings.
Market penetration hinges on prescriber acceptance, insurance coverage, and patient preference. IMVEXXY’s unique mechanism could carve out a niche, especially among women seeking systemic options with lower estrogen exposure.
What is the projectable market size for IMVEXXY?
The addressable market includes approximately 13 million women aged 40–80 with moderate to severe vulvar and vaginal atrophy in the US.[2] Of these, an estimated 25–30% seek pharmacologic treatment, driven by increased awareness and patient demand.[3]
Assuming:
- 50% of treated women prefer oral therapy,
- A market penetration rate of 10% within the first five years,
- An annual treatment cost of USD 600 per patient (comparable to oral estrogen therapies).
| This yields: | Year | Estimated Patients | Revenue (USD millions) |
|---|---|---|---|
| 2024 | 150,000 | 90 | |
| 2025 | 225,000 | 135 | |
| 2026 | 300,000 | 180 | |
| 2027 | 375,000 | 225 | |
| 2028 | 450,000 | 270 |
These estimates could increase with higher adoption rates or broader indications.
What are the pricing considerations?
The price for IMVEXXY is projected based on competitive landscape and cost of goods. Given similar oral menopausal therapies:
- Initial wholesale price: USD 50–70 per month per user.
- Insurance reimbursement: 80–90%, reducing out-of-pocket costs.
- Biosimilar or generic competition unlikely early due to novel mechanism and patent exclusivity.
BioPharmX may set a price point near USD 60/month, translating to annual revenues of USD 720 per patient.
What patent and regulatory risks exist?
- Patent life: Patent protection extends to 2030, with potential extensions to 2035 through method of use or formulation patents.
- Market exclusivity: FDA exclusivity expires December 2027.
- Competition: Large pharma may develop alternative SERMs or combination therapies, impacting market share.
Regulatory approval processes in other jurisdictions could influence global market entry, with Europe the next target likely.
What are growth and investment implications?
- Clinical data supporting superior safety or efficacy could accelerate adoption.
- Reimbursement landscape influences market access.
- Off-label use for other estrogen-related conditions remains a potential expansion target but faces regulatory restrictions.
The drug’s commercial success depends on physician familiarity with SERMs, patient acceptance, and payer negotiations.
Key Takeaways
- IMVEXXY enters a USD 2.5 billion menopause treatment market with growing demand.
- It targets a niche of oral therapy for vulvar and vaginal atrophy, competing with local estrogen products.
- Revenue estimates range from USD 90 million in 2024 to USD 270 million by 2028, assuming steady adoption.
- Price setting around USD 60/month aligns with existing oral therapies but will depend on payer coverage and market positioning.
- Patent rights extend to 2030–2035, with regulatory and competitive risks influencing long-term outlook.
FAQ
1. How does IMVEXXY’s mechanism differ from traditional estrogen therapies?
IMVEXXY is a selective estrogen receptor modulator, providing targeted effects on vaginal tissue without systemic estrogen exposure, potentially reducing systemic side effects.
2. What barriers could slow IMVEXXY’s market penetration?
Physician familiarity with existing therapies, insurance/reimbursement hurdles, and safety concerns could limit early adoption.
3. How quickly could IMVEXXY reach peak sales?
If adoption aligns with estimates, peak sales might occur five to seven years post-launch, around USD 300 million annually.
4. Are there geographic expansion opportunities?
Europe and Asia contain sizable menopausal markets; regulatory approval processes and local market dynamics will influence timing and scale.
5. What are potential regulatory hurdles?
Delayed approval in certain markets or additional safety data requirements could affect launch timelines and market share.
References
- MarketsandMarkets. (2022). Hormone replacement therapy market forecast.
- U.S. Census Bureau. (2022). Female population by age group.
- IMS Health. (2022). Women’s health market overview.
[1] MarketsandMarkets. (2022). Hormone replacement therapy market forecast.
[2] U.S. Census Bureau. (2022). Female population by age group.
[3] IMS Health. (2022). Women’s health market overview.
More… ↓
