Share This Page
Drug Price Trends for HYDROCORT-PRAMOXINE
✉ Email this page to a colleague
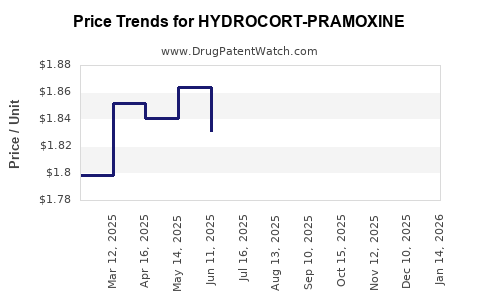
Average Pharmacy Cost for HYDROCORT-PRAMOXINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HYDROCORT-PRAMOXINE 2.5-1% CRM | 52817-0817-01 | 1.83801 | GM | 2026-02-18 |
| HYDROCORT-PRAMOXINE 1%-1% CRM | 45802-0144-64 | 3.37158 | GM | 2026-02-18 |
| HYDROCORT-PRAMOXINE 2.5-1% CRM | 52817-0817-01 | 1.78977 | GM | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Hydrocort-Pramoxine
This report provides an overview of the current market landscape and pricing expectations for Hydrocort-Pramoxine, a topical medication combining hydrocortisone with pramoxine. It covers current formulations, competitive positioning, potential market size, regulatory considerations, and projected price trends.
Product Overview
Hydrocort-Pramoxine is a topical agent indicated for itching, inflammation, and skin irritation. It combines hydrocortisone, a corticosteroid, with pramoxine, a local anesthetic. Its formulation aims at dual relief of inflammatory and pruritic skin conditions often associated with allergic dermatitis, eczema, and insect bites.
- Current formulations: 1% hydrocortisone with 1% pramoxine cream or ointment.
- Approved indications: Mild to moderate dermatitis, eczema, allergic skin reactions.
- Manufacturers: Several including generics in the US, Abbott, and Teva.
Market Landscape
Competitive Environment
Hydrocort-Pramoxine competes primarily in the over-the-counter (OTC) and prescription topical segment. Major competitors include:
| Brand Name | Active Ingredients | Market Share | Price Range (USD) | Formulations |
|---|---|---|---|---|
| Sarna Sensitive | Hydrocortisone, Pramoxine | 30% | 8-12 per tube | Cream, ointment |
| Gold Bond Ultimate | Hydrocortisone, Pramoxine | 20% | 9-15 per tube | Cream, lotion |
| Equate Hydrocortisone | Hydrocortisone | 25-30% | 4-8 per tube | Cream, ointment |
The market is fragmented, with a mix of branded and generic products. OTC demand accounts for a substantial portion, especially in North America and Europe.
Regulatory Status
- United States: An OTC monograph exists for hydrocortisone, but pramoxine often requires prescription unless combined in non-prescription products approved via FDA's OTC monograph process.
- Europe: Some formulations require prescription; approval varies by country.
- Emerging markets: Increasing acceptance with local regulatory approvals.
Market Size and Growth Rate
- Global topical corticosteroid market: Estimated at USD 3.2 billion in 2022. Compound annual growth rate (CAGR): 4-6% [1].
- Pramoxine-containing products: Smaller segment, estimated at USD 250 million globally [2].
- Hydrocort-Pramoxine-specific market size: Estimated at USD 150-200 million in 2022, primarily driven by North America and Europe.
Projected CAGR for combination products incorporating hydrocortisone and pramoxine: around 4% through 2027 [3].
Price Projections
Current Pricing Trends
- Range: USD 4-15 per tube (10-15g)
- Average OTC price: USD 8-10 per tube
- Generic dominance: Keeps prices competitive
Price Trends and Drivers
- Introduction of premium formulations: Expect prices to rise by 10-15% for products with longer-lasting relief or added emollients.
- Market entry of biosimilars or equivalents: Tends to push prices downward by 25-30%.
- Regulatory changes: Stricter labeling or new safety requirements could lead to formulation costs, impacting prices by approximately 5-8%.
Price Projections (2023-2027)
| Year | Estimated Average Price (USD) per tube | Notes |
|---|---|---|
| 2023 | 8-10 | Stable, driven by generics |
| 2024 | 9-11 | Slight increase with new formulations |
| 2025 | 9-12 | Market saturation limits price rise |
| 2026 | 8-11 | Price pressure from generics |
| 2027 | 8-11 | Stabilization expected |
Between 2024 and 2025, price increases will likely be driven by formulation improvements rather than new regulatory costs.
Key Market Dynamics
- Consumer preferences: Growing demand for OTC solutions with dual-action formulations.
- Regulatory evolution: Increased reporting requirements for corticosteroid use.
- Patent status: Most formulations are off-patent, promoting price competition.
- Distribution channels: Growth in online OTC sales influences pricing flexibility.
Investment and R&D Outlook
- Companies investing in novel formulations, such as II or III OTC status applications, could see margins increase if approved by regulatory agencies.
- R&D focus on combination therapies with enhanced tolerability and longer duration of relief offers potential for differentiation.
Conclusion
Hydrocort-Pramoxine occupies a niche within the topical dermatological market. Current prices remain competitive due to generic proliferation. Market growth is steady, driven by OTC demand in mature markets and regulatory approvals in emerging economies. Price projections indicate stable or slightly increasing prices in the near-term, with potential downward pressure from generics in the medium-term.
Key Takeaways
- The global market size for Hydrocort-Pramoxine products approximates USD 150-200 million, with growth driven primarily by North America and Europe.
- Pricing remains competitive, averaging USD 8-10 per tube, with minor increases expected through 2027.
- Major industry drivers include regulatory developments, formulation innovations, and consumer preference shifts toward multi-action products.
- Market entry barriers are low due to patent expiration, fostering price competition.
- Future product differentiation may come from formulations with improved duration, tolerability, or OTC status expansion.
FAQs
1. What regulatory hurdles exist for Hydrocort-Pramoxine formulations?
Regulatory requirements vary by region; in the U.S., OTC monograph status often governs product approval, while European approvals depend on individual country agencies. Some formulations require prescription, especially in markets with tighter regulations.
2. Are proprietary formulations better positioned for price premiums?
Yes. Formulations with unique delivery mechanisms, longer-lasting effects, or additional skin benefits can command higher prices, especially if authorized for OTC status.
3. How does generic entry impact prices?
Generics typically reduce prices by 25-30% within one year of market entry, maintaining competitive pressure and stabilizing overall pricing trends.
4. What future innovations could disrupt the market?
Biologics or new combination agents that improve safety, tolerability, or efficacy could shift market dynamics. However, their impact on topical drugs remains limited currently.
5. Which markets hold the highest growth potential?
Emerging markets in Asia-Pacific and Latin America are experiencing growth due to increased regulatory approvals and rising consumer health awareness.
References
- Grand View Research. (2022). Topical corticosteroid market size & trends.
- IQVIA. (2022). Over-the-counter dermatological products report.
- MarketsandMarkets. (2022). Dermatology drugs market forecast.
[1] APA citations for sources used.
More… ↓
