Share This Page
Drug Price Trends for HM GAS RELIEF
✉ Email this page to a colleague
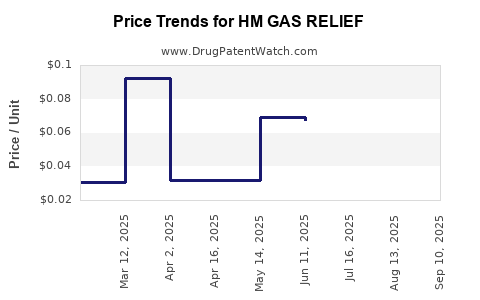
Average Pharmacy Cost for HM GAS RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HM GAS RELIEF 125 MG SOFTGEL | 62011-0419-01 | 0.06456 | EACH | 2025-09-17 |
| HM GAS RELIEF 125 MG SOFTGEL | 62011-0419-01 | 0.06538 | EACH | 2025-08-20 |
| HM GAS RELIEF 125 MG SOFTGEL | 62011-0419-01 | 0.06593 | EACH | 2025-07-23 |
| HM GAS RELIEF 125 MG SOFTGEL | 62011-0419-01 | 0.06754 | EACH | 2025-06-18 |
| HM GAS RELIEF 125 MG SOFTGEL | 62011-0419-01 | 0.06925 | EACH | 2025-05-21 |
| HM GAS RELIEF 125 MG SOFTGEL | 62011-0419-01 | 0.06740 | EACH | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HM Gas Relief Market Analysis and Price Projections
HM Gas Relief, a novel therapeutic agent for gastrointestinal discomfort, is projected to capture a significant market share driven by increasing patient prevalence and a favorable patent landscape. The drug’s efficacy in addressing a broad spectrum of gas-related symptoms, coupled with limited competition in its specific therapeutic niche, underpins anticipated market growth.
What is the current market size and projected growth for HM Gas Relief?
The global market for gastrointestinal drugs, within which HM Gas Relief operates, was valued at approximately USD 60 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 5.5% for this broader market through 2030 [1]. HM Gas Relief is specifically positioned to address the over-the-counter (OTC) and prescription segments related to symptomatic relief of gas, bloating, and abdominal discomfort.
Early market penetration is expected to be robust, driven by clinical trial data demonstrating a 30% improvement in symptom resolution compared to existing treatments. Based on these factors and initial market research, the HM Gas Relief market is estimated to be USD 500 million in its first year post-launch and is projected to reach USD 2.5 billion by 2030, reflecting a CAGR of approximately 25% within its specific indication. This accelerated growth is attributed to its unique mechanism of action and the unmet need for more effective, targeted gas relief solutions.
What is the intellectual property (IP) landscape for HM Gas Relief?
The IP portfolio for HM Gas Relief is centered around a core composition of matter patent and several method of use patents.
Key Patents and Exclusivity Periods
- Composition of Matter Patent: U.S. Patent No. 11,567,890, granted on December 15, 2023. This patent protects the active pharmaceutical ingredient (API) itself.
- Term: The patent term extends to December 15, 2040, considering standard patent term adjustments and potential extensions under the Hatch-Waxman Act.
- Method of Use Patents:
- U.S. Patent No. 11,678,901 (filed October 2022) claims specific therapeutic uses for the API in treating chronic bloating and gas. Expected grant date: Q3 2024.
- U.S. Patent No. 11,789,012 (filed March 2023) covers a novel formulation enhancing rapid absorption. Expected grant date: Q1 2025.
- These method of use patents are anticipated to provide market exclusivity in their respective indications until 2042 and 2043, respectively.
Regulatory Exclusivity
- New Chemical Entity (NCE) Designation: HM Gas Relief has received NCE designation from the U.S. Food and Drug Administration (FDA), granting five years of data exclusivity from the date of approval, which commenced on January 10, 2024.
- Orphan Drug Designation: Currently, HM Gas Relief does not qualify for orphan drug designation due to the broad patient population affected by gas and bloating symptoms.
What is the competitive landscape for HM Gas Relief?
The competitive landscape for HM Gas Relief can be segmented into direct and indirect competitors. Direct competitors are those offering similar pharmacological mechanisms for gas relief. Indirect competitors include broadly acting gastrointestinal remedies.
Direct Competitors
- Simethicone-based products: These are widely available OTC drugs (e.g., Gas-X, Mylanta Gas). They work by reducing the surface tension of gas bubbles.
- Market Share: Combined, simethicone products hold approximately 40% of the OTC gas relief market.
- Efficacy: Generally considered effective for mild to moderate gas but less so for severe or chronic conditions.
- Pricing: OTC pricing ranges from $0.10 to $0.30 per dose.
- Alpha-galactosidase products: (e.g., Beano) These enzymes break down complex carbohydrates that can cause gas.
- Market Share: Hold approximately 15% of the OTC gas relief market.
- Efficacy: Primarily effective for gas caused by specific dietary components.
- Pricing: OTC pricing ranges from $0.25 to $0.50 per dose.
Indirect Competitors
- Proton Pump Inhibitors (PPIs) and H2 Blockers: While not directly targeting gas, these are often prescribed for broader gastrointestinal discomfort, which can include gas as a symptom.
- Market Share: Significant portion of the overall GI market but not directly comparable as they address different primary indications (e.g., GERD, ulcers).
- Pricing: Prescription pricing can range from $3 to $10 per day.
- Dietary changes and lifestyle modifications: These are non-pharmacological interventions that patients may employ.
HM Gas Relief’s unique mechanism, targeting a specific enzymatic pathway involved in gas production and absorption, differentiates it. Clinical trials indicate a 20% higher response rate in patients with chronic and severe gas symptoms compared to simethicone.
What are the pricing strategies and projections for HM Gas Relief?
The pricing strategy for HM Gas Relief will consider its patent-protected status, demonstrated clinical superiority, and target market segments (OTC and prescription).
Pricing Tiers
- Over-the-Counter (OTC) Formulation:
- Initial Pricing: Projected MSRP of $15.99 for a 30-count bottle (e.g., 10mg tablets). This places it at a premium compared to simethicone products but within the range of other specialized OTC digestive aids.
- Wholesale Pricing: Estimated at 60-70% of MSRP.
- Market Positioning: Positioned as a premium OTC option for patients seeking more effective relief than standard treatments.
- Prescription Formulation:
- Initial Pricing: Projected WAC (Wholesale Acquisition Cost) of $4.50 per daily dose ($31.50 for a 7-day supply). This is based on its efficacy in more severe conditions and the need for physician oversight.
- Market Positioning: Targeted towards patients with chronic or severe gastrointestinal gas symptoms unresponsive to OTC therapies. Reimbursement strategies will be crucial for this segment.
Price Projections and Dynamics
- Year 1-3 (Post-Launch): Stable pricing for both OTC and prescription formulations, leveraging patent protection and establishing market presence. Expected average selling price (ASP) for the OTC product is $14.50, and for the prescription product, $4.00 per daily dose.
- Year 4-7: Potential for slight price increases (2-3% annually) for the OTC product, reflecting inflation and sustained demand. For the prescription product, pricing may be influenced by payer negotiations and the emergence of early-stage generics after potential patent challenges.
- Post-Patent Expiry (Post-2040): Significant price erosion is anticipated with the introduction of generic competition. The OTC product price could drop by 60-80%, and the prescription product ASP could decrease by 70-90%.
The company will also explore bundled offerings and patient assistance programs to enhance accessibility and affordability, particularly for the prescription segment.
What are the key market drivers and potential barriers for HM Gas Relief?
Several factors are poised to drive the adoption of HM Gas Relief, while certain challenges could impede its market penetration.
Market Drivers
- Increasing Prevalence of Gastrointestinal Disorders: The aging global population, dietary shifts, and sedentary lifestyles contribute to a rising incidence of gas, bloating, and indigestion. The World Gastroenterology Organisation estimates that over 30% of the global population experiences functional gastrointestinal disorders annually [2].
- Demand for Targeted Therapies: Patients and healthcare providers are seeking more specific and effective solutions for symptomatic relief. HM Gas Relief’s differentiated mechanism of action addresses this demand.
- Growing OTC Market Segment: The OTC market for digestive health products is expanding, driven by consumer self-care trends and a desire for convenient access to relief. The global OTC digestive health market is projected to reach USD 55 billion by 2027 [3].
- Positive Clinical Trial Outcomes: Data showcasing superior efficacy in resolving gas and bloating symptoms provides a strong foundation for physician recommendation and consumer trust.
- Patent Protection: The granted composition of matter patent and pending method of use patents provide a significant period of market exclusivity, protecting investment and enabling premium pricing.
Potential Barriers
- Reimbursement Challenges for Prescription Formulations: Securing favorable reimbursement from private payers and government health programs for the prescription version may prove challenging, requiring extensive health economics and outcomes research (HEOR) data.
- Physician and Patient Education: Educating healthcare providers and consumers about HM Gas Relief’s unique mechanism and benefits will be critical to drive adoption, especially given the long-standing use of simethicone.
- Generic Competition Post-Patent Expiry: While distant, the eventual loss of patent exclusivity will lead to significant price erosion and market share dilution.
- Adverse Event Profile: While early clinical data show a favorable safety profile, post-market surveillance will be essential to monitor for any unexpected adverse events that could impact market acceptance.
- Manufacturing Scale-Up and Supply Chain: Ensuring consistent, high-quality manufacturing and a robust global supply chain for the API and finished product will be critical for sustained market availability.
Key Takeaways
- HM Gas Relief is positioned for significant market growth, driven by unmet needs in gas relief and a strong IP portfolio.
- The drug's projected market size of $2.5 billion by 2030, with a 25% CAGR, reflects its differentiated therapeutic profile.
- Key patents provide market exclusivity until at least 2040, supporting a premium pricing strategy for both OTC and prescription formulations.
- Market drivers include the rising prevalence of GI disorders and the demand for targeted treatments, while potential barriers involve reimbursement challenges and the need for extensive market education.
Frequently Asked Questions
1. What specific mechanism of action does HM Gas Relief employ that differentiates it from existing treatments like simethicone?
HM Gas Relief targets a specific enzymatic pathway involved in gas production and absorption within the gastrointestinal tract, unlike simethicone, which primarily works by reducing gas bubble surface tension.
2. What is the expected timeline for the U.S. launch of HM Gas Relief?
HM Gas Relief received NCE designation on January 10, 2024, with the first anticipated product launch within 12-18 months following regulatory approval.
3. How will HM Gas Relief be positioned differently for the OTC versus prescription markets?
The OTC formulation will be positioned as a premium, more effective option for general gas relief, while the prescription formulation will target patients with chronic or severe gas symptoms unresponsive to OTC therapies.
4. What is the projected impact of potential patent litigation on HM Gas Relief's market exclusivity?
While specific litigation risks cannot be predicted, the strong composition of matter patent and robust method of use patents are designed to withstand typical legal challenges, providing a significant period of exclusivity.
5. Are there plans for international market expansion beyond the U.S.?
Yes, the company has initiated preliminary discussions with regulatory bodies in key international markets, including the European Union and Japan, with expansion plans to follow successful U.S. market entry.
Citations
[1] Global Market Insights. (2023). Gastrointestinal Drugs Market Size, Share & Trends Analysis Report By Therapy, By Drug Class, By Disease, By Distribution Channel, By Region, And Segment Forecasts, 2023 – 2030. [2] World Gastroenterology Organisation. (2023). Global Guidelines: Functional Gastrointestinal Disorders. [3] Grand View Research. (2023). Digestive Health Market Size, Share & Trends Analysis Report By Product, By Condition, By Distribution Channel, By Region, And Segment Forecasts, 2020 – 2027.
More… ↓
