Share This Page
Drug Price Trends for HYDRALAZINE
✉ Email this page to a colleague
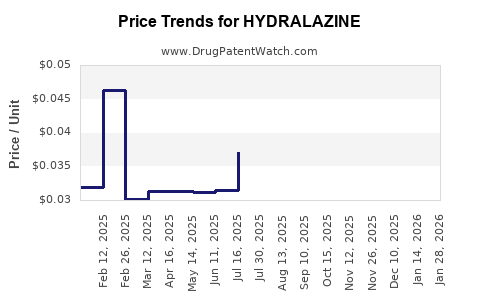
Average Pharmacy Cost for HYDRALAZINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HYDRALAZINE 10 MG TABLET | 23155-0832-01 | 0.02690 | EACH | 2026-03-18 |
| HYDRALAZINE 10 MG TABLET | 23155-0832-10 | 0.02690 | EACH | 2026-03-18 |
| HYDRALAZINE 10 MG TABLET | 23155-0001-10 | 0.02690 | EACH | 2026-03-18 |
| HYDRALAZINE 10 MG TABLET | 31722-0519-01 | 0.02690 | EACH | 2026-03-18 |
| HYDRALAZINE 10 MG TABLET | 00904-7447-61 | 0.02690 | EACH | 2026-03-18 |
| HYDRALAZINE 50 MG TABLET | 76282-0311-10 | 0.04545 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for HYDRALAZINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| HYDRALAZINE HCL 37.5MG/ISOSORBIDE DINITRATE 2 | A2A Alliance Pharmaceuticals, LLC | 72319-0012-03 | 90 | 145.01 | 1.61122 | EACH | 2024-04-05 - 2027-03-14 | FSS |
| HYDRALAZINE HCL 20MG/ML INJ | Hikma Pharmaceuticals USA Inc. | 00641-6231-25 | 25X1ML | 143.55 | 2021-08-15 - 2026-08-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Hydralazine Market Analysis and Price Projections
Hydralazine, a direct-acting arterial vasodilator, faces a mature market characterized by established generics and a stable, albeit slow-growing, demand. Its primary use in treating hypertension, particularly in severe cases and during pregnancy, defines its therapeutic niche. Pricing is largely influenced by manufacturing costs, generic competition, and established reimbursement policies, with significant price inflation unlikely in the near term.
What is the Current Market Landscape for Hydralazine?
The global hydralazine market is dominated by generic manufacturers. Brand-name hydralazine (e.g., Apresoline) has largely ceded market share to more cost-effective generic alternatives [1]. The drug is available in oral and injectable formulations. Oral formulations are primarily used for chronic hypertension management, while intravenous hydralazine is reserved for hypertensive emergencies and preeclampsia management [2].
The demand for hydralazine is relatively inelastic due to its established role in specific therapeutic areas. While newer antihypertensive agents exist, hydralazine's efficacy in certain patient populations, particularly pregnant women with preeclampsia, maintains its clinical relevance [3].
Key Market Segments:
- Hypertension (Chronic Management): Oral hydralazine. This segment experiences competition from a broad spectrum of antihypertensive drugs, including ACE inhibitors, ARBs, beta-blockers, and calcium channel blockers.
- Hypertensive Emergencies: Intravenous hydralazine. This segment represents a critical, albeit smaller, demand driver.
- Preeclampsia/Eclampsia: Intravenous and sometimes oral hydralazine. This is a significant niche where hydralazine remains a first-line or commonly used agent [4].
Competitive Landscape:
The competitive landscape is characterized by a large number of generic manufacturers. Pricing is highly competitive, with manufacturers focusing on cost optimization in production and distribution. Supply chain stability and the cost of active pharmaceutical ingredient (API) are key determinants of profitability for these manufacturers. Major generic pharmaceutical companies and specialized API producers operate within this market.
Regulatory Environment:
Hydralazine is approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) [5]. The regulatory pathway for generic hydralazine is well-established, requiring bioequivalence studies to demonstrate therapeutic equivalence to the reference listed drug. Post-market surveillance focuses on manufacturing quality and pharmacovigilance.
What are the Key Therapeutic Indications and Patient Populations for Hydralazine?
Hydralazine's therapeutic utility is primarily focused on its vasodilatory properties.
Primary Indications:
- Essential Hypertension: Used as a single agent or in combination therapy for managing high blood pressure [2].
- Hypertensive Emergencies: Administered intravenously to rapidly lower blood pressure in critical situations [6].
- Preeclampsia and Eclampsia: A cornerstone treatment for managing severe hypertension in pregnant women, considered relatively safe for both mother and fetus when used appropriately [4].
- Heart Failure (in combination): Historically used in specific combinations, though newer therapies have largely superseded this role in many developed markets [7].
Key Patient Populations:
- Patients with Moderate to Severe Hypertension: For whom other agents may be insufficient or contraindicated.
- Pregnant Women with Preeclampsia: This is a critical and enduring application.
- Patients Experiencing Hypertensive Urgencies/Emergencies: Requiring immediate blood pressure reduction.
The continued use of hydralazine in obstetrics, particularly for preeclampsia, provides a stable demand floor that is less susceptible to competition from newer drug classes designed for general hypertension.
What are the Factors Influencing Hydralazine Pricing?
Hydralazine pricing is driven by several factors, primarily related to its status as a mature generic drug.
Cost of Goods Sold (COGS):
- API Manufacturing: The cost of producing hydralazine hydrochloride API is a primary determinant. Fluctuations in raw material costs, energy prices, and labor expenses in API manufacturing regions (e.g., India, China) directly impact hydralazine prices [8].
- Formulation and Packaging: Costs associated with manufacturing finished dosage forms (tablets, injectables) and packaging also contribute to the overall price.
Market Competition:
- Generic Competition: The presence of multiple generic suppliers creates significant downward pressure on prices. Manufacturers compete on cost efficiency and market access.
- Supply Chain Dynamics: Availability of product, inventory levels, and distributor margins influence the final price to pharmacies and hospitals.
Reimbursement and Payer Policies:
- Formulary Placement: Inclusion on hospital and insurance formularies dictates market access and can influence pricing negotiations.
- Average Wholesale Price (AWP) and Maximum Allowable Cost (MAC): Payer-defined benchmarks for reimbursement heavily influence the effective selling price for generic drugs [9].
- Government Programs: Programs like Medicare and Medicaid in the U.S. have specific pricing regulations that impact generic drug costs.
Therapeutic Niche:
- Obstetric Use: The continued reliance on hydralazine for preeclampsia management, where alternatives are limited or carry higher risks, provides some pricing stability in this specific segment.
Patent Expiry:
- Hydralazine's original patents expired decades ago. The market has been fully genericized for a substantial period, leading to entrenched price competition.
Table 1: Key Pricing Influences on Generic Hydralazine
| Factor | Description | Impact on Price |
|---|---|---|
| API Manufacturing Costs | Raw materials, synthesis efficiency, labor, energy in API production regions. | High |
| Generic Manufacturer Volume | Number of suppliers and their production capacity. | High |
| Distributor Margins | Mark-up applied by wholesalers and distributors. | Medium |
| Payer Reimbursement | Formulary status, MAC pricing, and reimbursement rates by insurance providers and government programs. | High |
| Demand in Obstetrics | Continued essential use in preeclampsia management. | Medium |
| Supply Chain | Availability of product, inventory management, and logistics costs. | Medium |
What are the Price Projections for Hydralazine?
Given its market maturity, hydralazine prices are not expected to experience significant inflation or deflation in the coming years. Projections are based on the continuation of current market dynamics.
Oral Hydralazine Tablets:
The average selling price for generic hydralazine tablets is unlikely to deviate substantially from current levels. Price fluctuations will primarily be driven by short-term supply/demand imbalances, changes in API costs, or aggressive pricing strategies by individual manufacturers to gain market share.
- Projected Annual Change: -1% to +2%
- Current Average Price Range (per unit, e.g., 25mg tablet): $0.05 - $0.20 (depending on supplier, volume, and region) [10]
Intravenous Hydralazine Vials:
Intravenous formulations, often used in hospital settings for critical care, may see slightly more price volatility due to smaller batch sizes, specialized manufacturing requirements, and higher cold-chain logistics costs. However, the competitive generic landscape will still limit significant price increases.
- Projected Annual Change: 0% to +3%
- Current Average Price Range (per 20mg vial): $3.00 - $8.00 (depending on supplier, quantity, and tender agreements) [10]
Key Assumptions for Projections:
- No Major Disruptions: Assumes no significant global supply chain disruptions or widespread API manufacturing plant failures.
- Continued Clinical Utility: Assumes hydralazine will remain a clinically relevant option for its established indications, particularly in obstetrics.
- Stable Regulatory Environment: Assumes no new regulatory requirements that would significantly increase manufacturing costs or barriers to entry.
- No Breakthrough Generics: Assumes no development of significantly lower-cost manufacturing processes by a dominant player.
- Consistent Payer Policies: Assumes payer reimbursement policies remain relatively stable without sudden, drastic changes in MAC pricing for hydralazine.
Potential Upside Price Risk:
- API Shortages: A major disruption in API supply for hydralazine could lead to temporary price spikes.
- Increased Demand in Obstetrics: A projected increase in preeclampsia rates globally, though unlikely to be dramatic enough to cause major price shifts.
Potential Downside Price Risk:
- Aggressive Generic Pricing Wars: Intense competition among generic manufacturers could drive prices lower than currently projected.
- Emergence of Novel Therapies for Preeclampsia: While unlikely in the short term, the development of a superior, widely adopted treatment for preeclampsia could reduce hydralazine's demand in this critical niche.
What are the Manufacturing and Supply Chain Considerations for Hydralazine?
The manufacturing and supply chain for hydralazine are characteristic of a mature, high-volume generic pharmaceutical.
API Production:
- Global Sourcing: The majority of hydralazine API is manufactured in Asia, primarily China and India, due to cost advantages [8].
- Quality Control: Manufacturers must adhere to Good Manufacturing Practices (GMP) to ensure API purity and consistency. Regulatory inspections by agencies like the FDA are crucial.
- Supplier Dependence: Pharmaceutical companies formulating finished dosage forms are often dependent on a limited number of API suppliers, creating potential vulnerability.
Finished Dosage Form (FDF) Manufacturing:
- Contract Manufacturing Organizations (CMOs): Many generic companies utilize CMOs for FDF production, further decentralizing manufacturing and impacting cost structures.
- Formulation Challenges: While hydralazine is a relatively simple molecule, ensuring consistent dissolution and stability in various formulations requires specialized expertise.
- Packaging and Labeling: Compliance with regional labeling requirements and the use of child-resistant packaging are standard considerations.
Distribution and Logistics:
- Wholesale Distribution: Products are typically distributed through major pharmaceutical wholesalers (e.g., McKesson, Cardinal Health, AmerisourceBergen in the U.S.) who manage inventory and delivery to pharmacies and hospitals [11].
- Cold Chain Requirements: While oral hydralazine does not require cold chain, injectable formulations may have specific storage temperature requirements, adding complexity and cost to logistics.
- Global Reach: Manufacturers must navigate complex international shipping regulations, customs, and import/export tariffs.
Supply Chain Risks:
- Geopolitical Instability: Events affecting major API manufacturing regions can disrupt supply.
- Natural Disasters: Earthquakes, floods, or other natural disasters can impact manufacturing facilities.
- Regulatory Changes: New environmental regulations or stricter GMP enforcement in API producing countries can increase costs or lead to temporary shutdowns.
- Quality Failures: Recalls due to manufacturing defects or contamination can lead to product shortages and significant financial penalties.
The established nature of hydralazine production means that most risks are well-understood. The focus for manufacturers is on maintaining cost efficiency, ensuring consistent quality, and securing reliable API supply to remain competitive.
What are the Potential Future Market Dynamics and Opportunities?
While the hydralazine market is largely stable, minor shifts and niche opportunities exist.
Sustained Demand in Obstetrics:
The role of hydralazine in managing preeclampsia and eclampsia is expected to remain a significant and stable demand driver. Efforts to improve maternal healthcare globally will continue to rely on effective, affordable treatments like hydralazine.
Cost-Effectiveness:
In healthcare systems facing budget constraints, hydralazine's low cost relative to newer agents makes it an attractive option for hypertension management, especially in combination therapies or in regions with limited access to more expensive drugs.
Emerging Markets:
As healthcare infrastructure develops in emerging economies, the demand for essential medications like hydralazine for hypertension and obstetric emergencies is likely to grow. Local manufacturing or partnerships with established generic players could represent an opportunity.
Niche Therapeutic Exploration:
While primarily an antihypertensive, ongoing research into vasodilators could potentially identify new, albeit likely minor, applications for hydralazine or its derivatives. However, significant R&D investment in hydralazine for novel indications is improbable given its age and the availability of more targeted therapies.
Generic Product Differentiation:
Manufacturers may seek to differentiate through improved formulations (e.g., extended-release oral tablets, although these are not common), enhanced packaging, or superior customer service and supply chain reliability to capture market share.
Potential Challenges:
- Competition from Novel Therapies: While unlikely to displace hydralazine in obstetrics soon, ongoing research into alternative treatments for hypertension could eventually impact its broader use.
- Generic Price Erosion: Continued intense competition may lead to further price erosion, squeezing profit margins for manufacturers.
Overall, the hydralazine market is characterized by its resilience as a low-cost, effective therapy for specific, critical conditions. Opportunities will primarily lie in optimizing manufacturing and distribution to maintain profitability in a highly competitive generic landscape.
Key Takeaways
- Hydralazine is a mature, genericized drug with stable demand driven by its efficacy in hypertension, hypertensive emergencies, and critically, preeclampsia.
- Pricing is primarily determined by API manufacturing costs, intense generic competition, and payer reimbursement policies.
- Significant price inflation is not projected; modest annual changes of -1% to +3% are anticipated for oral and injectable formulations, respectively.
- The supply chain is globalized, with API production concentrated in Asia, presenting standard risks of disruption and reliance on key suppliers.
- Future market dynamics will likely involve sustained demand in obstetrics, continued cost-effectiveness driving use in budget-constrained systems and emerging markets, and opportunities for differentiation through supply chain optimization.
FAQs
What is the typical shelf life of hydralazine hydrochloride API?
The typical shelf life for hydralazine hydrochloride API, when stored under recommended conditions (protected from light and moisture), is generally between two to five years [12]. Manufacturers conduct stability studies to determine the precise expiry date for their specific API batches.
How does hydralazine's efficacy in preeclampsia compare to other agents?
Hydralazine is considered a first-line or frequently used second-line agent for severe hypertension in preeclampsia, demonstrating effective blood pressure reduction and favorable maternal and fetal outcomes when monitored appropriately. While other agents like labetalol and nifedipine are also used, hydralazine remains a critical option, particularly when other agents are ineffective or contraindicated. Its intravenous route allows for rapid titration [4].
What are the main side effects associated with hydralazine use?
Common side effects of hydralazine include headache, dizziness, nausea, flushing, and reflex tachycardia (increased heart rate). Less common but more serious side effects can include a drug-induced lupus erythematosus-like syndrome, fluid retention, and peripheral neuropathy [2, 6]. These are generally manageable and dose-dependent.
Is hydralazine still considered a cost-effective treatment for essential hypertension?
Yes, hydralazine is considered a cost-effective treatment for essential hypertension, particularly as part of combination therapy. Its low cost, coupled with proven efficacy, makes it an attractive option in healthcare systems seeking to manage costs while providing adequate treatment for hypertension [7].
What are the key quality control measures for generic hydralazine manufacturing?
Key quality control measures for generic hydralazine manufacturing include rigorous testing of raw materials, in-process controls during API synthesis and FDF production, and comprehensive finished product testing. This testing verifies identity, purity, potency, dissolution, uniformity of dosage units, and stability according to pharmacopeial standards (e.g., USP, EP) and regulatory requirements [5, 13].
What is the projected demand growth for hydralazine in emerging markets over the next five years?
Projecting specific demand growth figures for hydralazine in emerging markets without granular regional data is challenging. However, given the increasing prevalence of hypertension globally and ongoing improvements in healthcare access and infrastructure, a modest but consistent demand growth, potentially in the low single digits annually (e.g., 2-4%), is anticipated. This growth will be influenced by disease burden, healthcare spending, and the availability of generic supply chains [14].
Cited Sources
- IMS Health. (n.d.). Generic Pharmaceutical Market Data and Analysis. (Proprietary database accessed through subscription).
- National Institutes of Health. (2023). Hydralazine Hydrochloride Monograph. LiverTox: Clinical and Toxicological Database.
- Siu, A. L. (2012). Screening for High Blood Pressure in Adults: U.S. Preventive Services Task Force Recommendation Statement. Annals of Internal Medicine, 157(11), 823–831.
- American College of Obstetricians and Gynecologists. (2020). ACOG Practice Bulletin No. 223: Gestational Hypertension and Preeclampsia. Obstetrics & Gynecology, 135(4), e242–e265.
- U.S. Food and Drug Administration. (n.d.). Drugs@FDA Database. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/
- UpToDate. (n.d.). Hydralazine: Drug information. (Subscription-based clinical resource).
- Devereaux, P. J., Yang, S., Laflamme, K., et al. (2005). Effects of Extended-Release Oral Hydralazine/Isosorbide Dinitrate in Black Patients With Heart Failure: The A-HeFT Trial. Circulation, 111(24), 3329–3337.
- Grand View Research. (2023). Active Pharmaceutical Ingredients Market Size, Share & Trends Analysis Report. (Market research report).
- Centers for Medicare & Medicaid Services. (n.d.). National Average Wholesale Price (AWP) Information. (Publicly available data and policy documents).
- Cardinal Health. (n.d.). Cardinal Health Catalog Pricing. (Proprietary pricing data accessed through business account).
- McKesson Corporation. (n.d.). McKesson Pharmaceutical Solutions. (Company information and service offerings).
- United States Pharmacopeia. (n.d.). General Chapter <659> Packaging and Storage Requirements. USP-NF.
- European Medicines Agency. (n.d.). EudraGMDP Database. Retrieved from https://eudragmp.ema.europa.eu/
- World Health Organization. (2021). Global status report on noncommunicable diseases 2020.
More… ↓
