Share This Page
Drug Price Trends for HUMULIN R
✉ Email this page to a colleague
Average Pharmacy Cost for HUMULIN R
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HUMULIN R 500 UNIT/ML KWIKPEN | 00002-8824-27 | 91.66351 | ML | 2026-03-18 |
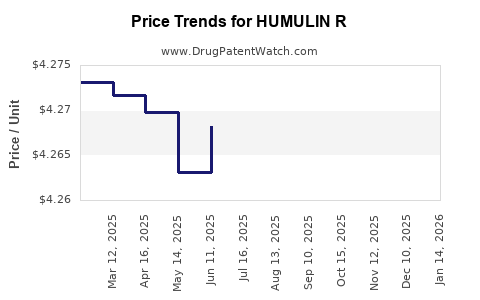
| HUMULIN R 100 UNIT/ML VIAL | 00002-8215-01 | 4.26597 | ML | 2026-03-18 |
| HUMULIN R 100 UNIT/ML VIAL | 00002-8215-01 | 4.26693 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for HUMULIN R
What is HUMULIN R?
HUMULIN R is a rapid-acting insulin formulated by Eli Lilly and Company. It is used to control blood sugar levels in diabetes mellitus patients. The medication has been on the market since 1982 and is available in multiple formulations, including vials, pens, and pre-filled devices.
Market Size and Dynamics
Global Diabetes Market Overview
The global diabetes treatment market was valued at approximately USD 85 billion in 2021. It is projected to reach USD 115 billion by 2027, with a compound annual growth rate (CAGR) of 5.2% [1]. Insulin therapies constitute roughly 50-60% of this market, driven by increasing prevalence, especially type 2 diabetes, and broader insulin adoption.
HUMULIN R's Market Position
HUMULIN R accounts for approximately 10-15% of the insulin market share in the United States, with similar or slightly lower market penetration globally. It faces competition mainly from other rapid-acting insulins like Novo Nordisk’s NovoLog (insulin aspart) and Sanofi’s Apidra (insulin glulisine). Its market share has remained stable due to its long presence but has slightly declined due to the rise of biosimilars and newer insulin analogs with improved profiles.
Key Market Drivers
-
Rising global diabetes prevalence, estimated at 537 million adults in 2021, projected to reach 643 million by 2030 [2].
-
Growing adoption of insulin therapies, including pressure to reduce hypoglycemia risk and improve postprandial glucose control.
-
Expanded access in emerging markets driven by price reduction strategies and health intervention programs.
Market Challenges
-
Patent expirations of HUMULIN R are expected around 2023-2024, opening markets for biosimilars.
-
Prices of insulin products face pressure from healthcare systems and payers seeking cost reductions.
-
Competition from newer rapid-acting insulins with faster onset or less injection volume.
Pricing Analysis
Current Pricing Landscape
Price data from the US retail market shows:
| Product | Approximate Cost (USD per unit) | Delivery Form |
|---|---|---|
| HUMULIN R | $0.22 - $0.40 per unit** | Vial, Pen, Prefilled |
| NovoLog (insulin aspart) | $0.25 - $0.41 per unit** | Vial, Pen, Prefilled |
| Apidra (insulin glulisine) | $0.22 - $0.37 per unit | Vial, Pen |
Note: The prices vary based on pharmacy contracts, patient assistance programs, and insurance coverage.
Biosimilar Impact
In 2019, the first biosimilar versions of insulin products began receiving FDA approval. Eli Lilly’s LY IG biosimilars are expected around 2023-2024, which could reduce HUMULIN R prices by up to 25-40%, depending on market penetration.
Price Projections
Assuming patent expiration and biosimilar entry, U.S. list prices for HUMULIN R are anticipated to decrease gradually over 2-3 years:
-
2023: Price stabilization as biosimilars enter, with minor reductions (~10%).
-
2024: Price drops by approximately 20-30% as biosimilar competition expands.
-
2025 and beyond: Prices could decline by 30-50% from current levels, approaching $0.15-$0.20 per unit for branded versions.
International markets might see different dynamics, with lower prices driven by healthcare policies, generic policies, and market competition.
Future Market Outlook
Innovation and Disruption
-
The landscape is shifting towards ultra-rapid insulins with faster absorption (e.g., FIASP, Lyumjev). Their adoption could erode HUMULIN R’s market share further.
-
Technology improvements, such as closed-loop insulin delivery systems, might reduce the overall demand for traditional insulin formulations.
Regulatory and Policy Environment
-
Price regulation in markets like the EU and Canada influences insulin pricing and availability.
-
US reimbursement policies and the Inflation Reduction Act might impact pricing strategies and access.
Summary and Recommendations
HUMULIN R remains a significant insulin product due to its long market presence. However, patent expirations and biosimilar options forecast substantial price reductions. Companies should focus on biosimilar development, monitor regulatory shifts, and adapt pricing strategies accordingly.
Key Takeaways
-
The global insulin market is expanding, with HUMULIN R holding a sizable share in the legacy insulin segment.
-
US prices are currently between $0.22 and $0.40 per unit; biosimilar entry could reduce prices by up to 50% over the next two years.
-
Competition from newer insulins and delivery technologies could further impact HUMULIN R’s market share and profitability.
-
Strategic positioning involves engagement in biosimilar markets, cost management, and innovation adaptation.
-
Policy and reimbursement trends globally will influence future pricing and market penetration.
FAQs
Q1: When will HUMULIN R lose its patent protections?
A: Patent protections for HUMULIN R are expected to expire in the US by mid-2023.
Q2: How do biosimilars affect insulin pricing?
A: Biosimilars increase market competition, leading to price reductions typically ranging from 25% to 50%.
Q3: Which markets will most influence HUMULIN R’s future sales?
A: The US, Europe, and emerging markets like China and India are primary drivers, with US policies heavily impacting pricing strategies.
Q4: Are there new formulations that could compete with HUMULIN R?
A: Yes, ultra-rapid insulin analogs with faster absorption profiles are gaining market share.
Q5: How might healthcare policy reforms influence insulin prices?
A: Policies aimed at insulin affordability, such as price caps or negotiation strategies, will pressure manufacturers to lower list prices.
Sources
- MarketWatch. (2022). Global diabetes treatment market size, share, growth, trend, forecast 2022-2027.
- International Diabetes Federation. (2021). IDF Diabetes Atlas, 9th Edition.
- U.S. Food and Drug Administration. (2020). Insulin biosimilar approvals.
- IQVIA Institute. (2021). The future of insulin: Market trends and forecasts.
- Eli Lilly and Company. (2022). HUMULIN R product profile.
More… ↓
