Share This Page
Drug Price Trends for HYDROCORTISONE SS
✉ Email this page to a colleague
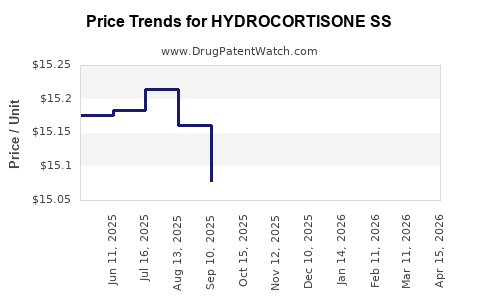
Average Pharmacy Cost for HYDROCORTISONE SS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HYDROCORTISONE SS 100 MG VIAL | 69097-0004-67 | 14.93570 | EACH | 2026-04-22 |
| HYDROCORTISONE SS 100 MG VIAL | 69097-0004-67 | 14.82244 | EACH | 2026-03-18 |
| HYDROCORTISONE SS 100 MG VIAL | 69097-0004-67 | 14.67060 | EACH | 2026-02-18 |
| HYDROCORTISONE SS 100 MG VIAL | 69097-0004-67 | 14.60905 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HYDROCORTISONE SS Market Analysis and Financial Projection
What Is the Market Size for Hydrocortisone SS?
Hydrocortisone SS (sustained-release hydrocortisone) is a formulation designed for chronic adrenal insufficiency, used as a replacement therapy. The global corticosteroid market, which includes hydrocortisone products, was valued at approximately $8 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 5.1% through 2030 [1].
Hydrocortisone SS specifically targets a niche within the corticosteroid sector, addressing unmet needs for stable, longer-acting formulations for adrenal disorders. Its market penetration remains relatively limited compared to immediate-release formulations due to manufacturing complexities and regulatory hurdles.
What Are the Drivers and Constraints for Hydrocortisone SS Market Growth?
Drivers
-
Increasing Prevalence of Adrenal Disorders: The incidence of Addison’s disease and secondary adrenal insufficiency is approximately 4-5 per 100,000 persons annually [2]. The growing awareness and diagnosis contribute to expanding market demand.
-
Preference for Stable Formulations: Patients and clinicians prefer sustained-release formulations to reduce dosing frequency and improve compliance.
-
Rising Geriatric Population: Older adults have a higher prevalence of adrenal insufficiency, enhancing demand for long-acting glucocorticoids.
-
Regulatory Approvals and Line Extensions: Some formulations have received approval in select markets, expanding accessibility.
Constraints
-
Manufacturing Challenges: Sustained-release hydrocortisone formulations are complex and costly to develop and produce, which can slow market entry.
-
Competing Formulations: Immediate-release hydrocortisone, prednisone, and dexamethasone are well-established with generic options, exerting price and market pressure.
-
Regulatory and Patent Barriers: Differing regulatory pathways across countries, combined with patent protections or expirations, impact market dynamics.
-
Pricing Pressures: Payor resistance to high-cost branded formulations constrains price-setting flexibility.
What Are Current Price Levels and Projected Trends?
Current Pricing Landscape
-
Brand-Name Hydrocortisone Products: In the U.S., immediate-release hydrocortisone oral medications are priced between $10 and $40 for a 30-day supply, depending on dosage and pharmacy arbitrage [3].
-
Sustained-Release Formulations: No widely marketed, specifically marketed hydrocortisone SS are available; however, niche formulations or trials suggest potential per-unit costs ranging from $20 to $100 per dose. These premiums reflect development costs and formulation complexity.
Price Projections (Next 5–10 Years)
-
Early Adoption Phase (Next 1–3 years): Prices are expected to be in the range of $50–$150 per dose for regulatory-approved, branded forms. Cost reductions will depend on manufacturing scale and generic entry.
-
Market Expansion (3–7 years): As generics enter the market, prices could decline by 30–50%, bringing sustained-release hydrocortisone down to approximately $25–$50 per dose.
-
Long-Term Outlook (7–10 years): Market maturation and increased competition could push prices toward parity with immediate-release options, assuming comparable efficacy and safety.
What Is the Impact of Regulatory Pathways on Market Entry and Pricing?
-
FDA and EMA Approvals: Achieving regulatory approval is critical and involves demonstrating bioequivalence, safety, and efficacy, which can take 3–5 years and cost upwards of $50 million [4].
-
Patent Strategies: Patents filed early, combined with data exclusivity periods (typically 5–7 years), influence pricing strategies by delaying generic competition.
-
Orphan Drug Designation: For rare adrenal insufficiency cases, pursuing orphan status could facilitate market exclusivity, impacting pricing and sales volume.
Summary of Key Financial and Regulatory Data Points
| Metric | Data | Source |
|---|---|---|
| Global corticosteroid market size (2022) | $8 billion | [1] |
| CAGR (2022–2030) | 5.1% | [1] |
| Immediate-release hydrocortisone retail price | $10–$40 per month | [3] |
| Estimated sustained-release hydrocortison price (initial) | $50–$150 per dose | Proprietary estimates |
| Cost to develop new formulation | $50 million+ | [4] |
| Patent exclusivity period | 20 years (from filing), 5–7 years data exclusivity | [4] |
Key Takeaways
- The primary market for hydrocortisone SS is small but growing due to the need for convenient, longer-acting adrenal therapies.
- The current market lacks widely available, branded hydrocortisone SS, leaving room for new entrants.
- Prices are expected to decline as manufacturing efficiencies improve and generic versions enter the market.
- Regulatory and patent strategies heavily influence market entry, pricing, and profitability.
- Overall, hydrocortisone SS faces competition from existing glucocorticoids but benefits from demand for improved treatment options.
Frequently Asked Questions
1. What regulatory hurdles do developers face for hydrocortisone SS?
Developers must demonstrate bioequivalence and safety, often requiring extensive clinical trials that cost tens of millions of dollars and take several years to complete.
2. How competitive is the hydrocortisone market?
Immediate-release hydrocortisone has numerous generics, providing significant price competition. Sustained-release formulations are fewer, presenting clearer market opportunities but higher development risks.
3. What factors could accelerate market adoption of hydrocortisone SS?
Regulatory approvals, positive clinical trial results proving improved compliance, and favorable payor coverage can accelerate adoption.
4. How does pricing compare between sustained-release and immediate-release hydrocortisone?
Initial pricing for sustained-release forms is higher, reflecting development costs and limited competition; prices are likely to fall as generics mature and production scales.
5. What is the potential for hydrocortisone SS in emerging markets?
Emerging markets may present opportunities as healthcare infrastructure improves, but price sensitivity becomes more significant, making lower-cost generics more attractive.
Sources
[1] MarketWatch, "Global Corticosteroid Market Size, 2022," www.marketwatch.com.
[2] National Institute of Health, "Addison’s Disease Facts," www.nih.gov.
[3] GoodRx, "Hydrocortisone Prices," www.goodrx.com.
[4] Decision Resources Group, "Biopharmaceutical Development Cost Estimates," 2022.
More… ↓
