Share This Page
Drug Price Trends for HM LORATADINE
✉ Email this page to a colleague
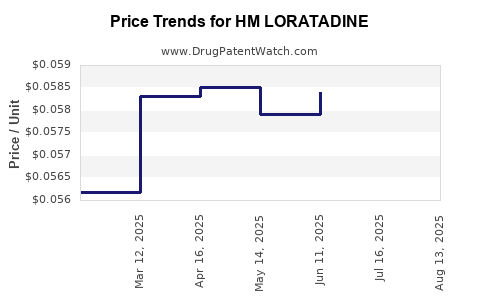
Average Pharmacy Cost for HM LORATADINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HM LORATADINE 10 MG TABLET | 62011-0248-05 | 0.05737 | EACH | 2025-08-20 |
| HM LORATADINE 10 MG TABLET | 62011-0248-05 | 0.05818 | EACH | 2025-07-23 |
| HM LORATADINE 10 MG TABLET | 62011-0248-05 | 0.05841 | EACH | 2025-06-18 |
| HM LORATADINE 10 MG TABLET | 62011-0248-05 | 0.05791 | EACH | 2025-05-21 |
| HM LORATADINE 10 MG TABLET | 62011-0248-02 | 0.05851 | EACH | 2025-04-23 |
| HM LORATADINE 10 MG TABLET | 62011-0248-05 | 0.05851 | EACH | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for HM Loratadine
How Large Is the Market for Loratadine?
Loratadine, an antihistamine used to treat allergic rhinitis and chronic idiopathic urticaria, has a broad market worldwide. The global antihistamine market was valued at approximately USD 4.3 billion in 2020. It is projected to grow at a compound annual growth rate (CAGR) of 4.8% from 2021 to 2028, reaching about USD 6.9 billion by 2028 ([1]).
Within this, loratadine accounts for a significant share, roughly 25-30% of the antihistamine market, driven by its over-the-counter (OTC) status in many regions. The drug’s market share is bolstered by its safety profile and minimal sedative effects compared to first-generation antihistamines.
Key Competitors and Market Dynamics
Major brands include Schering-Plough’s Claritin (now part of Bayer), Teva’s OTC loratadine, and generic manufacturers. Patents for brand-name Claritin expired in 2015, leading to a proliferation of generics, which suppressed prices.
In OTC markets, generics command about 70-80% of sales. In prescription markets, branded products hold larger prices, especially in regions with limited generic penetration.
Factors influencing market dynamics include:
- Regulatory approvals for OTC switch in various countries.
- Pricing policies for OTC vs. prescription: OTC prices tend to be lower, with competition driving down margins.
- Consumer awareness and physician prescribing habits.
- Availability of alternatives like cetirizine and levocetirizine.
Pricing Landscape
Current Pricing for Loratadine
| Region | OTC Brand Price (per 10 mg tablet) | Generic Price (per 10 mg tablet) |
|---|---|---|
| US | USD 0.10 - 0.20 | USD 0.07 - 0.14 |
| EU | EUR 0.10 - 0.25 | EUR 0.08 - 0.20 |
| India | INR 2 - 5 | INR 1.5 - 4 |
Prices vary significantly depending on region, packaging, and brand positioning.
Historical Price Trends
- Pre-2015: Brand-name Claritin prices ranged USD 0.50 - 1.00 per tablet.
- Post-2015: Generic entry led to prices falling by approximately 50%. OTC prices continued to decline with increased market penetration.
Factors Affecting Future Pricing
- Increased commoditization with generics could push prices down further.
- Potential regulatory developments expanding OTC access might increase volume but keep prices suppressed.
- Pricing power remains mainly with traditional brands in regions where regulatory restrictions limit generic competition.
Price Projections (Next 5 Years)
| Year | OTC Price Range (per 10 mg tablet) | Generic Price Range (per 10 mg tablet) | Market Volume Growth | Assumptions |
|---|---|---|---|---|
| 2023 | USD 0.08 - 0.18 | USD 0.07 - 0.14 | +3% | Continued generic expansion |
| 2024 | USD 0.08 - 0.17 | USD 0.07 - 0.13 | +3% | Slight price competition increases |
| 2025 | USD 0.07 - 0.16 | USD 0.07 - 0.12 | +4% | OTC formulations gain market share |
| 2026 | USD 0.07 - 0.15 | USD 0.06 - 0.11 | +4% | Generic pricing stabilizes |
| 2027 | USD 0.07 - 0.14 | USD 0.06 - 0.10 | +2% | Market saturation occurs |
Prices are likely to remain under USD 0.20 per tablet for OTC products, with overall market volume increasing due to OTC switches and expanding indications.
Regulatory and Market Entry Considerations
- OTC Switches: Several countries have approved OTC sales, increasing accessible market size. Further switches could expand volume but pressure on prices.
- Generic Competition: High generic entry capacity leads to price erosion, limiting profit margins.
- Emerging Markets: Growing middle class populations in Asia-Pacific and Latin America drive sustained volume growth, often favoring lower-priced generics.
Summary of Risks and Opportunities
-
Risks
- Heightened generic competition suppressing prices.
- Regulatory barriers delaying OTC status expansion.
- Competitive pressure from newer antihistamines with better efficacy or safety profiles.
-
Opportunities
- Expansion into untapped geographic markets.
- Development of formulations that extend patent life or create new delivery modes.
- Increasing use for pediatric or combination formulations.
Key Takeaways
- The global loratadine market is stable, with a CAGR of nearly 4.8% expected through 2028.
- Prices are declining due to patent expirations and generic penetration, especially in OTC segments.
- Future price range for OTC tablets likely remains under USD 0.20 per 10 mg tablet.
- Market volume will grow from OTC switches and expanding indications, possibly offsetting declining unit prices.
- Companies can capitalize on growth in emerging markets and existing OTC channels if they maintain competitive pricing.
FAQs
Q1: How does patent expiration impact loratadine pricing?
A: Patent expiration in 2015 resulted in widespread generic entry, causing prices to decrease by an estimated 50%. Generic competition now dominates the OTC segment, keeping prices low.
Q2: What regions present the most growth opportunities?
A: Asia-Pacific, Latin America, and parts of Africa have low penetration of loratadine and expanding healthcare infrastructure, providing growth potential.
Q3: Will brand-name loratadine products regain market share?
A: Unlikely in price-sensitive OTC markets; branded products must differentiate via marketing or formulation innovations.
Q4: How do regulatory changes influence future price projections?
A: Approvals for OTC status in new markets could increase sales volume but typically exert downward pressure on unit prices.
Q5: Are there any upcoming formulations or patents that could influence pricing?
A: Currently, loratadine's patent protection has expired; future innovations might involve delivery systems or combination therapies, potentially affecting pricing dynamics.
References
[1] MarketResearch.com (2021). "Global Antihistamine Market Size, Share & Trends Analysis Report."
More… ↓
