Share This Page
Drug Price Trends for HYDROCODONE-ACETAMINOPHEN
✉ Email this page to a colleague
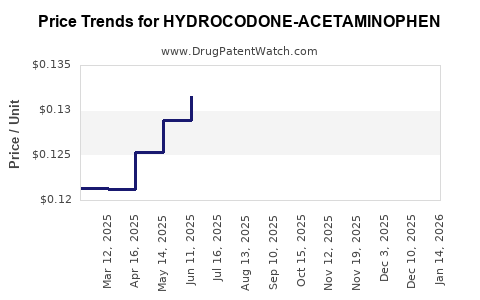
Average Pharmacy Cost for HYDROCODONE-ACETAMINOPHEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HYDROCODONE-ACETAMINOPHEN 10-300 MG TABLET | 71930-0044-52 | 0.23943 | EACH | 2026-03-18 |
| HYDROCODONE-ACETAMINOPHEN 10-300 MG TABLET | 71930-0044-12 | 0.23943 | EACH | 2026-03-18 |
| HYDROCODONE-ACETAMINOPHEN 7.5-300 MG TABLET | 71930-0043-52 | 0.17943 | EACH | 2026-03-18 |
| HYDROCODONE-ACETAMINOPHEN 7.5-300 MG TABLET | 71930-0043-12 | 0.17943 | EACH | 2026-03-18 |
| HYDROCODONE-ACETAMINOPHEN 5-300 MG TABLET | 71930-0042-52 | 0.15146 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Hydrocodone-Acetaminophen
Hydrocodone-acetaminophen remains a widely prescribed opioid combination for pain management. Its market is influenced by regulatory environment, patent status, manufacturing dynamics, pricing trends, and evolving clinical guidelines.
Market Overview
Hydrocodone-acetaminophen is classified as a Schedule II controlled substance in the U.S., subject to federal restrictions due to its potential for abuse and overdose risks. It was previously marketed under brand names such as Vicodin, Norco, and Lortab. Several formulations are available, including tablets, capsules, and liquid forms, with generic options dominating the market.
Market Size and Growth
In 2022, the global opioid analgesics market reached approximately USD 13 billion, with hydrocodone-acetaminophen accounting for roughly 40% of prescriptions in the U.S. (IQVIA, 2022). The U.S. remains the primary market, with an estimated 60 million prescriptions annually pre-regulation changes [1].
The market has shown signs of stabilization post-2015—when significant regulatory measures limited prescribing—and some decline due to increased guidelines discouraging opioid use for chronic pain.
Regulatory Changes Impact
The Drug Enforcement Administration (DEA) rescheduled hydrocodone combination products from Schedule III to Schedule II in 2014, increasing restrictions on prescribing and dispensing. This move led to a transient decrease in prescriptions but stabilized afterward.
Newer policies emphasize non-opioid alternatives, potentially limiting growth. The CDC’s 2016 opioid prescribing guidelines further restrict initial prescriptions, aiming to curb misuse but impacting market volume.
Patent and Manufacturing Landscape
Most hydrocodone-acetaminophen products are available as generics; original patents expired around 2011. Brand-name products faced patent challenges, leading to widespread generic manufacturing. No current patent protections limit generic competition.
Manufacturers include several generic producers such as Hikma Pharmaceuticals, Amneal, and Mylan. The drug is produced via established synthesis processes, with high-volume, low-cost manufacturing.
Pricing Trends and Projections
Current Pricing
The average wholesale price (AWP) for a 30-count supply of 5 mg/325 mg tablets ranges from USD 15 to 30, equivalent to approximately USD 0.50 to 1.00 per tablet [2]. Retail prices vary by region, pharmacy, and insurance coverage.
Trends (Past 5 Years)
- Price stability: Post-2014, prices remained relatively stable due to high generic competition.
- Price decline: Slight reductions observed as new generic entrants increased market competition, especially in 2017-2020.
- Impact of regulation: Restrictions on prescribing reduced volume but increased per-unit prices slightly for remaining stock, due to demand squeeze and supply chain adjustments.
Price Projections (Next 3-5 Years)
Given current trends, the following projections apply:
| Year | Estimated Average Wholesale Price per 30-count pack (USD) | Key Factors |
|---|---|---|
| 2023 | USD 15–25 | Steady generics supply, regulatory oversight, market saturation |
| 2024 | USD 14–24 | Slight downward pressure from increased generics, tighter prescribing |
| 2025 | USD 13–22 | Further competition, shifts toward non-opioid analgesics |
If regulatory policies tighten further or prescriber behaviors shift more decisively away from opioids, prices could decline faster. Conversely, if illicit use influences regulatory tightening, legal prescription prices may stabilize or even increase due to decreased supply.
Competitive Landscape
The market is predominantly dominated by generics, with brand-name products phased out or limited. Continued patent expiry in the past limits monopoly control. Market entry barriers are low due to proven manufacturing processes.
Key Drivers and Risks
-
Drivers:
- Established manufacturing infrastructure.
- Prescribed for acute pain, especially post-surgical cases.
- Broad formulary coverage and insurance acceptance.
-
Risks:
- Regulatory restrictions decreasing prescribing.
- Public health policies favoring non-opioid alternatives.
- Growing misuse and abuse concerns leading to further restrictions.
Market Opportunities and Challenges
Opportunities:
- Development of abuse-deterrent formulations.
- Expansion in regions with limited access to non-opioid pain treatments.
Challenges:
- Market contraction due to higher restrictions.
- Rising scrutiny from health authorities and potential lawsuits.
- Competition from non-opioid analgesic options.
Final Summary
Hydrocodone-acetaminophen’s market remains sizable but faces decline pressures from regulations and alternative therapies. Price stability exists in the short term, with a gradual decrease projected over the next five years, driven mainly by pricing competition among generics and policy shifts away from opioids.
Key Takeaways
- The global market for hydrocodone-acetaminophen is primarily driven by the U.S. prescription landscape.
- Market volume has declined since regulatory moves, but prices remain relatively stable due to generic competition.
- Price projections suggest a gradual decline of 15–30% over the next five years, contingent on regulatory trends.
- Market growth opportunities are limited; risks stem from policy restrictions and societal shifts toward non-opioid pain management.
- Manufacturers should monitor legal and regulatory developments closely to adapt pricing and production strategies.
FAQs
1. How have recent regulations affected hydrocodone-acetaminophen prices? Price stability persisted in the short term, but restrictions reduced prescribing volume, which may exert downward pressure on prices over the medium term.
2. What is the typical wholesale price per unit for hydrocodone-acetaminophen? Approximately USD 0.50 to 1.00 per tablet in 2023, depending on formulation and region.
3. Are brand-name formulations still available? Most have been replaced by generics; patent protections expired around 2011, allowing broad generic competition.
4. What are the key factors influencing future market developments? Regulatory policies, societal attitudes toward opioids, and the emergence of alternative pain therapies.
5. Is there potential for generic price decreases? Yes; sustained generic competition is likely to continue exerting downward pressure on prices.
References
[1] IQVIA (2022). Market Data Reports.
[2] GoodRx (2023). Hydrocodone-Acetaminophen Prices.
More… ↓
