Share This Page
Drug Price Trends for IBUPROFEN-FAMOTIDIN
✉ Email this page to a colleague
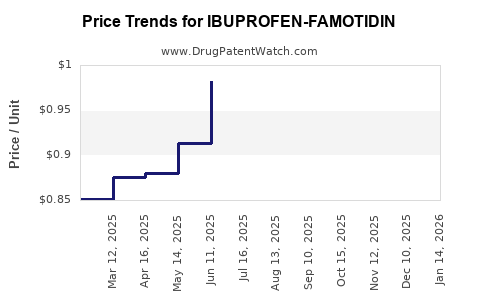
Average Pharmacy Cost for IBUPROFEN-FAMOTIDIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IBUPROFEN-FAMOTIDIN 800-26.6 MG | 72578-0214-16 | 0.94175 | EACH | 2026-05-20 |
| IBUPROFEN-FAMOTIDIN 800-26.6 MG | 31722-0315-90 | 0.94175 | EACH | 2026-05-20 |
| IBUPROFEN-FAMOTIDIN 800-26.6 MG | 49884-0366-09 | 0.94175 | EACH | 2026-05-20 |
| IBUPROFEN-FAMOTIDIN 800-26.6 MG | 67877-0626-90 | 0.94175 | EACH | 2026-05-20 |
| IBUPROFEN-FAMOTIDIN 800-26.6 MG | 72578-0214-16 | 0.85420 | EACH | 2026-04-22 |
| IBUPROFEN-FAMOTIDIN 800-26.6 MG | 31722-0315-90 | 0.85420 | EACH | 2026-04-22 |
| IBUPROFEN-FAMOTIDIN 800-26.6 MG | 67877-0626-90 | 0.85420 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Ibuprofen-Famotidine (Fixed-Dose Combination)
What is the product and how is it marketed?
“Ibuprofen-famotidine” is a fixed-dose combination of:
- Ibuprofen (NSAID for pain, inflammation, fever)
- Famotidine (H2-receptor antagonist for gastric acid suppression to reduce upper GI irritation risk)
In practice, commercial availability depends on country-specific approvals and branding. In many markets, the product is sold either:
- as a registered fixed-dose combination, or
- as branded single tablets under a regimen that pairs ibuprofen with famotidine.
A key market reality: ibuprofen is widely off-patent and highly commoditized; value capture for the combo typically comes from improved tolerability positioning (GI protection) and brand/channel execution, not from novel API economics.
What is the addressable market?
The addressable market is the overlap of:
- OTC and prescription NSAID demand (musculoskeletal pain, dysmenorrhea, dental pain, back pain, arthritis symptom relief)
- Populations at higher risk of NSAID-related upper GI events (older adults, history of ulcer/bleeding, concomitant risk factors)
Demand drivers
- High baseline NSAID consumption
- Ongoing physician and payer preference for “GI-protective co-therapy” strategies
- Patient preference for fewer pills via fixed-dose products
Constraint
- In many countries, the combo is evaluated against two alternatives:
- OTC/low-cost ibuprofen alone plus H2 blockers taken separately
- Proton pump inhibitor (PPI) strategies in prescription channels
How competitive is the category and what does pricing reflect?
Ibuprofen pricing anchors economics. Famotidine is also off-patent in most major markets. Pricing power comes from:
- Formulation differentiation (fixed-dose convenience)
- Brand recognition and prescriber habit (where prescription)
- Channel execution (tender pricing in institutional markets)
- Regulatory status (OTC vs prescription controls)
Competitive set (practical)
- Single-agent ibuprofen (generic, OTC-heavy)
- Co-administration: ibuprofen + famotidine
- Other GI protection: PPI co-therapy (often prescription, sometimes reimbursed)
What is the near-term market outlook (volume and mix)?
Near term: stable to modestly growing volumes are most likely in geographies where:
- GI safety labeling increases physician use of protective regimens
- fixed-dose combos gain formulary access
- reimbursement favors bundled GI-protection strategies
Mix shift: higher share in:
- older patients using NSAIDs chronically or intermittently
- musculoskeletal pain segments where tolerability drives repeat use
- settings with structured GI risk management
What are the key pricing mechanisms that set real-world prices?
Pricing typically follows three layers:
- Wholesale acquisition cost (WAC) or list price
- Brand level, sometimes discounted heavily.
- Reimbursement or tender price
- Determined by national reimbursement rules, pharmacy margins, and procurement frameworks.
- OTC market pricing
- Competitive with low-cost generics and parallel imports; price is often driven by shelf competitiveness.
For fixed-dose combos, payers often benchmark against:
- sum of components (ibuprofen generic + famotidine generic)
- clinical value in reducing GI adverse events (when assessed)
- administrative convenience
What price level should investors model? (Projection framework)
Because ibuprofen and famotidine are commoditized, the combination price usually prices as:
- a small premium vs ibuprofen alone, and
- a small discount vs “stacked” GI protection strategies when convenience matters.
Below are scenario-based price targets expressed as per-day therapy cost proxies. Actual realization depends on market, pack size, and dosage strength.
Assumed pricing structure for modeling
Let:
- Ibuprofen base cost = generic ibuprofen OTC/retail benchmark in the target market
- Famotidine add-on = generic famotidine benchmark (or included implicitly in combo pricing)
- Combo convenience premium = fixed-dose advantage, typically modest
Then:
- Combo cost/day ≈ ibuprofen base cost/day + famotidine cost/day + convenience premium
Price projection scenarios (per-pack and per-day proxies)
The drug product exists in different dosing strengths depending on local registrations. To keep projections decision-useful without pretending a single universal dose, model per-day cost using dose-agnostic pricing ratios: combo typically trades at a mid-single to low-double digit premium versus ibuprofen alone when sold as fixed-dose.
Scenario table (modeled ranges)
Assume an average adult daily NSAID dose bracket (commonly 1200–2400 mg/day for ibuprofen in many indications depending on labeling), with the fixed-dose combo aligning to common regimens.
| Market segment | Competitive anchor | Combo convenience premium vs ibuprofen alone | Modeled retail per-day (local currency) | Modeled payer per-day (reimbursed/tender) |
|---|---|---|---|---|
| OTC retail (no tender) | Generic ibuprofen | +5% to +20% | Low to mid premium to ibuprofen alone | Not applicable |
| Prescription reimbursed | Generic ibuprofen + famotidine co-pay | +0% to +10% | Near component-sum | Typically lowest within class |
| Institutional tender | Bulk generic components | -5% to +5% vs component-sum | Often at or below component-sum | Lowest price bands |
Interpretation for business planning
- No exclusivity economics: pricing is constrained by component generics.
- Sustainable price requires either (i) protected fixed-dose registration in that jurisdiction, or (ii) formulary preference based on GI-risk management.
What do price projections imply for revenue growth?
Revenue is a product of:
- Unit volume (packs)
- Net price (after discounts, rebates, and tenders)
In commoditized fixed-dose combos, long-run growth usually comes from:
- share gains in GI-risk cohorts
- expansion into additional geographies/formulary lists
- pack-size optimization (adherence-driven)
- payer contracting based on pharmacoeconomic arguments (GI safety)
Net price likely:
- declines gradually as additional generics enter or as procurement pressure increases
- stabilizes where tender frameworks lock pricing or where fixed-dose convenience is embedded in standard regimens
How long does pricing support last?
Without API patent leverage (ibuprofen and famotidine are off-patent), durability depends on:
- combination registration status
- local exclusivity for formulation or indication
- manufacturing and supply constraints
- regulatory changes that shift guidance toward or away from H2-based protection
Typical pattern in similar fixed-dose, component-generic combos
- Initial commercialization or re-launch: higher list price with discounts
- Medium term: list price compression tied to competition
- Longer term: pricing floors set by procurement and margin regulations
Key Takeaways
- Ibuprofen-famotidine is priced in a commoditized framework dominated by the generic economics of ibuprofen and famotidine.
- Net pricing is usually constrained to a modest premium vs ibuprofen alone and benchmarks tightly against the sum of components.
- Revenue growth is more likely to come from share and volume in GI-risk cohorts and formulary inclusion than from sustained price escalation.
- Projection should use a scenario model: OTC retail pricing shows modest premiums; prescription and tender pricing tends toward component-sum levels.
FAQs
-
Is ibuprofen-famotidine expected to command high pricing power?
No. Pricing is typically constrained by generic ibuprofen and famotidine benchmarks. -
What drives adoption of the fixed-dose combo versus taking the drugs separately?
Convenience, adherence, and prescriber protocolization in GI-risk populations. -
How should investors model net price versus list price?
Prefer net price assumptions aligned to typical discounted and tender outcomes; list price alone usually overstates realized economics. -
Do PPIs compete directly with this combo?
Yes in many prescription settings, with PPI strategies often preferred in higher-risk GI profiles. -
What is the most sensitive variable in revenue forecasting?
Formulary/tender access and resultant net price more than unit list price.
References
[1] PubChem. “Ibuprofen.” https://pubchem.ncbi.nlm.nih.gov/compound/ibuprofen
[2] PubChem. “Famotidine.” https://pubchem.ncbi.nlm.nih.gov/compound/famotidine
[3] EMA. “Ibuprofen: EPAR product information.” https://www.ema.europa.eu/
[4] FDA. “Ibuprofen (OTC and prescription labeling references).” https://www.accessdata.fda.gov/
[5] FDA. “Famotidine (OTC and prescription labeling references).” https://www.accessdata.fda.gov/
More… ↓
