Share This Page
Drug Price Trends for HM IBUPROFEN
✉ Email this page to a colleague
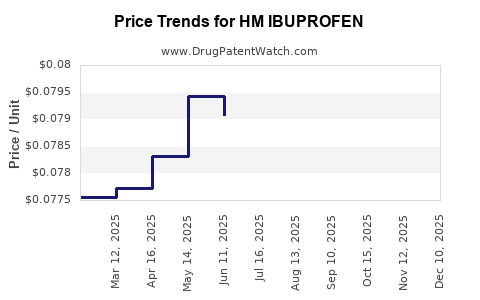
Average Pharmacy Cost for HM IBUPROFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HM IBUPROFEN 200 MG SOFTGEL | 62011-0368-01 | 0.08038 | EACH | 2025-12-17 |
| HM IBUPROFEN 200 MG SOFTGEL | 62011-0368-01 | 0.08020 | EACH | 2025-11-19 |
| HM IBUPROFEN 200 MG SOFTGEL | 62011-0368-01 | 0.07985 | EACH | 2025-10-22 |
| HM IBUPROFEN 200 MG SOFTGEL | 62011-0368-01 | 0.07918 | EACH | 2025-09-17 |
| HM IBUPROFEN 200 MG SOFTGEL | 62011-0368-01 | 0.07975 | EACH | 2025-08-20 |
| HM IBUPROFEN 200 MG SOFTGEL | 62011-0368-01 | 0.07948 | EACH | 2025-07-23 |
| HM IBUPROFEN 200 MG SOFTGEL | 62011-0368-01 | 0.07907 | EACH | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HM IBUPROFEN Market Analysis and Financial Projection
What is the Market Landscape for Ibuprofen (HM IBUPROFEN)?
Ibuprofen is a non-steroidal anti-inflammatory drug (NSAID) primarily used for pain relief, fever reduction, and inflammation control. Market dynamics are driven by generic drug proliferation, OTC availability, and regional regulatory frameworks.
Globally, the NSAID market, including ibuprofen, is valued at approximately $10 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of around 3.5% until 2027[1]. The market's growth is supported by increasing prevalence of chronic pain, growing aging populations, and healthcare policies favoring OTC analgesics.
Major manufacturers include Boots, Himalaya Drug Company, and global brands like Advil and Motrin. New formulations (e.g., liquid gels, sustained-release) and extensions into combination drugs enhance market share.
How is Pricing Structured for HM IBUPROFEN?
Price points for ibuprofen products vary significantly based on formulation, brand, and regional market. Over-the-counter (OTC) products generally cost between $0.05 and $0.20 per tablet or capsule. Prescription formulations, especially higher-dose or specialized delivery systems, are priced higher, typically $1 to $5 per dose.
For branded OTC products, the average retail price in the US is approximately $10 for a bottle of 100 tablets, translating to $0.10 per tablet. Generic versions are generally priced 30–50% lower, around $0.05 to $0.07 per tablet.
Bulk purchasing or wholesale prices tend to be lower, approximately $0.02 to $0.04 per tablet. These variations influence revenue and profit margins across different regions.
What Are Price Projections for HM IBUPROFEN?
Market projections for ibuprofen, including HM IBUPROFEN, suggest stable pricing in the short term, with potential downward pressure due to increased generic competition:
-
OTC prices are expected to remain within the $0.05–$0.20 per tablet range over the next five years, assuming no major formulation innovations or patent protections.
-
Higher-dose prescription formulations may see marginal increases or stabilization at $1–$3 per dose, driven by healthcare policy changes and insurance reimbursements.
-
Volume growth, especially in emerging markets, could offset price declines, maintaining revenue streams for manufacturers.
Regulatory approvals or restrictions could impact pricing. For example, tighter regulation on OTC sales might push consumers toward prescription options, leading to higher prices but reduced volume.
How Will Patent Status Impact HM IBUPROFEN Pricing?
Most ibuprofen formulations are off-patent globally, with exclusive rights expiring nearly two decades ago. Patent expirations foster generic entry, driving prices downward and increasing market competition.
However, if HM IBUPROFEN incorporates novel delivery mechanisms or combines with other APIs, patent protections could extend, supporting higher prices for new proprietary versions.
What Are the Key Market Risks Affecting Price Projections?
-
Regulatory Changes: Stricter OTC regulations or restrictions can limit sales volume, pressuring prices downward.
-
Competition: Entry of generics and biosimilars drives prices lower. Market saturation could diminish profit margins.
-
Supply Chain Disruptions: Raw material shortages or logistics issues can increase production costs temporarily, affecting pricing strategies.
-
Consumer Trends: Growing preferences for natural or alternative therapies may impact demand, influencing pricing.
What Are Competitive Strategies for HM IBUPROFEN?
Manufacturers often adopt one or more of the following:
-
Price Competition: Lower prices to increase market share, especially in price-sensitive regions.
-
Product Differentiation: Develop formulations with faster onset, longer duration, or fewer side effects.
-
Brand Loyalty: Invest in marketing and packaging to maintain consumer preference, even against generics.
-
Expansion into New Markets: Target regions with rising healthcare access, such as Asia-Pacific.
Final Observations
The market for HM IBUPROFEN will remain predominantly competitive, with pricing primarily influenced by generic entry and regional regulatory frameworks. Innovations that extend patent life or offer superior therapeutic profiles could support higher price points temporarily. Long-term, price declines tied to generic competition will be inevitable, balanced by volume growth in emerging markets.
Key Takeaways
-
The global ibuprofen market is valued at $10 billion (2022) with a 3.5% CAGR through 2027.
-
OTC prices range from $0.05–$0.20 per tablet; generics are priced lower, affecting margins.
-
Prices are expected to remain stable short-term, with downward pressure due to increased generic competition.
-
Patent protections are unlikely to influence pricing unless HM IBUPROFEN introduces proprietary formulations.
-
Market risks include regulatory shifts, competition, supply chain issues, and shifting consumer preferences.
FAQs
1. How do patent expirations affect ibuprofen prices?
Patent expirations enable generic manufacturers to enter the market, significantly reducing prices and increasing competition.
2. Will HM IBUPROFEN see price increases from formulation innovations?
Potentially. If HM IBUPROFEN develops proprietary delivery systems or combination drugs, they could command higher prices temporarily.
3. Are regional regulatory differences impacting pricing?
Yes. Strict OTC regulations in certain countries can reduce market penetration and influence price structures.
4. How significant is the market growth in emerging regions?
Emerging markets offer considerable growth potential due to increasing healthcare access and rising demand, which can offset price declines.
5. What is the outlook for premium or branded ibuprofen products?
They may sustain higher prices if they demonstrate clinical advantages or belong to niche segments, but widespread generic competition limits long-term positioning.
References
[1] MarketsandMarkets, "NSAID Market," 2022.
More… ↓
