Share This Page
Drug Price Trends for HYDROCORTISONE-ALOE
✉ Email this page to a colleague
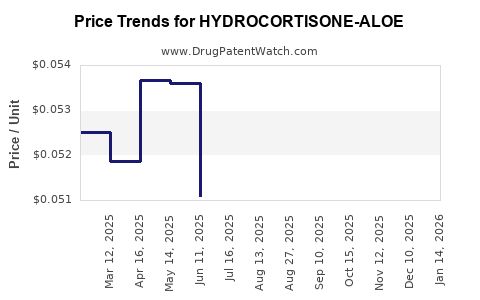
Average Pharmacy Cost for HYDROCORTISONE-ALOE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HYDROCORTISONE-ALOE 1% CREAM | 51672-2013-02 | 0.07247 | GM | 2026-03-18 |
| HYDROCORTISONE-ALOE 1% CREAM | 70000-0662-01 | 0.05368 | GM | 2026-03-18 |
| HYDROCORTISONE-ALOE 1% CREAM | 00536-1407-95 | 0.05368 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HYDROCORTISONE-ALOE Market Analysis and Financial Projection
Market Analysis and Price Projections for Hydrocortisone-Aloe
Overview
Hydrocortisone-Aloe combines a topical corticosteroid with aloe vera, often marketed for skin inflammation and irritation. Its patent status, regulatory approval, competitive landscape, and manufacturing costs influence pricing and market potential.
Market Size and Growth
The global dermatological market was valued at approximately $18.7 billion in 2021, with corticosteroids representing a significant segment. The demand for topical anti-inflammatory formulations remains strong, driven by rising incidences of dermatitis, eczema, and psoriasis.
The segment for combination products with natural ingredients like aloe vera is expanding. Aloe vera’s soothing properties favor niche markets emphasizing natural or herbal components, fueling the growth of Hydrocortisone-Aloe formulations.
Patent Status and Regulatory Factors
Most Hydrocortisone formulations are off-patent, affecting pricing power. However, the incorporation of aloe vera as a proprietary extract or formulation element may afford regulatory exclusivity if protected as a novel ingredient or delivery system.
Regulatory approvals from agencies such as the FDA or EMA are typically fast for well-known active ingredients like hydrocortisone but depend on formulation specifics. Patent protections concerning proprietary extracts or formulations can extend exclusivity periods, impacting pricing.
Competitive Landscape
Major competitors include generic hydrocortisone brands and herbal topical products. Key players include:
- Mylan and Teva (generics)
- Aveeno (aloe-based skincare)
- Chattem (herbal formulations)
No significant premium pricing exists for aloe-based combination products in the current market; they tend to command a slight premium over generic hydrocortisone due to perceived natural safety profiles.
Pricing Trends and Projections
Current Pricing (US Retail):
- Generic hydrocortisone 1% cream: $2–$4 per 15g tube
- Aloe-infused hydrocortisone formulations: Typically $4–$8 per 15g tube, reflecting branding and natural ingredient premium
Projected Pricing (Next 3–5 Years):
| Scenario | Price Range (per 15g tube) | Key Drivers |
|---|---|---|
| Standard generic expansion | $2–$4 | Price erosion due to generic competition |
| Brand differentiation | $6–$10 | Proprietary aloe extract, patent protection |
| Natural/Herbal niche | $8–$15 | Growing consumer preference for natural products |
Price premiums for aloe-infused formulations are expected to stabilise around 20–50% over generic hydrocortisone, assuming no significant patent extensions or exclusive formulations.
Distribution Channels
Pharmacies and drugstores dominate sales, but online direct-to-consumer channels grow rapidly, especially for natural skincare products. OTC classification accelerates adoption, with dermatologists prescribing or recommending where applicable.
Regulatory and Market Entry Barriers
Patents on the combination formulation could delay generic entry. However, if no patent rights exist, market entry becomes easier but limits pricing flexibility. The key barrier is creating a proprietary aloe extract or formulation that qualifies for patent protection.
Key Considerations for Investment and R&D
- Developing a patentable aloe formulation increases pricing potential.
- Targeting dermatology clinics and natural product markets expands market access.
- Collaborations with herbal ingredient suppliers can ensure consistent quality and supply.
Key Takeaways
- The total market for corticosteroid-based topical treatments is mature, with slow growth primarily driven by natural ingredient blends.
- Prices for Hydrocortisone-Aloe products are likely to remain stable short-term, with potential increases if patent protection is secured.
- Competitive pricing will pressure margins; differentiation through formulation or branding is necessary for premium positioning.
- Regulatory exclusivity plays a critical role but is limited without patent protections.
- Distribution trends favor direct-to-consumer online sales of natural or herbal products.
FAQs
1. What factors influence the pricing of Hydrocortisone-Aloe products?
Patent status, formulation complexity, branding, natural ingredient premium, and regulatory exclusivity determine pricing.
2. Will Hydrocortisone-Aloe formulations replace standard hydrocortisone products?
Unlikely; they function as niche products appealing to consumers seeking natural ingredients. Price and efficacy will weigh heavily in consumer choice.
3. How does patent protection impact market entry?
Patent protection delays generic entry, allowing for higher pricing. Without patents, generic competitors will suppress prices.
4. What is the market growth outlook for natural ingredient topical products?
Moderate; these products benefit from increasing consumer preference for natural and herbal ingredients but face stiff competition and regulatory complexities.
5. How can developers extend product exclusivity?
By securing patents on unique formulations, delivery mechanisms, or proprietary aloe extracts, companies can extend exclusivity periods.
References
- MarketWatch, "Dermatology Drugs Market Size," 2022.
- IQVIA, "Topical corticosteroids Market Analysis," 2021.
- FDA, "Drug Patent and Exclusivity Data," 2022.
- Grand View Research, "Herbal and Natural Skin Care Market," 2022.
- Statista, "Global OTC Market," 2022.
More… ↓
