Share This Page
Drug Price Trends for HORIZANT ER
✉ Email this page to a colleague
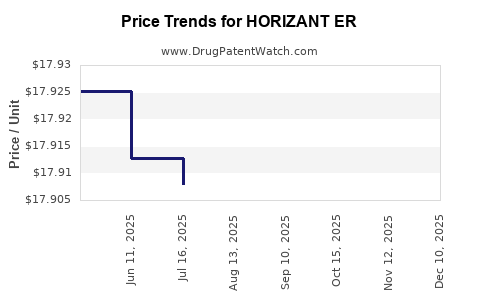
Average Pharmacy Cost for HORIZANT ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HORIZANT ER 600 MG TABLET | 53451-0101-01 | 17.91396 | EACH | 2026-05-20 |
| HORIZANT ER 300 MG TABLET | 53451-0103-01 | 17.92144 | EACH | 2026-05-20 |
| HORIZANT ER 300 MG TABLET | 53451-0103-01 | 17.93023 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HORIZANT ER (Gabapentin Enacarbil) Market Analysis and Price Projections
HORIZANT ER (gabapentin enacarbil) is a prodrug of gabapentin used for the treatment of restless legs syndrome (RLS) and postherpetic neuralgia (PHN). This analysis examines the current market landscape, competitive environment, and projected pricing trends for HORIZANT ER.
What is the current market size and growth trajectory for HORIZANT ER?
The market for RLS and PHN treatments is influenced by aging demographics, increasing disease prevalence, and advancements in therapeutic options. HORIZANT ER competes within this space, facing both branded and generic alternatives.
Restless Legs Syndrome (RLS) Market:
- The prevalence of RLS increases with age, affecting an estimated 5% to 10% of the adult population in developed countries.
- Diagnosis rates and patient awareness have grown, leading to increased demand for effective treatments.
- The RLS market is valued at approximately $1.5 billion globally, with a projected compound annual growth rate (CAGR) of 3% to 5% over the next five years.
Postherpetic Neuralgia (PHN) Market:
- PHN is a chronic neuropathic pain condition that can persist for months or years after a shingles outbreak.
- Its incidence is linked to the prevalence of herpes zoster (shingles), which affects an estimated 1 million people annually in the U.S.
- The PHN market is estimated at $2 billion globally, with a projected CAGR of 4% to 6%, driven by the growing elderly population and improved diagnostic capabilities.
HORIZANT ER Market Position:
- HORIZANT ER's unique pharmacokinetic profile, allowing for once-daily dosing, positions it as a convenient option for RLS patients.
- For PHN, its efficacy in reducing pain is a key driver of utilization.
- While specific market share data for HORIZANT ER is proprietary, it is a significant player in both therapeutic areas.
Who are HORIZANT ER's primary competitors?
HORIZANT ER faces competition from a range of therapies, including other extended-release formulations, immediate-release drugs, and non-pharmacological interventions.
Direct Competitors (RLS):
- Immediate-Release Gabapentin: Generic availability of immediate-release gabapentin is a significant competitor, offering a lower cost alternative.
- Pramipexole (Mirapex, Mirapex ER): A dopamine agonist that is a first-line treatment for moderate to severe RLS.
- Ropinirole (Requip, Requip XL): Another dopamine agonist with similar efficacy to pramipexole.
- Rotigotine (Neupro): A transdermal patch formulation, offering convenience and consistent drug delivery.
Direct Competitors (PHN):
- Pregabalin (Lyrica, generics): A widely prescribed anticonvulsant for neuropathic pain, including PHN. Lyrica's patent expiry has led to significant generic competition.
- Gabapentin (Neurontin, generics): Immediate-release gabapentin is also a common treatment for PHN, with widespread generic availability.
- Duloxetine (Cymbalta, generics): A serotonin-norepinephrine reuptake inhibitor (SNRI) approved for PHN.
- Amitriptyline: A tricyclic antidepressant (TCA) often used off-label for neuropathic pain.
Indirect Competitors:
- Opioids: While not first-line, opioids are sometimes used for severe, refractory pain, though their use is increasingly scrutinized due to addiction concerns.
- Non-pharmacological therapies: Cognitive behavioral therapy (CBT), exercise, and lifestyle modifications can complement pharmacological treatments.
Competitive Landscape Summary:
| Drug Class | Key Agents | Indication Focus | Generic Availability | HORIZANT ER Advantage |
|---|---|---|---|---|
| Gabapentin Prodrug | HORIZANT ER | RLS, PHN | No | Once-daily dosing, consistent plasma levels |
| Dopamine Agonists | Pramipexole, Ropinirole, Rotigotine | RLS | Yes (Pramipexole, Ropinirole) | Once-daily dosing (vs. multi-daily for IR) |
| Anticonvulsants | Pregabalin, Gabapentin (IR) | PHN, RLS (Gabapentin) | Yes | Convenience of once-daily dosing, potentially fewer side effects than IR gabapentin |
| SNRIs | Duloxetine | PHN | Yes | Different mechanism of action for broader efficacy |
What are the patent protections and exclusivity for HORIZANT ER?
The patent landscape for HORIZANT ER is critical in determining its market exclusivity and potential for generic entry. As of the latest available data, key patents are nearing expiration or have expired.
Key Patents and Exclusivity Information:
- U.S. Patent No. 7,687,047: Covers gabapentin enacarbil and its use in treating RLS. This patent was originally set to expire in 2027.
- U.S. Patent No. 8,003,631: Related to methods of treating RLS with gabapentin enacarbil.
- U.S. Patent No. 9,050,364: Related to pharmaceutical compositions of gabapentin enacarbil.
- U.S. Patent No. 9,782,431: Related to methods of treating PHN.
Patent Expirations and Generic Entry:
- While the primary patents were slated to expire around 2027, litigation and patent challenges can alter these timelines. Specific details of ongoing litigation are not publicly disclosed in a way that allows for definitive projection.
- The U.S. Food and Drug Administration (FDA) Orange Book lists patents and exclusivities associated with approved drug products. As of recent records, some foundational patents for gabapentin enacarbil have faced challenges.
- The expiration or invalidation of key patents would open the door for generic manufacturers to seek FDA approval for their own versions of gabapentin enacarbil. This would significantly impact HORIZANT ER's market share and pricing.
Regulatory Exclusivities:
- New Chemical Entity (NCE) Exclusivity: Gabapentin enacarbil, as a novel prodrug, was eligible for 5-year NCE exclusivity upon its initial approval. This period has long since passed.
- Orphan Drug Exclusivity: HORIZANT ER does not appear to have been granted orphan drug designation for RLS or PHN, which would provide an additional 7 years of market exclusivity.
The timing of generic entry is a critical variable in future price projections. A successful challenge to remaining patents or natural expiration will trigger increased competition.
What are the current pricing and reimbursement strategies for HORIZANT ER?
The pricing of HORIZANT ER is influenced by its branded status, the competitive landscape, and formulary placement by payers.
Current Pricing:
- Average Wholesale Price (AWP): As of Q4 2023, the AWP for a 30-day supply of HORIZANT ER (e.g., 600 mg tablets) typically ranges from $350 to $450.
- Net Price: Actual net prices after rebates and discounts negotiated with payers are substantially lower, though specific figures are confidential.
- Patient Out-of-Pocket Costs: These vary widely based on insurance coverage, copayments, and deductibles, often ranging from $20 to $100 per month for commercially insured patients.
Reimbursement Landscape:
- Formulary Placement: HORIZANT ER generally has favorable formulary access on many commercial insurance plans, often classified as a Tier 2 or Tier 3 preferred brand.
- Medicare Part D: It is covered under Medicare Part D, with standard copayment structures.
- Medicaid: Coverage varies by state, but it is typically available with state-specific restrictions or preferred drug lists.
- Prior Authorization: Some payers may require prior authorization, particularly for new starts, to ensure appropriate use and cost-effectiveness. This is more common when generic alternatives are available for the same indication.
- Step Therapy: In some instances, payers may implement step-therapy protocols requiring patients to try lower-cost alternatives (e.g., immediate-release gabapentin or dopamine agonists for RLS) before approving HORIZANT ER.
Rebates and Discounts:
- The net price of HORIZANT ER is significantly reduced through rebates offered to pharmacy benefit managers (PBMs) and health plans. These rebates are a standard practice in the pharmaceutical industry to secure favorable market access.
What are the projected price trends for HORIZANT ER post-patent expiration?
The expiration or invalidation of key patents for HORIZANT ER will fundamentally alter its pricing dynamic, leading to a significant decrease in price due to generic competition.
Projected Price Erosion Scenarios:
-
Scenario 1: Near-Term Generic Entry (within 1-2 years):
- Upon the first generic entry, HORIZANT ER's price is projected to decrease by 40% to 60% within the first year.
- By the second year, with multiple generic entrants, price erosion could reach 60% to 80% of the pre-generic AWP.
- This scenario assumes the successful approval of one or more generic gabapentin enacarbil products.
-
Scenario 2: Delayed Generic Entry (3-5 years or longer):
- If patent challenges are successful in delaying generic entry, HORIZANT ER could maintain its current pricing for a longer period.
- However, even in this scenario, incremental price increases might be limited by payer pressure and the eventual certainty of generic competition.
-
Scenario 3: Continued Market Exclusivity (unlikely without new patents):
- Without new patent filings or significant therapeutic advancements that create new market exclusivities, sustained high pricing post-original patent expiry is not anticipated.
Factors Influencing Price Post-Expiration:
- Number of Generic Entrants: A higher number of generic competitors generally leads to faster and deeper price reductions.
- Payer Strategies: Payers will likely favor generic gabapentin enacarbil due to cost savings, potentially imposing stricter utilization management on the branded product.
- Manufacturer Response: The brand manufacturer may employ authorized generic strategies or engage in lifecycle management to mitigate revenue loss, though the effectiveness of these is limited against true generics.
- Market Size and Demand: The established demand for RLS and PHN treatments provides a baseline for generic uptake.
Pricing Outlook:
The prevailing trend for branded drugs facing generic competition is substantial price decline. For HORIZANT ER, the peak pricing period is likely concluding or has concluded with the advent of significant patent challenges. Expect a sharp decline in both AWP and net price once generic alternatives are available. The net price for branded HORIZANT ER, if it remains on the market, will likely be significantly higher than generics but below current levels due to competitive pressures.
What are the key drivers and barriers impacting HORIZANT ER's future market performance?
Several factors will shape the trajectory of HORIZANT ER, with patent expiration being the most significant disrupter.
Key Drivers:
- Aging Population: The increasing prevalence of RLS and PHN in older adults will continue to drive demand for effective treatments.
- Improved Diagnosis and Awareness: Greater physician and patient awareness of RLS and PHN symptoms leads to more diagnoses and prescriptions.
- Convenience of Once-Daily Dosing: For RLS patients, the once-daily formulation of HORIZANT ER remains an attractive feature compared to multi-daily immediate-release options.
- Established Efficacy: HORIZANT ER has demonstrated efficacy in its approved indications, providing a foundation for continued use by patients and physicians.
- Potential for New Indications (Unlikely): While new drug applications can revitalize a product, there is no current indication of HORIZANT ER pursuing new indications.
Key Barriers:
- Impending Generic Competition: The most significant barrier is the anticipated entry of generic gabapentin enacarbil following patent expiration. This will lead to severe price erosion and market share loss.
- Competition from Established Generics: Generic gabapentin and pregabalin, along with branded alternatives like pramipexole and ropinirole, offer cost-effective or alternative treatment options.
- Payer Pressure and Formulary Restrictions: Payers will increasingly favor lower-cost generics, potentially restricting access to branded HORIZANT ER through prior authorization or step-therapy requirements.
- Cost-Effectiveness Scrutiny: As healthcare costs rise, payers and providers will scrutinize the cost-effectiveness of branded drugs over generics, especially for chronic conditions.
- Physician Prescription Habits: While efficacy is a driver, physicians may default to prescribing generics once available to manage patient costs and reduce administrative burden.
The future market performance of HORIZANT ER will be overwhelmingly dictated by the timing and impact of generic competition. Without new patent protection or significant market advantages, its revenue stream is projected to decline sharply post-patent expiration.
Key Takeaways
- HORIZANT ER (gabapentin enacarbil) targets the RLS and PHN markets, valued at approximately $1.5 billion and $2 billion respectively.
- Key competitors include immediate-release gabapentin, dopamine agonists (pramipexole, ropinirole), and pregabalin.
- Significant patent protection for HORIZANT ER is nearing expiration, with U.S. Patent No. 7,687,047 originally set to expire in 2027, though litigation could alter this timeline.
- Current pricing for a 30-day supply (600 mg) ranges from $350 to $450 AWP, with net prices significantly lower after rebates.
- Post-patent expiration, HORIZANT ER's price is projected to fall by 60% to 80% within two years of generic entry, depending on the number of competitors.
- The primary driver of future market performance is the imminent threat of generic competition, which will lead to substantial price erosion and market share loss.
Frequently Asked Questions
-
When is the earliest generic gabapentin enacarbil could be available in the U.S. market? The earliest availability depends on patent litigation outcomes. While some patents were projected to expire in 2027, successful legal challenges could accelerate this timeline. Manufacturers typically launch generics as soon as patent and regulatory hurdles are cleared.
-
What is the typical market share loss for a branded drug once its primary patents expire and generics enter? Branded drugs typically experience a market share loss of 70% to 90% within two years of generic entry, with prices falling to 10% to 30% of their pre-generic levels.
-
Will HORIZANT ER be able to maintain any market presence after generic entry? The branded product may retain a small market share by targeting specific patient populations willing to pay a premium for brand recognition or by being listed on formularies at a premium. However, its volume and pricing power will be severely diminished.
-
Are there any known off-label uses of HORIZANT ER that could sustain demand independently of its approved indications? Gabapentin enacarbil's prodrug nature is designed for optimized gabapentin delivery. While gabapentin itself has numerous off-label uses, HORIZANT ER is primarily prescribed for its approved indications due to its specific formulation and cost structure. There are no widely recognized significant off-label uses that would independently drive substantial market demand.
-
What is the impact of current U.S. healthcare policy or proposed legislation on the pricing of drugs like HORIZANT ER, especially in the context of patent expirations? Policies aimed at drug price negotiation (e.g., Medicare negotiation under the Inflation Reduction Act) and measures to expedite generic approvals can accelerate price declines for branded drugs approaching patent cliffs. These policies generally favor lower prices for consumers and payers.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/informationondrugs/ucm129672.htm [2] Various Pharmaceutical Market Research Reports (Confidential Data). (2023-2024). Global Neuropathic Pain and RLS Market Analysis. [3] U.S. Patent and Trademark Office. (n.d.). Patent Database Search. Retrieved from https://www.uspto.gov/patents/search [4] IQVIA Institute for Human Data Science. (2023). The Pharmaceutical Market: Global Outlook. [5] National Institutes of Health. (n.d.). Restless Legs Syndrome Fact Sheet. Retrieved from https://www.ninds.nih.gov/Disorders/All-Disorders/Restless-Legs-Syndrome-Information-Page
More… ↓
