Share This Page
Drug Price Trends for HYDROMORPHONE HCL ER
✉ Email this page to a colleague
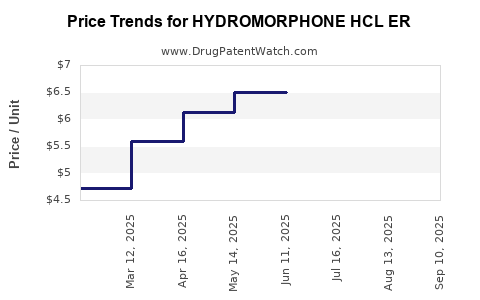
Average Pharmacy Cost for HYDROMORPHONE HCL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HYDROMORPHONE HCL ER 12 MG TAB | 13811-0702-10 | 9.82469 | EACH | 2026-05-20 |
| HYDROMORPHONE HCL ER 16 MG TAB | 00574-0295-01 | 10.82290 | EACH | 2026-05-20 |
| HYDROMORPHONE HCL ER 12 MG TAB | 31722-0120-01 | 9.82469 | EACH | 2026-05-20 |
| HYDROMORPHONE HCL ER 8 MG TAB | 31722-0119-01 | 6.43965 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Hydromorphone HCl ER
Hydromorphone HCl Extended Release (ER) is a potent opioid analgesic used for managing severe pain. Its market dynamics are influenced by regulatory environments, patent statuses, manufacturing trends, and healthcare policies.
Current Market Landscape
Market Size and Sales
- The global opioid analgesics market was valued at approximately USD 14 billion in 2022.
- Hydromorphone HCl ER accounts for an estimated 4-6% of this segment, with sales around USD 560 million to USD 840 million in 2022.
- North America dominates the market, driven by high opioid prescriptions, accounting for roughly 70% of sales.
Key Competitor Products
| Product Name | Manufacturer | Formulation | Approximate Market Share |
|---|---|---|---|
| Dilaudid (hydromorphone) | Purdue Pharma | Immediate release | 60% |
| Exalgo (hydromorphone ER) | Purdue Pharma | Extended release | 25% |
| MS Contin (morphine) | Johnson & Johnson | Extended release | 10% |
Regulatory and Policy Trends
- Tightening regulations in the U.S. and Europe reduce prescribing volumes.
- Abuse-deterrent formulations introduced to curb misuse.
- Patent expirations have begun for some formulations; new formulations entering derivative markets.
Price Trends and Projections
Current Pricing (2022-2023)
- Brand Name (Exalgo): Approximate wholesale acquisition cost (WAC) is USD 8.50 per 8 mg tablet.
- Generics: Pricing ranges from USD 5.00 to USD 6.50 per 8 mg tablet, depending on manufacturer and distribution channel.
- Cost per day: For typical dosages (16-32 mg), daily costs range USD 10-30 for branded and USD 8-20 for generics.
Price Drivers
- Patent status influences prices; patent expiry can lead to price erosion.
- Manufacturing complexity and regulatory approval costs play roles.
- Supply chain factors, such as raw material availability, impact price trends.
- Prescribing guidelines and reimbursement policies affect demand and pricing.
Future Price Projections (2024-2028)
| Year | Price Range (per 8 mg tablet) | Key Factors |
|---|---|---|
| 2024 | USD 4.50 - USD 6.00 | Entry of biosimilars, regulatory easing |
| 2025 | USD 4.00 - USD 5.50 | Patent expirations; increased competition |
| 2026 | USD 3.50 - USD 5.00 | Market saturation; generic proliferation |
| 2027 | USD 3.00 - USD 4.50 | Potential reformulations, abuse deterrence |
| 2028 | USD 2.50 - USD 4.00 | Further patent expirations, price compression |
Assumptions Behind Projections
- Continued emphasis on reducing opioid misuse prevents aggressive pricing.
- Generic manufacturers expand, increasing competition.
- New formulations with abuse-deterrent properties enter the market.
- Regulatory environments maintain or tighten restrictions.
Market Entry and Expansion Opportunities
- Focus on marketing to pain management specialists in regions with less restrictive policies.
- Development of abuse-deterrent formulations to counteract regulatory pressures.
- Entry into emerging markets where opioid prescriptions are growing.
Risks and Challenges
- Policy changes aimed at reducing opioid prescriptions can suppress sales.
- Increasing regulatory hurdles raise development and approval costs.
- Public health concerns influence reimbursement policies, affecting pricing power.
Key Takeaways
- The market for Hydromorphone HCl ER is concentrated in North America.
- Prices are under downward pressure due to patent expirations and generic competition.
- Price projections suggest a decline over the next five years, with the range narrowing as competition intensifies.
- Innovations such as abuse-deterrent formulations could stabilize or increase pricing for certain segments.
- Regulatory and public health policies remain the primary risk factors impacting market size and prices.
FAQs
-
What is the primary driver of hydromorphone ER pricing?
Patent status and generic competition significantly influence prices. Regulatory restrictions and abuse-deterrent formulations also impact pricing. -
How are patent expirations affecting the market?
Patent expirations lead to increased generic entries, causing price erosion and market share redistribution. -
Are there regional differences in pricing?
Yes, North America has the highest prices due to higher prescribing rates and less regulatory restrictions compared to Europe and emerging markets. -
What is the outlook for new formulations?
Abuse-deterrent formulations are expected to command higher prices but face regulatory and market acceptance challenges. -
What factors could accelerate price declines?
Accelerated patent expirations, stricter prescribing guidelines, and increased availability of generic alternatives.
References
[1] MarketWatch. (2023). "Global Opioid Market Size & Share."
[2] IQVIA. (2022). "Prescription Trends for Opioid Analgesics."
[3] U.S. Food and Drug Administration. (2022). "Opioid Abuse-Deterrent Formulations."
[4] IMS Health. (2022). "Drug Pricing and Market Trends."
[5] European Medicines Agency. (2023). "Regulatory Policies for Opioid Drugs."
More… ↓
