Share This Page
Drug Price Trends for IBUPROFEN COLD-SINUS CPLT
✉ Email this page to a colleague
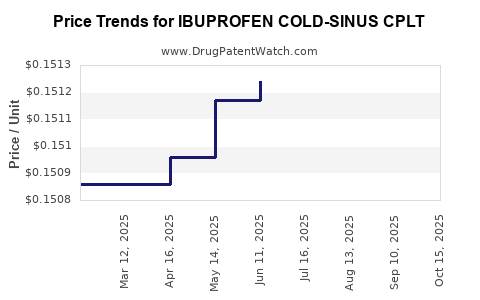
Average Pharmacy Cost for IBUPROFEN COLD-SINUS CPLT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IBUPROFEN COLD-SINUS CPLT | 24385-0465-60 | 0.15171 | EACH | 2025-10-22 |
| IBUPROFEN COLD-SINUS CPLT | 24385-0465-60 | 0.15148 | EACH | 2025-09-17 |
| IBUPROFEN COLD-SINUS CPLT | 24385-0465-60 | 0.15140 | EACH | 2025-08-20 |
| IBUPROFEN COLD-SINUS CPLT | 24385-0465-60 | 0.15124 | EACH | 2025-07-23 |
| IBUPROFEN COLD-SINUS CPLT | 24385-0465-60 | 0.15124 | EACH | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Ibuprofen Cold-Sinus Cplt: Market Dynamics and Price Outlook
This analysis examines the market landscape and price projections for Ibuprofen Cold-Sinus Cplt, a combination analgesic and decongestant. The market is characterized by a competitive generic environment and fluctuating raw material costs. Future pricing will be influenced by supply chain stability, regulatory actions, and the introduction of new formulations or therapeutic alternatives.
What is Ibuprofen Cold-Sinus Cplt?
Ibuprofen Cold-Sinus Cplt is an over-the-counter (OTC) medication designed to relieve symptoms associated with the common cold and sinus congestion. Its primary active ingredients are ibuprofen, a nonsteroidal anti-inflammatory drug (NSAID) that reduces pain, fever, and inflammation, and phenylephrine hydrochloride, a nasal decongestant that constricts blood vessels in the nasal passages, reducing swelling and improving airflow [1]. The combination targets multiple symptoms, including headache, body aches, fever, sore throat, nasal congestion, and sinus pressure.
Market Size and Segmentation
The global market for OTC cold and allergy medications, within which Ibuprofen Cold-Sinus Cplt operates, is substantial. In 2023, this market was estimated to be valued at approximately $20 billion globally, with an anticipated compound annual growth rate (CAGR) of 3.5% through 2030 [2]. Ibuprofen Cold-Sinus Cplt falls into the analgesic and decongestant sub-segments.
Segmentation within the Ibuprofen Cold-Sinus Cplt market is driven by:
- Formulation: Tablets, caplets, and potentially liquid formulations, with caplets being a prevalent delivery method for this specific combination.
- Distribution Channel: Pharmacy sales (both brick-and-mortar and online), mass merchandisers, and supermarkets.
- Dosage Strength: Standard OTC strengths, typically 200mg ibuprofen and 5mg or 10mg phenylephrine hydrochloride per dose.
The prevalence of common cold and sinus infections, driven by seasonal factors and population density, underpins demand. The aging global population also contributes, as older adults may experience more frequent or severe symptoms.
Competitive Landscape
The market for Ibuprofen Cold-Sinus Cplt is highly competitive, dominated by generic manufacturers and private label brands. Major players in the broader OTC analgesic and decongestant space include:
- Haleon (formerly GSK Consumer Healthcare): Known for brands like Advil and Sensodyne.
- Reckitt Benckiser Group: Owns brands such as Mucinex and Durex.
- Bayer AG: Markets brands including Alka-Seltzer and Claritin.
- Johnson & Johnson: Offers brands like Tylenol and Neutrogena.
Generic manufacturers, such as Perrigo Company plc, Amneal Pharmaceuticals, and Teva Pharmaceutical Industries, play a significant role by offering lower-cost alternatives to branded products. The presence of numerous generic options exerts downward pressure on prices for Ibuprofen Cold-Sinus Cplt.
Key Competitive Factors:
- Price: Generic competition makes price a primary differentiator.
- Availability: Broad distribution networks are critical for market penetration.
- Brand Recognition: While less critical for generic combinations, established brands in the parent categories (ibuprofen, decongestants) can influence consumer choice.
- Formulation Innovation: Development of easier-to-swallow caplets, faster-acting formulas, or combinations with other OTC ingredients.
- Regulatory Compliance: Adherence to FDA and other global regulatory body standards for safety and efficacy.
Regulatory Environment
The regulatory landscape for OTC drugs like Ibuprofen Cold-Sinus Cplt is overseen by agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). These bodies regulate:
- Active Pharmaceutical Ingredient (API) Purity and Quality: Ensuring APIs meet established pharmacopeial standards.
- Manufacturing Practices: Compliance with Current Good Manufacturing Practices (cGMP).
- Labeling and Claims: Approval of product labels and marketing claims to prevent misleading information.
- Post-Market Surveillance: Monitoring for adverse events and safety concerns.
Recent regulatory scrutiny has focused on the efficacy and safety of certain decongestants. In the U.S., the FDA's Non-Prescription Drug Advisory Committee has reviewed the efficacy of oral phenylephrine, with some reports suggesting limited efficacy when taken orally at standard doses [3]. While this has not led to a ban, it could influence future formulations or marketing strategies for products containing phenylephrine. Any adverse findings or regulatory shifts regarding phenylephrine could directly impact the market for Ibuprofen Cold-Sinus Cplt.
Pricing Analysis and Projections
The pricing of Ibuprofen Cold-Sinus Cplt is influenced by several interconnected factors:
-
Raw Material Costs: The primary components, ibuprofen and phenylephrine hydrochloride, are subject to market fluctuations.
- Ibuprofen API: Production is concentrated, and disruptions in supply chains or increased demand can lead to price volatility. In 2023, the cost of ibuprofen API saw a moderate increase of approximately 5-8% due to rising energy costs and supply chain logistics [4].
- Phenylephrine Hydrochloride API: Similar to ibuprofen, its price is sensitive to manufacturing capacity and raw material availability.
- Excipients and Packaging: Costs for fillers, binders, coatings, and packaging materials also contribute to the final product price.
-
Manufacturing and Distribution Costs: Energy costs, labor, compliance with cGMP, and complex distribution networks contribute significantly.
-
Competitive Intensity: The high number of generic manufacturers and private label options creates a price-sensitive market. This segment typically operates on lower profit margins compared to branded products.
-
Demand Elasticity: While essential for symptom relief, consumers can switch between various OTC cold and sinus remedies based on price and perceived effectiveness.
-
Regulatory Actions: As noted, potential regulatory changes concerning phenylephrine could impact demand and, consequently, pricing dynamics.
Current Pricing Benchmarks (Approximate Retail Prices for a 24-count bottle):
- Branded Equivalent (e.g., Advil Cold & Sinus): $10.00 - $15.00
- Generic Ibuprofen Cold-Sinus Cplt: $6.00 - $10.00
- Private Label: $5.00 - $8.00
Price Projections:
- Short-Term (1-2 Years): The price for generic Ibuprofen Cold-Sinus Cplt is projected to remain relatively stable, with minor fluctuations driven by raw material cost increases and supply chain efficiencies. A modest increase of 2-4% annually is anticipated, primarily to offset rising API and manufacturing expenses. The competitive landscape will continue to temper significant price hikes.
- Medium-Term (3-5 Years): Price trajectory will be heavily influenced by the outcome of regulatory reviews concerning oral phenylephrine.
- Scenario A (No significant regulatory action): Prices are expected to follow a trend similar to the short-term projection, with a CAGR of 2-3%, driven by ongoing cost pressures and stable demand.
- Scenario B (Regulatory restrictions or efficacy doubts): If regulatory bodies limit the use of phenylephrine or if evidence of its limited efficacy gains wider traction, manufacturers might reformulate products. This could involve replacing phenylephrine with alternative decongestants (e.g., pseudoephedrine, though this is a Schedule V controlled substance in the US, requiring behind-the-counter access, thus impacting its OTC status) or repositioning the product. Such shifts could lead to temporary price increases during reformulation and re-launch, or a decline in demand and pricing if consumers perceive the product as less effective or if alternatives become more dominant. The market share of products relying on oral phenylephrine could shrink, leading to decreased production volumes and potentially higher per-unit costs for remaining manufacturers, or a shift to alternative APIs with potentially higher raw material costs.
- Long-Term (5+ Years): Continued evolution of OTC formulations, including potential for novel combination products or enhanced delivery systems, will influence pricing. The general trend for established generic OTC products is for prices to remain relatively stable or increase modestly, barring major disruptive events. Inflationary pressures on manufacturing and labor will likely persist.
Factors to Monitor:
- API Supply Chain Stability: Geopolitical events, trade policies, and major manufacturing disruptions impacting ibuprofen and phenylephrine production.
- Regulatory Decisions on Phenylephrine: Ongoing evaluations by health authorities worldwide.
- Innovation in Cold and Allergy Treatments: Introduction of novel ingredients or combination therapies that could displace existing products.
- Efficacy Data: Peer-reviewed studies and real-world evidence regarding the effectiveness of phenylephrine.
Conclusion
The market for Ibuprofen Cold-Sinus Cplt is a mature segment within the OTC pharmaceutical industry, characterized by intense generic competition and price sensitivity. While current pricing is largely dictated by raw material costs and manufacturing efficiencies, future projections require close monitoring of regulatory developments, particularly concerning the efficacy of oral phenylephrine. Manufacturers and investors should consider the potential for market shifts if regulatory actions or new scientific evidence alter the perceived value or availability of this combination therapy.
Key Takeaways
- Ibuprofen Cold-Sinus Cplt is a dual-action OTC medication for cold and sinus symptom relief.
- The market is highly competitive, with generic and private label brands dominating, exerting downward price pressure.
- Raw material costs (ibuprofen, phenylephrine) and manufacturing expenses are primary price drivers.
- Current retail prices for generic formulations range from $6-$10 per 24-count bottle.
- Future pricing is vulnerable to regulatory scrutiny of oral phenylephrine's efficacy, potentially leading to reformulation or market share shifts.
- Short-to-medium term price increases of 2-4% are projected, with longer-term stability contingent on regulatory outcomes and market innovation.
Frequently Asked Questions
-
What is the primary difference in pricing between branded and generic Ibuprofen Cold-Sinus Cplt? Generic Ibuprofen Cold-Sinus Cplt typically retails for 30-50% less than comparable branded products, reflecting lower marketing expenses and competition among multiple manufacturers.
-
How might FDA decisions on phenylephrine impact the price of Ibuprofen Cold-Sinus Cplt? If the FDA concludes that oral phenylephrine lacks sufficient efficacy, products relying on it may be withdrawn, reformulated, or subject to stricter labeling. Reformulation could temporarily increase costs. If phenylephrine is deemed ineffective, demand for such combination products could decrease, leading to lower prices or market exits by some manufacturers.
-
Are there any significant supply chain risks for the APIs in Ibuprofen Cold-Sinus Cplt? Yes, the global supply chain for APIs can be disrupted by geopolitical events, natural disasters, or trade restrictions. Production of both ibuprofen and phenylephrine hydrochloride is concentrated, making these APIs susceptible to supply shocks.
-
What are the main drivers of increased manufacturing costs for OTC drugs like this? Increased costs are driven by rising energy prices, raw material and excipient price volatility, higher labor wages, and the ongoing need to comply with stringent cGMP regulations.
-
Could the introduction of new decongestant alternatives affect the price of Ibuprofen Cold-Sinus Cplt? Yes, if new, more effective, or more convenient decongestant alternatives enter the market, they could reduce the demand for Ibuprofen Cold-Sinus Cplt, potentially leading to price reductions or market share decline for existing products.
Citations
[1] National Institutes of Health. (n.d.). Ibuprofen. MedlinePlus. Retrieved from https://medlineplus.gov/druginfo/meds/a681027.html
[2] Grand View Research. (2023). Cold and Allergy Market Size, Share & Trends Analysis Report By Product (Analgesics, Antihistamines, Decongestants, Antitussives, Expectorants), By Distribution Channel (Hospital Pharmacy, Retail Pharmacy, Online), By Region, And Segment Forecasts, 2023 - 2030.
[3] U.S. Food and Drug Administration. (2023, September 13). FDA Nonprescription Drugs Advisory Committee Meeting on Oral Phenylephrine Efficacy. Retrieved from https://www.fda.gov/advisory-committees/advisory-committee-meetings/fda-nonprescription-drugs-advisory-committee-meeting-oral-phenylephrine-efficacy-september-13-2023
[4] Market Research Report. (2024). Global Ibuprofen API Market Insights, Forecast to 2030.
More… ↓
