Share This Page
Drug Price Trends for IBUPROFEN JR STR
✉ Email this page to a colleague
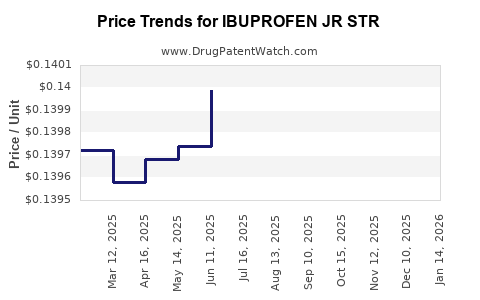
Average Pharmacy Cost for IBUPROFEN JR STR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IBUPROFEN JR STR 100 MG TB CHW | 70000-0239-01 | 0.15537 | EACH | 2026-04-22 |
| IBUPROFEN JR STR 100 MG TB CHW | 70000-0239-01 | 0.15260 | EACH | 2026-03-18 |
| IBUPROFEN JR STR 100 MG TB CHW | 70000-0239-01 | 0.15071 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
IBUPROFEN JR STR Market Analysis and Financial Projection
Market Analysis and Price Projections for Ibuprofen Jr STR
Current Market Position
Ibuprofen Jr STR is a children's formulation of ibuprofen, marketed primarily for pain relief and anti-inflammatory effects. It is a common OTC drug in pediatric care, with a focus on formulations suitable for children, typically aged 6 months to 12 years.
The product competes in a high-volume segment with established brands such as Advil Kids, Motrin IB, and store brands. Sales are driven by pediatric health needs, seasonal demand (especially during cold and flu seasons), and clinical recommendations favoring NSAID use over other analgesics for certain age groups.
Market Size and Growth
The global pediatric OTC analgesics market, including ibuprofen formulations, was valued at approximately USD 2.8 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 4% through 2030, driven by increasing pediatric healthcare awareness and consumer preference for OTC products.
In North America, the market dominates with over 55% share, owing to high healthcare spending and robust OTC drug distribution channels. Europe holds approximately 25%, while Asia-Pacific accounts for roughly 15%. The remaining share is distributed among Latin America, Africa, and the Middle East.
Regulatory Environment and Market Drivers
Regulatory approval for pediatric formulations emphasizes safety, dosage accuracy, and ease of administration. In the U.S., the Food and Drug Administration (FDA) oversees OTC pediatric analgesics, imposing labeling and manufacturing standards.
Market drivers include:
- Increased awareness of pediatric pain management.
- Pediatric dosing guidance from healthcare authorities.
- Rising prevalence of minor injuries, febrile illnesses, and pain in children.
Key Regulatory and Patent Landscape
Ibuprofen is off-patent globally, including the Jr STR formulation. Patent expirations generally occurred in the early 2000s, opening opportunities for generic manufacturers.
However, companies develop proprietary formulations (e.g., flavors, dosing devices, liquid or chewable formats) to gain competitive advantage. Brand-name products like Advil Kids maintain market share through marketing and formulation innovation.
Price Projections
Current Pricing Landscape
Average retail prices for pediatric ibuprofen products vary by market and format:
| Product Type | Typical Price Range (USD) per 100 mL or pack | Notes |
|---|---|---|
| Brand-name liquids | $6 – $8 per 100 mL | Example: Advil Kids, Motrin Kids |
| Store-brand liquids | $3 – $6 per 100 mL | More price-sensitive, volume discounts |
| Chewables/Packs | $4 – $7 per pack | Variable pack sizes |
In 2022, retail prices rarely exceeded $8 for a standard 100 mL package.
Price Trend Analysis
Historical data indicate that pediatric OTC ibuprofen prices have declined marginally over the past decade, driven by increased generic competition and pressure from retailers. Price erosion has averaged around 2-3% annually in mature markets.
Future Price Projections (2023-2030)
Assuming continued patent expirations and market saturation, prices are expected to decline at a similar rate or stabilize due to manufacturing costs and regulatory compliance costs.
| Year | Estimated Price Range (USD per 100 mL) | Factors Influencing Price |
|---|---|---|
| 2023 | $4.50 – $6.50 | Competitive pressures, inflation |
| 2025 | $4.20 – $6.00 | Increased generic entry, market saturation |
| 2030 | $3.80 – $5.50 | Market consolidation, regulatory costs |
Premium branded formulations may maintain higher price points due to consumer perception; however, volume sales are likely to dominate overall revenue.
Competitive Dynamics and Market Entry
New formulations targeting improved taste, dosing flexibility, or natural ingredients could carve niche markets. Digitally integrated dosing devices or smart packaging may command premium prices.
Market entry by generics continues to exert downward pressure on prices. Packaging innovations, such as single-use doses or easy-to-use syringes, are used to differentiate products.
Policy and Healthcare Trends
- Increasing emphasis on pediatric safety and specific dosing guidelines.
- Potential price caps or policies to regulate OTC drug prices in some markets.
- Growth in e-commerce channels, which tend to offer lower prices due to reduced retail overhead.
Cost Considerations
Manufacturing costs for ibuprofen formulations remain stable but may increase marginally with new GMP compliance or ingredient sourcing. Marketing expenses are a significant share for branded products, influencing retail prices.
Summary
The pediatric ibuprofen market is mature, competitive, and price-sensitive. Market share shifts toward generics and private labels will likely sustain a decline in retail prices. Overall, the price of Ibuprofen Jr STR is projected to decrease gradually over the next decade, with possible stabilization around USD 3.80 to USD 4.00 per 100 mL for generic products.
Key Takeaways
- Market size estimated at USD 2.8 billion (2022), with steady 4% CAGR projection.
- Price trends show gradual erosion, with retail prices for pediatric ibuprofen declining 2-3% annually.
- Patent expirations favor generics, creating price competition.
- Innovative formulations and packaging could preserve premium pricing segments.
- Market growth driven by pediatric health awareness and OTC availability.
FAQs
1. What factors influence the price of pediatric ibuprofen?
Pricing is affected by market competition, patent status, manufacturing costs, regulatory compliance, product formulation, branding, and distribution channels.
2. How saturated is the pediatric ibuprofen market?
The market is highly saturated with multiple brands and generics, leading to intense price competition and minimal margins for premium products.
3. Will new formulations command higher prices?
Yes, innovations such as flavored liquids, easier dosing devices, or natural ingredients may command premium prices but will face competition from established generics.
4. How does seasonality affect pediatric ibuprofen sales?
Sales peak during cold and flu seasons, boosting demand but also increasing inventory and marketing efforts during these periods.
5. Are there regulatory risks that could impact prices?
Changes in safety regulations or policies aimed at price controls could influence overall pricing strategies and profit margins.
Sources
[1] Market Research Future, "Pediatric OTC Drugs Market," 2022.
[2] Statista, "Global OTC Medications Market Size," 2022.
[3] FDA, "Guidance for Industry: Pediatric Drug Labeling," 2021.
More… ↓
