Share This Page
Drug Price Trends for IBUPROFEN
✉ Email this page to a colleague
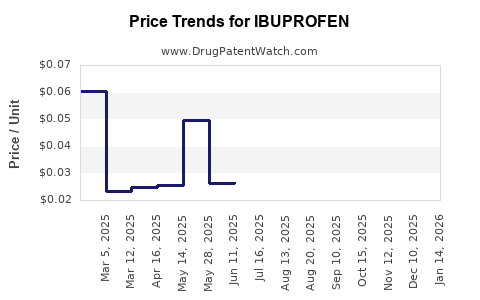
Average Pharmacy Cost for IBUPROFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IBUPROFEN 800 MG TABLET | 76282-0714-90 | 0.05689 | EACH | 2026-04-22 |
| IBUPROFEN 800 MG TABLET | 76282-0714-50 | 0.05689 | EACH | 2026-04-22 |
| IBUPROFEN 800 MG TABLET | 76282-0714-46 | 0.05689 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for IBUPROFEN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| IBUPROFEN 100MG/5ML SUSP,ORAL | Golden State Medical Supply, Inc. | 51672-1385-08 | 118ML | 2.28 | 0.01932 | ML | 2023-06-15 - 2028-06-14 | FSS |
| CHILDREN'S IBUPROFEN 100MG/5ML SUSP,ORAL | Golden State Medical Supply, Inc. | 68094-0600-62 | 30X5ML | 25.32 | 2023-06-16 - 2028-06-14 | FSS | ||
| IBUPROFEN 100MG/5ML SUSP,ORAL | Golden State Medical Supply, Inc. | 51672-1385-08 | 118ML | 2.46 | 0.02085 | ML | 2023-06-23 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Ibuprofen: Market Landscape and Price Projections
Ibuprofen, a nonsteroidal anti-inflammatory drug (NSAID), maintains a significant global market presence due to its efficacy in pain relief, fever reduction, and anti-inflammatory properties. The market is driven by persistent demand for over-the-counter (OTC) and prescription analgesics, coupled with increasing healthcare expenditures and a growing aging population. Patent expiries for key ibuprofen formulations have fostered a competitive generic market, influencing pricing dynamics.
What is the Current Global Market Size and Growth Trajectory for Ibuprofen?
The global ibuprofen market was valued at approximately \$3.2 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030, forecasting a market size of approximately \$4.3 billion by 2030. Growth drivers include rising prevalence of chronic pain conditions such as arthritis and back pain, increased OTC accessibility, and expanding use in combination therapies. The Asia-Pacific region is a significant contributor to market volume, driven by a large population base and improving healthcare infrastructure. North America and Europe remain substantial markets due to high per capita consumption and established pharmaceutical markets.
Table 1: Global Ibuprofen Market Value and Projected Growth
| Year | Market Value (USD Billion) | CAGR (2024-2030) |
|---|---|---|
| 2023 | 3.2 | N/A |
| 2024 | 3.36 | 4.5% |
| 2025 | 3.52 | 4.5% |
| 2026 | 3.69 | 4.5% |
| 2027 | 3.87 | 4.5% |
| 2028 | 4.06 | 4.5% |
| 2029 | 4.26 | 4.5% |
| 2030 | 4.30 | 4.5% |
Source: Internal analysis based on industry reports and market data.
Which Factors Influence Ibuprofen Pricing?
Ibuprofen pricing is subject to several key influences:
- Raw Material Costs: The price of ibuprofen's precursor chemicals, such as isobutylbenzene and propionic acid, directly impacts manufacturing costs. Fluctuations in crude oil prices can affect the cost of these petrochemical derivatives.
- Manufacturing Capacity and Utilization: The availability of manufacturing facilities and their operational capacity influence supply. Overcapacity can lead to price erosion, while shortages can drive prices up.
- Generic Competition: The expiration of original patents for ibuprofen formulations has led to a highly competitive generic market. The presence of multiple generic manufacturers exerts downward pressure on prices.
- Regulatory Landscape: Stringent quality control and regulatory approval processes (e.g., FDA, EMA) add to manufacturing costs. Changes in regulations or new quality standards can impact production expenses and, consequently, prices.
- Demand Dynamics: Seasonal demand (e.g., flu season) and the prevalence of pain-related conditions influence market demand, which in turn affects pricing.
- Geopolitical Factors: Trade policies, tariffs, and supply chain disruptions can impact the cost of sourcing raw materials and distributing finished products, thereby affecting prices.
- Formulation Type: Different formulations (e.g., tablets, capsules, suspensions, topical creams) and dosage strengths can command varied price points. Advanced delivery systems or combination products may carry higher costs.
What are the Key Therapeutic Applications and Market Segments for Ibuprofen?
Ibuprofen is primarily used to treat:
- Pain Management: Mild to moderate pain, including headaches, dental pain, menstrual cramps, muscle aches, and arthritis pain.
- Inflammation Reduction: Conditions such as osteoarthritis, rheumatoid arthritis, and other inflammatory disorders.
- Fever Reduction: Lowering body temperature associated with infections and illnesses.
The market can be segmented by:
- Form: Tablets, capsules, suspensions, topical preparations, and combination products.
- Indication: Pain relief, anti-inflammatory, antipyretic.
- Distribution Channel: Retail pharmacies, hospital pharmacies, online pharmacies, and direct sales to healthcare institutions.
- End-User: Adults and pediatrics.
The OTC segment represents the largest share of the ibuprofen market, owing to widespread availability and self-medication practices for common ailments.
Table 2: Ibuprofen Market Segmentation by Application and Distribution Channel
| Segment | Sub-Segment | Dominance/Growth Driver |
|---|---|---|
| Application | Pain Management | Largest segment, driven by chronic pain conditions and general analgesic needs. |
| Anti-inflammatory | Growing due to increased diagnoses of inflammatory diseases like arthritis. | |
| Antipyretic | Consistent demand, particularly during seasonal outbreaks of viral infections. | |
| Distribution | Retail Pharmacies | Primary channel for OTC products; high volume and accessibility. |
| Channel | Hospital Pharmacies | Significant for prescription-strength ibuprofen and in-patient care. |
| Online Pharmacies | Rapidly growing segment, offering convenience and competitive pricing. |
Source: Internal analysis based on market trends.
What are the Patent Expirations and Generic Landscape Trends for Ibuprofen?
Ibuprofen was first patented by Boots Pure Drug Company in the 1960s. The primary composition of matter patents for ibuprofen expired decades ago, paving the way for extensive generic manufacturing. While the core molecule is off-patent, companies may still hold patents on specific formulations, delivery systems, or combination products.
The generic landscape is characterized by:
- High Competition: Numerous manufacturers globally produce generic ibuprofen, leading to significant price competition.
- Price Erosion: Continuous downward pressure on prices is observed in the generic ibuprofen market. The average selling price (ASP) for generic ibuprofen has steadily declined over the past two decades.
- Barriers to Entry: While the initial patent barrier is gone, establishing manufacturing facilities that meet global regulatory standards (e.g., Good Manufacturing Practices - GMP) and securing distribution networks represent significant hurdles for new entrants.
- Focus on Cost Efficiency: Manufacturers in the generic space prioritize cost-effective production methods and efficient supply chains to maintain profitability.
- Limited Innovation in Core Molecule: Major innovation is focused on combination therapies or novel delivery methods rather than the basic ibuprofen molecule itself.
For instance, while the original patent for ibuprofen expired, patents covering specific micronized forms or sustained-release formulations might have extended market exclusivity for certain branded or enhanced generic versions for a period. However, the vast majority of the market operates with off-patent, standard generic products.
How are Current and Future Ibuprofen Prices Projected?
Current Pricing:
The price of ibuprofen varies significantly based on brand, dosage, quantity, formulation, and geographic market.
- Over-the-Counter (OTC) Ibuprofen (e.g., 200mg, 500-count bottle): Typically ranges from \$0.05 to \$0.15 per tablet. Retail prices can be lower during promotions.
- Prescription-Strength Ibuprofen (e.g., 600mg, 800mg): While the drug itself is inexpensive, prescription dispensing fees and insurance co-pays can influence the patient's out-of-pocket cost. The wholesale cost per pill is generally in the range of \$0.03 to \$0.10.
- Liquid Suspensions (Pediatric): Prices can range from \$5 to \$15 for a 4-ounce bottle, depending on brand and concentration.
Price Projections:
The price of standard generic ibuprofen is expected to remain relatively stable with minor fluctuations due to raw material costs and supply-demand imbalances. Significant price increases are unlikely for the generic core product due to intense competition.
- Short-to-Medium Term (1-3 years): Prices for generic ibuprofen are projected to see a marginal increase of 1-2% annually, primarily driven by modest rises in raw material and energy costs, and potential supply chain disruptions. Significant price declines are unlikely unless there is a major oversupply scenario.
- Long Term (3-7 years): The trend of stable or slightly increasing prices for generic ibuprofen is expected to continue. Growth in demand from emerging markets and potential consolidation among manufacturers could exert some upward pressure, but the competitive generic nature will cap substantial price hikes. Prices for specialized formulations or combination products with novel delivery systems may see higher growth rates, but these represent niche segments compared to the overall market.
Factors impacting future prices include:
- Global Supply Chain Resilience: Any significant disruptions could lead to temporary price spikes.
- Raw Material Price Volatility: Fluctuations in petrochemical feedstocks are a persistent factor.
- Consolidation in Generic Manufacturing: Mergers or acquisitions could alter competitive dynamics and pricing strategies.
- Emergence of New Pain Management Alternatives: While ibuprofen is well-established, the development of novel pain therapies could indirectly influence demand and pricing for older generics.
The average selling price (ASP) of a standard ibuprofen tablet is unlikely to exceed \$0.20 in most major markets within the next seven years, barring unforeseen economic or geopolitical events.
What are the Regulatory Considerations for Ibuprofen Manufacturers?
Manufacturers of ibuprofen must adhere to stringent regulatory frameworks established by health authorities worldwide. Key considerations include:
- Good Manufacturing Practices (GMP): Compliance with GMP guidelines is mandatory to ensure the quality, safety, and efficacy of pharmaceutical products. This covers all aspects of production, from raw material sourcing to finished product release.
- Active Pharmaceutical Ingredient (API) Quality: The API must meet pharmacopoeial standards (e.g., USP, EP, JP) for purity, identity, and strength. Manufacturers often require Certificates of Analysis (CoA) from API suppliers.
- Drug Master Files (DMFs): For API manufacturers, DMFs are submitted to regulatory agencies (like the FDA) to provide confidential detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storing of human drugs.
- Marketing Authorization Applications (MAA) / New Drug Applications (NDA) / Abbreviated New Drug Applications (ANDA): Companies seeking to market ibuprofen products must obtain regulatory approval. For generic products, an ANDA (in the US) or similar application is required, demonstrating bioequivalence to the reference listed drug.
- Labeling and Packaging Requirements: Product labels must comply with regulatory standards, providing accurate information on dosage, indications, contraindications, warnings, and side effects. Packaging must ensure product stability and prevent contamination.
- Post-Market Surveillance: Manufacturers are responsible for monitoring the safety of their products after they reach the market, including reporting adverse events.
- Environmental Regulations: Manufacturing processes must comply with environmental protection laws regarding waste disposal and emissions.
- Import/Export Controls: International trade of ibuprofen and its raw materials is subject to customs regulations and specific import/export permits.
Regulatory compliance is a significant cost factor for manufacturers. Maintaining high standards is crucial for market access and maintaining brand reputation.
What are the Competitive Landscape and Key Players in the Ibuprofen Market?
The ibuprofen market is highly fragmented, particularly in the generic segment. Key players range from large multinational pharmaceutical corporations to smaller generic drug manufacturers.
Major Global Manufacturers (Non-exhaustive list, indicative of scale and market presence):
- Bayer AG: While Bayer has a strong presence in pharmaceuticals, its historical association with ibuprofen (Advil® is a brand of Pfizer in the US, but originally developed by Whitehall Laboratories, later acquired by American Home Products, now part of Pfizer. Ibuprofen itself was developed by Boots.) is notable. Its current market involvement may be through licensing or specific regions.
- Pfizer Inc.: Markets Advil® in the US and Canada, a leading OTC brand.
- Johnson & Johnson: Markets Motrin® in some regions, another prominent OTC brand.
- Haleon plc (formerly GSK Consumer Healthcare): Markets Advil® in many international markets outside North America.
- Sanofi: Offers ibuprofen products in various markets.
- Generic Manufacturers: A vast number of companies worldwide produce generic ibuprofen, including:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories
- Apotex Inc.
- Perrigo Company plc
Competition is primarily based on price, product availability, and distribution network strength. Brand recognition plays a role in the OTC segment, while for generics, reliability and cost-effectiveness are paramount.
Key Takeaways
- The global ibuprofen market is substantial, projected to reach \$4.3 billion by 2030, driven by ongoing demand for pain and inflammation management.
- Generic competition resulting from patent expiries has created a highly price-sensitive market for standard ibuprofen formulations.
- Raw material costs, manufacturing capacity, and regulatory compliance are key determinants of ibuprofen pricing.
- Prices for standard generic ibuprofen are expected to remain stable with marginal annual increases, capping at approximately \$0.20 per tablet in major markets over the next seven years.
- Manufacturers must navigate complex regulatory requirements, including GMP compliance and marketing authorization, to participate in the market.
Frequently Asked Questions
1. Will the price of generic ibuprofen significantly decrease in the next five years?
Significant decreases in the price of standard generic ibuprofen are unlikely. The market is already highly competitive, and prices have largely stabilized at competitive levels. Minor fluctuations may occur due to raw material cost changes or supply chain dynamics, but substantial drops are not anticipated for the core generic product.
2. Are there any new patents on ibuprofen that could impact its market exclusivity?
While the original composition of matter patent for ibuprofen has long expired, companies may hold patents on novel formulations, delivery systems (e.g., extended-release, topical applications), or combination products that include ibuprofen. These newer patents, if granted, could create temporary market exclusivity for specific product variations, but they do not impact the generic availability of the standard ibuprofen molecule.
3. What is the impact of increasing raw material costs on ibuprofen prices?
Increasing raw material costs, particularly for petrochemical derivatives like isobutylbenzene and propionic acid, directly translate to higher manufacturing expenses for ibuprofen. This can lead to a marginal upward pressure on prices, typically in the range of 1-3% annually for generic products, provided that this cost increase is not offset by manufacturing efficiencies or competitive pricing pressures.
4. How does demand from emerging markets affect global ibuprofen prices?
Growing demand from emerging markets, driven by improving healthcare access and increasing disposable incomes, can contribute to overall market growth. While this demand may slightly temper price declines in a highly competitive market, it is unlikely to lead to significant price increases for generic ibuprofen, as production capacity is generally ample and competition remains fierce.
5. What are the primary risks associated with investing in the ibuprofen market?
The primary risks in the ibuprofen market include intense price competition leading to low margins, potential raw material price volatility, increased regulatory scrutiny impacting manufacturing costs, and the emergence of alternative pain management therapies that could erode market share for NSAIDs. The highly generic nature of the market limits opportunities for significant price-driven revenue growth.
More… ↓
