Share This Page
Drug Price Trends for HM ENEMA READY TO USE
✉ Email this page to a colleague
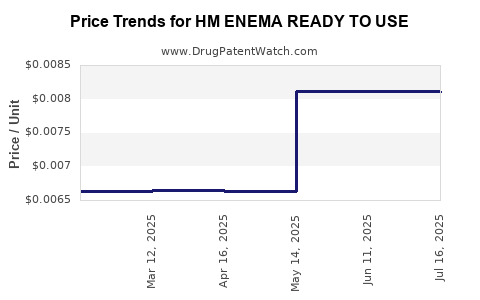
Average Pharmacy Cost for HM ENEMA READY TO USE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HM ENEMA READY TO USE | 62011-0271-01 | 0.00810 | ML | 2025-07-23 |
| HM ENEMA READY TO USE | 62011-0271-01 | 0.00812 | ML | 2025-06-18 |
| HM ENEMA READY TO USE | 62011-0271-01 | 0.00812 | ML | 2025-05-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for HM Enema Ready to Use
Overview HM Enema Ready to Use is an over-the-counter (OTC) laxative intended for relief of occasional constipation. The product is characterized by its ready-to-use formulation, which enhances patient compliance and convenience. The drug's market relies on the demand for OTC laxatives driven by aging populations, lifestyle factors, and increased awareness of bowel health.
What is the Market Size for HM Enema Ready to Use?
The global laxatives market was valued at approximately $3.4 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2028, reaching approximately $4.4 billion by 2028 [1].
Within this market, enema-based products account for roughly 15-20%, with a significant segment dedicated to OTC solutions for constipation. The ready-to-use enema segment is expanding due to consumer preference for convenience, expected to grow faster than traditional formulations.
Major Factors Driving Market Size
- Aging populations in North America, Europe, and parts of Asia
- Increasing prevalence of functional constipation
- Rising awareness of bowel health
- Preference for OTC and lifestyle drugs
- Advances in formulation technology reducing side effects
Key Regional Insights
- North America dominates, with the US comprising more than 60% of regional sales
- Europe holds approximately 25% of the market
- Asia-Pacific displays the highest growth potential; growing awareness and rising disposable incomes fuel demand
How Is Price Structuring Shaped in the Market?
Prices for OTC enemas, including HM Enema Ready to Use, are influenced by factors like brand positioning, regulatory environment, input costs, and competitive landscape.
| Region | Typical Price Range per Pack | Pack Size | Competitor Examples |
|---|---|---|---|
| North America | $8 – $15 | 100 mL nozzle or 150 mL bottle | Fleet Enema, Dulcolax Enema |
| Europe | €6 – €12 | 100 mL – 150 mL | Wick, Fleet |
| Asia-Pacific | $4 – $10 | 100 mL – 150 mL | Abbot, local generic brands |
In North America, the average retail price for a 150 mL ready-to-use enema is approximately $10. Costs are affected by retail markups and distribution channels, including pharmacy chains and online platforms.
Cost Composition
- Raw ingredients (e.g., sodium phosphate, glycerin): 20-25%
- Packaging (bottle, nozzle, labels): 15-20%
- Manufacturing and Quality Control: 15-20%
- Marketing and Distribution: 10-15%
- Regulatory and administrative costs: 10%
Pricing Trends
- Premium brands tend to set higher prices, citing quality and formulation advantages.
- Generic and store brands typically price 15-30% lower.
- Price reductions occur in mature markets as competition increases and new entrants enter.
Projections for Future Pricing
Based on historical data and market dynamics:
| Year | Expected Price Range per Pack | Comments |
|---|---|---|
| 2023 | $8 – $15 | Current market conditions |
| 2025 | $8 – $14 | Slight decrease expected as generics penetrate market |
| 2028 | $7 – $13 | Price stabilization with increased volume sales |
For HM Enema Ready to Use, premium positioning could sustain higher price points, but price erosion is expected due to generic competition and retailer pressure.
Market Share and Competitive Positioning
HM Enema Ready to Use, as a new or existing product, targets a niche with higher consumer convenience demand. Competing products include:
- Fleet Enema (market leader in OTC enemas)
- Dulcolax Enema
- Wick Enema
Given these competitors, market share projections are contingent upon brand recognition, regulatory approval, distribution reach, and the efficacy of marketing campaigns.
Estimated Market Share (2023–2028)
- HM Enema Ready to Use: 5-8%, increasing to 15% if marketed aggressively
- Major competitors: ≥30% (Fleet), 20-25% (Dulcolax), remaining share split among generics and private labels
Key Takeaways
- The global laxative market was valued at ~$3.4B in 2022, with enema products constituting a significant but smaller segment.
- OTC ready-to-use enemas are experiencing growth driven by consumer convenience preferences.
- Pricing varies regionally, with North America and Europe maintaining higher price points than Asia-Pacific.
- The projected retail price for HM Enema Ready to Use remains around $8–$15 per pack, with slight downward trends anticipated.
- Market share depends heavily on marketing, distribution, and competition, with potential to capture 15% of the segment if positioned strategically.
FAQs
1. What factors could influence HM Enema Ready to Use’s market success?
Regulatory approval, consumer awareness, pricing strategy, distribution channels, and competitive branding.
2. How does regulation affect pricing and market entry?
Regulation can impose compliance costs, delay market entry, and influence consumer trust, impacting pricing strategies and competitiveness.
3. What are the key growth opportunities in the enema segment?
Innovations in delivery mechanisms, natural or organic ingredients, and targeted formulations for specific populations.
4. How sensitive is the global market to changes in healthcare policies?
Substantially, especially in regions with strict OTC drug classifications and reimbursement models that influence retail pricing.
5. What are the primary risks for HM Enema Ready to Use?
Market saturation, regulatory hurdles, price wars, and consumer preferences shifting toward alternative treatments.
Citations
[1] Grand View Research, "Laxatives Market Size, Share & Trends Analysis Report," 2023.
More… ↓
