Last updated: February 13, 2026
Hydroxychloroquine (HCQ), a generic antimalarial and autoimmune drug, gained widespread attention during the COVID-19 pandemic, leading to fluctuating demand and pricing. The current market environment reflects a shift toward stable, long-term use primarily in autoimmune disorders like lupus and rheumatoid arthritis.
Market Overview
Production and Suppliers
- Major producers include Novartis, Teva, Mylan, and Sandoz.
- Generic manufacturers dominate global supply, resulting in low unit costs.
Regulatory Status
- Approved for malaria, lupus, rheumatoid arthritis in most jurisdictions.
- FDA revoked emergency use authorization for COVID-19 treatment in June 2020 due to lack of efficacy.
Demand Trends
- Steady demand for autoimmune indications.
- COVID-19-related surge has diminished; no sustained increase expected.
| Geographic Market Distribution |
Region |
Market Share (%) |
Key Factors |
| North America |
45 |
Established autoimmune treatments, regulatory stability |
| Europe |
25 |
Similar to North America, high prevalence of autoimmune conditions |
| Asia-Pacific |
20 |
Growing autoimmune diagnosis, emerging manufacturing |
| Rest of World |
10 |
Lower access, supply constraints |
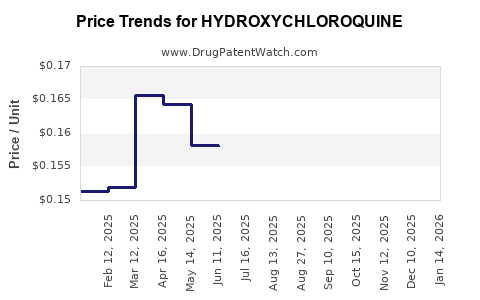
Pricing Dynamics
Pre-Pandemic Pricing
- Wholesale acquisition cost (WAC): $0.10 - $0.25 per tablet (200 mg).
- Typical pack: 60 tablets, average retail: ~$15–$25.
Pandemic-Driven Price Fluctuations
- During 2020, prices doubled in some regions due to supply shortages.
- Short-lived spike; supply recovered by late 2020, prices normalized.
Current Prices
- WAC: approximately $0.10–$0.15 per tablet (200 mg).
- Retail: $10–$15 for a 60-tablet pack.
Supply Chain and Cost Factors
- Raw material costs have remained stable.
- Manufacturing efficiencies and increased competition keep prices low.
- Regulatory barriers are minimal for approved indications.
Future Price Projections
Short-Term Outlook (next 1-2 years)
- Prices stabilize around current levels due to consistent demand for approved uses.
- No significant supply disruptions foreseen, barring new global health emergencies.
Medium to Long-Term Outlook (3-5 years)
- Prices likely stay within the current low range as generic saturation continues.
- Slight decline possible if production costs decrease or patent/litigation barriers evolve.
- Use in COVID-19 treatment unlikely to reemerge as a major demand driver.
Key Factors Influencing Market Trajectory
- Regulatory Decisions: Approval or revocation of emergency use across different countries.
- New Indications: Exploration of HCQ for other diseases could marginally influence demand.
- Patent and Litigation: HCQ’s patent expiration in most markets has led to increased generics, reducing prices.
- Supply Chain Stability: Raw material availability and manufacturing capacity influence prices.
Competitive Landscape
| Company |
Market Share |
Focus Area |
Key Strengths |
| Novartis |
Leading |
Production, R&D for autoimmune applications |
Wide global reach |
| Teva |
Major |
Cost-efficient manufacturing |
Large distribution network |
| Mylan |
Significant |
Generic production |
Price competitiveness |
| Sandoz |
Notable |
Biosimilar and generic offerings |
Regulatory expertise |
Final Observations
Hydroxychloroquine remains a low-cost, widely available generic with stable demand primarily driven by autoimmune disorders. Its price stability is expected to persist unless new regulatory policies or medical evidence alter its use profile significantly.
Key Takeaways
- Hydroxychloroquine prices have stabilized post-pandemic, remaining low due to generic competition.
- No foreseeable price spikes unless demand shifts for new indications or supply disruptions occur.
- The global market is segmented, with North America and Europe holding most of the demand for approved uses.
- Cost factors remain stable; production is consolidated among a few large suppliers.
- Future market dynamics will hinge on regulatory decisions, emerging research, and supply chain stability.
FAQs
1. Will hydroxychloroquine prices increase due to new COVID-19 variants?
Unlikely. The drug is no longer authorized for COVID-19 treatment in most markets. Demand remains steady for autoimmune diseases.
2. Are there new patents or formulations in development?
No significant patents. Most formulations are off-patent, with generic manufacturers dominating the market.
3. How does hydroxychloroquine compare with other autoimmune drugs in pricing?
It is considerably cheaper than biologics and newer therapies, making it a cost-effective option.
4. Could geopolitical issues impact supply or pricing?
Potentially, especially if raw material supplies are disrupted or if export restrictions are introduced.
5. What factors could lead to a price decline?
Market oversaturation, increased competition, or regulatory hurdles limiting off-label uses.
References
- FDA. "Hydroxychloroquine Sulfate Tablets, Guidance for Industry," 2020.
- IQVIA. "Global Pharmaceutical Market Trends," 2022.
- Statista. "Leading Pharmaceutical Manufacturers by Revenue," 2022.
- World Health Organization. "Guidelines on the Treatment of Autoimmune Disorders," 2021.
- American Society of Rheumatology. "Hydroxychloroquine Usage Trends," 2022.